- Title
-
Macrophages protect Talaromyces marneffei conidia from myeloperoxidase-dependent neutrophil fungicidal activity during infection establishment in vivo
- Authors
- Ellett, F., Pazhakh, V., Pase, L., Benard, E.L., Weerasinghe, H., Azabdaftari, D., Alasmari, S., Andrianopoulos, A., Lieschke, G.J.
- Source
- Full text @ PLoS Pathog.
|
T. marneffei infection of zebrafish at 28°C. (A-C) Histological time-course following T. marneffei inoculation of zebrafish, stained by Grocott methanamine silver and Evan’s Blue counterstain (i), with adjacent hematoxylin and eosin-stained sections (ii). mpi, minutes post infection; dpi, days post infection. Red arrowheads indicate fungal conidia. White arrowheads indicate Duct of Cuvier (A) and leukocyte with intracellular fungal elements (C). (D) T. marneffei CFU time-course at 28°C following intravascular inoculation of target dose of approximately 150 fungal conidia (actual dose verified by 0 dpi CFU). Different colors indicate 3 independent experiments, mean±SEM, n≥5 embryos/group/experiment. *p = 0.016, **p = 0.0059, ***p = 0.0003 for statistical comparison between 3 and 4 dpi. (E) Embryo survival following intravascular inoculation in (D). Data are pooled embryos from 3 experiments: n = 474 uninfected, 339 infected. NS: not significant. |
|
T. marneffei infection of zebrafish at 33°C. (Ai-iv) Histology of infected zebrafish embryos at different stages of T. marneffei infection. Sections were stained with Grocott methanamine silver to visualize fungi and counterstained with Nuclear Fast Red. Scale bar: 10 μm. (i) Phagocytosed conidium (arrowhead) within host leukocyte in the circulation at 20 minutes post infection. (ii) At 2 dpi, two fungal morphologies were observed: intracellular fungi assumed typical yeast morphology (upper arrowhead), while extracellular fungi displayed an elongated form (lower arrowhead). (iii) Yeast morphology of T. marneffei within a circulating infected leukocyte (arrowhead) at 3 dpi. (iv) Granuloma formation at 4 dpi. (B)FACS-purified, cytospun, T. marneffei-laden leukocytes prepared from infected embryos at 4 dpi, incubated at 33°C during infection. Grocott methanamine silver with Nuclear Fast Red counterstain. (i and ii) show neutrophils laden with hyphal forms (empty arrowheads), representative of all neutrophils observed. (iii) shows two macrophages laden with yeast forms (full arrowheads), the dominant morphology observed. (iv) shows a macrophage containing fungal cells of both yeast and hyphal morphologies. Scale bar: 10 μm. (C) Three representative CFU time-courses of infection at 33°C. In contrast to infections at 28°C (Fig 1D), no consistent decrease in CFU was observed at 4 dpi. Data are mean±SD of 3 independent replicates, n = 5 embryos (pooled)/timepoint/experiment. (D) Kaplan-Meier life table analysis for the treatment groups shown in (E). Significantly reduced survival occurred in infected groups compared to uninfected groups. n = 297 for uninfected, 319 for infected. P-value from Gerhan-Breslow-Wilcoxon Test. (E) Measurement of neutrophil populations during infection using Neutrophil Units. A significant increase in neutrophil population size occurred in the infected group compared to uninfected group over 1–4 dpi. Data are mean+SEM from 3 independent experiments. n = 5 embryos/group/timepoint in each experiment. P-values from unpaired two-tailed t-test. |
|
Zebrafish mount a Csf3r-dependent granulopoietic response to T. marneffei infection. (A) Low magnification images of infected and uninfected whole Tg(mpx:EGFP) embryos at 0, 3 and 4 dpi, displaying the profound infection-driven increase in EGFP-expressing neutrophil numbers. (B, C) Neutrophil (B) and macrophage (C) population expansion in infected versus uninfected embryos from 0–4 dpi. These data derive from enumerating neutrophil and macrophage numbers in the same embryos, of genotype Tg(mpx:EGFP/mpeg1:Gal4FF/UAS-E1b:Eco.nfsB-mCherry). Data displayed are mean±SD, n = 4 embryos/group. These data replicate other data derived from separately scoring these leukocyte populations in different embryos, in n≥2 independent other experiments. (D) Neutrophil population sizes at 4 dpi represented by Neutrophil Units in embryos injected with antisense morpholino oligonucleotides targeted to knockdown csf3-receptor (csf3r-MO) or interleukin-6 receptor (il6ra-MO and gp130-MO) Data are mean±SEM, n = 10 embryos/group/experiment, n≥3 experiments. Intragroup +/- infection p-values from 2-tailed unpaired t-test; p-values from intergroup comparisons involving csf3r-MO from 1-tailed unpaired t-test, given the a priori expectation of a reduction in neutrophil values in csf3r-deficiency [65, 95] (E) Kaplan-Meier life table analysis for the treatment groups shown in (C). Numbers/group (uninfected/infected): Control MO, 474/326; csf3r-MO, 443/233; il6ra-MO, 462/330; gp130-MO, 465/313. P-values from Gerhan-Breslow-Wilcoxon Test. |
|
Macrophages dominate T. marneffei phagocytosis during infection establishment. (A, B) Confocal imaging of leukocytes and calcofluor-stained acuD:RFP fungal conidia in Tg(mpx:EGFP) fluorescent leukocytes. (A) shows a germinated, RFP-expressing, blue calcofluor-stained conidium (red arrow) within a non-fluorescent vacuole of a bright EGFPhigh neutrophil. Z-stack sections (i-iii) orientated as in schematic diagram. (B) shows a dull EGFPlow macrophage laden with multiple RFP-expressing conidia. (C,D) Phagocytosis of calcofluor-stained fungal conidia by a macrophage (C) and neutrophil (D) in Tg(mpeg1:mCherry/mpx:EGFP) embryo (C) inoculated in a somite. The still images are from S1 and S2 Movies respectively. The macrophage extends a long process towards the conidium (red arrowhead); conidial phagocytosis is followed by macrophage movement over the site of phagocytosis and out of the field. In each panel, maximum projection z-stack image is supplemented by xz and yz projections, orientated as in the schematic diagram. (E-G) Confocal imaging of intracellular calcofluor-stained conidia within a neutrophil (E) and macrophage (F) of a Tg(mpeg1:mCherryCAAX/mpx:EGFPCAAX) embryo, with dimensional projections as in (C). In each case, the lower right yellow-boxed panel is a detail of the yellow-boxed area in the upper left xy maximum intensity projection, displaying the cross-section (white dashed line) for which a fluorescence intensity channel profile is shown in G. Arrowheads indicate peaks of fluorescence corresponding to neutrophil (Gi, green) and macrophage (Gii, red) cellular membranes. (H) Enumeration of conidia phagocytosis (i) by macrophages (red lines) and neutrophils (green lines) reflects higher macrophage recruitment (ii) following somite infection. Lines represent individual infected zebrafish followed over time. N = 10 embryos/group imaged on n = 8 different days. P-values from Mann-Whitney test: ns = not significant, * = <0.05, ** = <0.01, *** = <0.001, **** = <0.0001. |
|
Neutrophils and macrophages play opposing roles during establishment of T. marneffei infection. (A) Representative images of 52 hpf Tg(mpeg1:mCherry/mpx:EGFP) embryos injected with antisense morpholino oligonucleotides to perturb the balance of neutrophil and macrophage populations. (B) T. marneffei CFU numbers at 24 hpi corresponding to the aligned treatment groups in panels (Ai-iv), for wild-type (WT) and myeloperoxidase-deficient (mpx-/-) genotypes as shown. Data are mean±SEM for n≥3 experiments, n≥5 embryos/group/experiment. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Depletion of the macrophage niche increases conidial destruction during establishment of infection. (A) Ablation of macrophages decreases conidial viability in the first 24 hpi. (i) and (ii) show representative images of nitroreductase-dependent metronidazole-mediated macrophage ablation following treatment of transgenic embryos (ii) versus untreated control embryos (i). Ablation efficiency is quantified in (S9A Fig). (B) T. marneffei CFU numbers at 24 hpi in macrophage-replete control (Tg(mpeg1:Gal4FF/UAS-E1b:Eco.nfsB-mCherry) negative, treated with 10 mM metronidazole) compared to macrophage-depleted (Tg(mpeg1:Gal4FF/ UAS- E1b:Eco.nfsB-mCherry) positive, treated with 10 mM metronidazole). (C) Ablation of neutrophils increases conidial viability in the first 24 hpi. (i) and (ii) show representative images of Nitroreductase-dependent metronidazole-mediated neutrophil ablation in treated (ii) versus diluent-treated control embryos (i). Ablation efficiency is quantified in (S9B and S9C Fig). (D) T. marneffei CFU numbers at 24 hpi in neutrophil-replete control (Tg(mpx:KalTA4/UAS-E1b:Eco.nfsB-mCherry) negative, treated with 10 mM metronidazole) compared to neutrophil-depleted (Tg(mpx:KalTA4/ UAS- E1b:Eco.nfsB-mCherry) positive, treated with 10 mM metronidazole) Data are mean±SEM, n≥5 embryos/group/experiment, n≥3 experiments. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Additional features of zebrafish T. marneffei infection at 28°C. (A-C) Maximum intensity projection confocal fluorescence z-stacks depicting stages of acuD:RFP T. marneffei infection in Tg(mpx:EGFP) zebrafish, allowing observation of fluorescent leukocytes, with calcofluor pre-staining allowing visualization of fungal conidia and RFP expression demonstrating their germination. Scale bars: 20 μm. dpi, days post infection. (A) Germination of conidia at 1 dpi with extension of RFP-positive germ tubes from extracellular calcofluor-stained conidia (arrowhead) adjacent to Tg(mpx:EGFP) leukocytes. (B) Destruction of fungal cells identified by RFP-positive debris within a neutrophil vacuole at 2 dpi (boxed). (C) Filamentous fungal cell growth (arrowhead) stretches and ruptures some leukocytes at 2 dpi. (D-F) Histology of infected zebrafish at different stages of T. marneffei infection. Fungal cells are stained black with Grocott’s methenamine silver stain against an Evan’s Blue counterstain (i, left panels). Tissues are visualized by hematoxylin and eosin staining of adjacent sections (ii, right panels). Scale bars: 20 μm. (D) Early granuloma formation at 2 dpi with accumulation of leukocytes around an infection focus (red arrowhead). (E) Infection focus in the brain at 3 dpi (red arrowhead). (F) Organising granuloma at 4 dpi, with epithelioid leukocytes surrounding a necrotic centre containing fungal debris (lower red arrowhead). Infected leukocyte nearby (upper red arrowhead) suggests dissemination of infection from the granuloma by leukocytes. (G) Low-power fluorescence image superimposed on brightfield image, showing granuloma formation at 4 dpi in tissue adjacent to the caudal hematopoietic tissue (CHT). Fluorescent Tg(mpx:EGFP) leukocytes (white arrowhead) have accumulated around a focus of germinated acuD:RFP T. marneffei cells (red arrowhead). (H) Maximum intensity projection of confocal z-stack showing accumulation of EGFP positive leukocytes around a focus of RFP-expressing germinated T. marneffei. Invasive filamentous growth (red arrowhead) radiates from the margins of the granuloma centre (white arrowhead). Scale bar: 20 μm. (A-C) Maximum intensity projection confocal fluorescence z-stacks depicting stages of acuD:RFP T. marneffei infection in Tg(mpx:EGFP) zebrafish, allowing observation of fluorescent leukocytes, with calcofluor pre-staining allowing visualization of fungal conidia and RFP expression demonstrating their germination. Scale bars: 20 μm. dpi, days post infection. (A) Germination of conidia at 1 dpi with extension of RFP-positive germ tubes from extracellular calcofluor-stained conidia (arrowhead) adjacent to Tg(mpx:EGFP) leukocytes. (B) Destruction of fungal cells identified by RFP-positive debris within a neutrophil vacuole at 2 dpi (boxed). (C) Filamentous fungal cell growth (arrowhead) stretches and ruptures some leukocytes at 2 dpi. (D-F) Histology of infected zebrafish at different stages of T. marneffei infection. Fungal cells are stained black with Grocott’s methenamine silver stain against an Evan’s Blue counterstain (i, left panels). Tissues are visualized by hematoxylin and eosin staining of adjacent sections (ii, right panels). Scale bars: 20 μm. (D) Early granuloma formation at 2 dpi with accumulation of leukocytes around an infection focus (red arrowhead). (E) Infection focus in the brain at 3 dpi (red arrowhead). (F) Organising granuloma at 4 dpi, with epithelioid leukocytes surrounding a necrotic centre containing fungal debris (lower red arrowhead). Infected leukocyte nearby (upper red arrowhead) suggests dissemination of infection from the granuloma by leukocytes. (G) Low-power fluorescence image superimposed on brightfield image, showing granuloma formation at 4 dpi in tissue adjacent to the caudal hematopoietic tissue (CHT). Fluorescent Tg(mpx:EGFP) leukocytes (white arrowhead) have accumulated around a focus of germinated acuD:RFP T. marneffei cells (red arrowhead). (H) Maximum intensity projection of confocal z-stack showing accumulation of EGFP positive leukocytes around a focus of RFP-expressing germinated T. marneffei. Invasive filamentous growth (red arrowhead) radiates from the margins of the granuloma centre (white arrowhead). Scale bar: 20 μm. (TIF) |
|
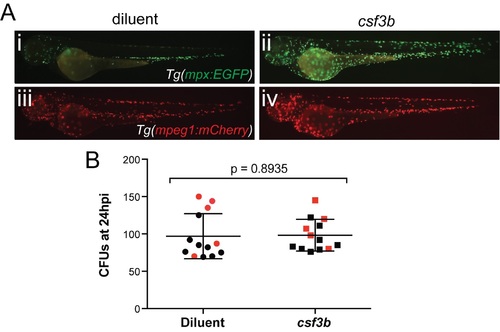
Expansion of neutrophils by csf3b overexpression does not enhance fungal clearance. |
|
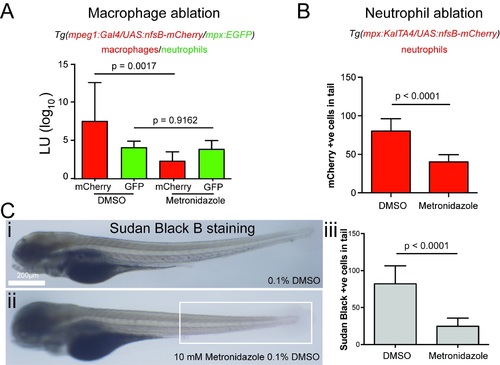
Quantification of nitroreductase-mediated leukocyte ablation. |