- Title
-
Stable vascular connections and remodeling require full expression of VE-cadherin in zebrafish embryos
- Authors
- Montero-Balaguer, M., Swirsding, K., Orsenigo, F., Cotelli, F., Mione, M., and Dejana, E.
- Source
- Full text @ PLoS One
|
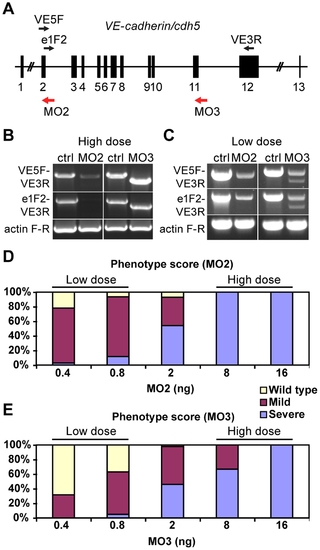
VE-cadherin morpholinos downregulate VE-cadherin expression in a dosage dependent manner. (A) Schematic representation of zebrafish VE-cadherin/cdh5 gene with exons represented as black boxes. The positions of the morpholinos employed in this study are indicated with red arrows, and the primers used for the analysis of splice variants as black arrows (VE5F, e1F2 and VE3R). (B–C) Expression of VE-cadherin was analyzed by RT-PCR using two sets of primers. (B) Upon injection of high doses (16 ng) of VE-cadherin morpholinos, aberrant splicing of VE-cadherin was observed for both MO2 and MO3 when using primer pair VE5F-VE3R. Primer pair e1F2-VE3R allowed the detection of endogenous wild type VE-cadherin transcript in controls (Ctrl) and proved the absence of this transcript in MO2 injected embryos (left panel in B). This primer pair confirmed the altered splicing for MO3 injected embryos (right panel in B). Actin was used as an internal control. (C) Injection of low doses of morpholino (0.8 ng) caused partial knockdown of VE-cadherin. These embryos expressed both wild type VE-cadherin and aberrant spliced transcripts. (D–E) Histograms show the quantification of the effect of VE-cadherin morpholinos at 54 hpf. Both MO2 (D) and MO3 (E) cause increased severity of the phenotype increasing the doses injected. With low doses (0.8 ng) most of the embryos present circulation and hemorrhages (mild phenotype). Conversely, injection of high doses of either one morpholino (16 ng) cause a highly penetrant phenotype, with complete absence of blood circulation (severe phenotype). Intermediate amounts of morpholino lead to both mild and severe phenotype in different percentages. MO2 presented higher penetrance than MO3. |
|
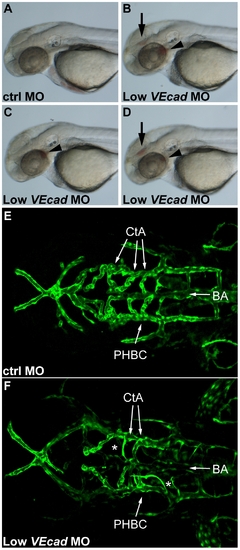
Partial VE-cadherin knockdown causes vascular instability and hemorrhages. (A–D) Bright field images of 52 hpf embryos. Lateral views, anterior to the left. Embryos injected with low doses (0.8 ng) of VE-cadherin morpholino (Low VEcadMO) develop single (C) or multiple hemorrhages (B and D) that were evident at 52 hpf and appeared in the brain (arrows) and/or aortic arches (arrowheads). No or very rare hemorrhages were observed in embryos injected with mismatch control morpholino (ctrlMO) at the same concentration (A). (E, F) Confocal images of Tg(flk1:EGFP) embryos at 54 hpf. Dorsal views, anterior to the left. In embryos injected with ctrlMO (E), central arteries (CtA) extend from the basilar artery (BA) towards forebrain and midbrain, and drain into the primordial hindbrain channel (PHBC). In contrast, in Low VEcadMO injected embryos (F) only few CtAs are formed, and those which are present fail to fuse to BA and lack lumen (asterisks). |
|
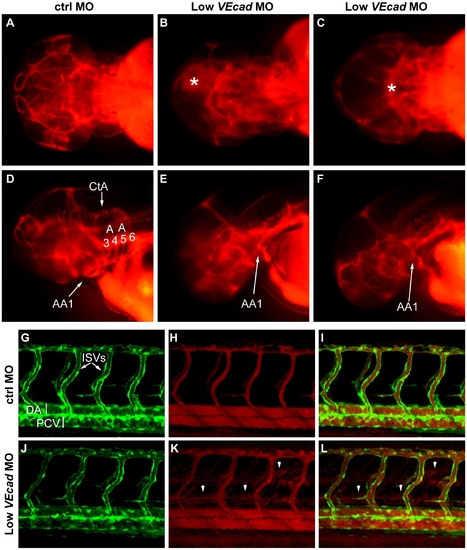
Partial VE-cadherin knockdown affects cranial and trunk vessel integrity. (A–F) Microangiographies performed at 52 hpf. (A–C) Dorsal views, (D–F) lateral views of the embryos above, anterior to the left. Control embryos (A, D) have a functional and complex cranial vascular network with lumenized aortic arches (AA) and central arteries (CtA). Microangiograms of embryos injected with low dose (0.8 ng) of VE-cadherin morpholino that developed hemorrhages (Low VEcadMO) show defects in head perfusion. B, E and C, F correspond to two different embryos with the same treatment. Extravasation of the injected dextran is observed at the site of the hemorrhages (asterisk in B and C). In addition, only the first aortic arch (AA1) is fully formed in the mild morphants but aortic arches 3 through 6 do not exhibit circulation (see panels E, F). (G–L) Microangiographies performed in Tg(flk1:EGFP) embryos at 54 hpf. Lateral views of trunk vessels are shown. (G, J) EGFP positive vessels, (H, K) microangiograms performed with rhodamine-dextran, and merge images (I, L). In control embryos (G–I) intersegmental vessels (ISVs), dorsal aorta (DA) and posterior cardinal vein (PCV) are fully lumenized and the injected dextran remains inside this primary vascular network. In some VE-cadherin mild morphants (Low VEcadMO) these vessels are apparently normal (J) but leakage is observed in the somites and regions surrounding the ISVs (arrowheads in panels K,L). |
|
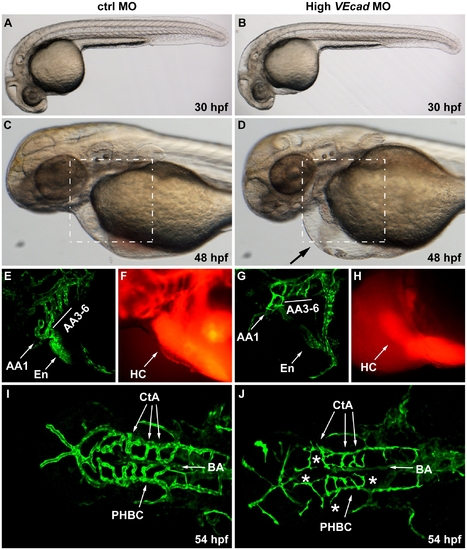
Complete VE-cadherin abrogation impairs the formation of a functional vascular network. (A–D) Bright field images of embryos injected with 16 ng of ctrlMO and VE-cadherin morpholino (High VEcadMO). VE-cadherin severe morphants present a normal morphological development at 30 hpf (B) but blood circulation is not established (see movies in supplementary material). At 48 hpf lack of circulation persists in this morphants with severe phenotype, and pericardial edema is observed (arrow in panel D). (E–H) Views of the endocardial and aortic arch region (highlighted box in C and D) at 52 hpf. Endocardium (En) and aortic arch vessels (AA) are visualized using a flk1:EGFP background (E, G) while microangiograms show interconnected lumenized vessels filled with rhodamine-dextan (F). VE-cadherin severe morphants form endocardium (En in panel G) that is not openly connected to the vasculature, neither to the aortic arch 1 (AA1) nor to the CCV, and retains the microinjected dextran in the heart chambers (HC). Rudiments of AAs without lumen are present in VE-cadherin morphants (G). (I, J) Dorsal views of Tg(flk1:EGFP) embryos at 54 hpf. In embryos injected with high dose of VE-cadherin morpholino (J) cranial vessels are apparently in place but central arteries (CtA) fail to form the connection to the basilar artery (BA), do not correctly lumenize and small sprouting vessels are present (asterisks). |
|
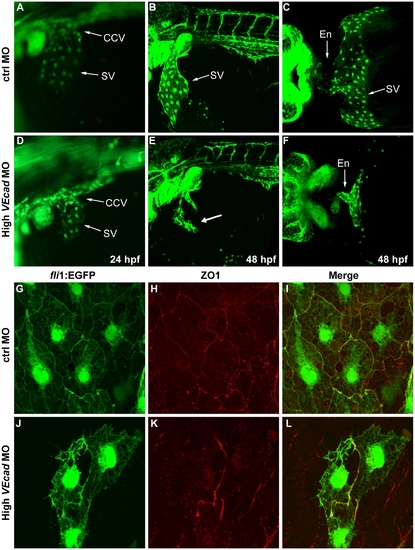
Depletion of VE-cadherin affects the formation of the sinus venosus. (A–F) Fluorescence images of fli1:EGFP embryos at the level of the common cardinal vein (CCV) and sinus venosus (SV). At 24 hpf a number of endothelial cells expand out of the CCV to form the SV both present in control embryos (A). In VE-cadherin severe morphant embryos (High VEcadMO) a rudiment of SV appears at 24 hpf (D). At 48 hpf the SV is fully formed in control embryos (B) while in high VEcadMO only few endothelial cells are present (arrow in panel E). Ventral views of fixed embryos allow the visualization of the endocardium (En) connected to a fully formed SV in control embryos (C). In contrast, high VEcadMO do have a tubular endocardium (En in panel F) but the SV is not formed. The absence of this structure impairs the connection of the CCV to the heart tube and the closure of the circulatory loop. (G–L) Immunofluorescence analysis by confocal microscopy of endothelial cells from the SV at 48 hpf. Fli1:EGFP positive endothelial cells visualized in G and J present intercellular junctions, marked by antibody staining against ZO1 (H, K). The few endothelial cells present in this SV region in high VEcadMO injected embryos show an aberrant morphology, but form some junctions at the sites of contact. |
|
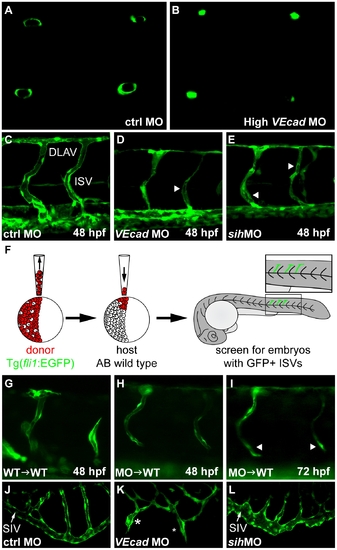
VE-cadherin expression influences lumen formation. (A–B) Optical cross sections of intersegmental vessels (ISVs) at 48 hpf in Tg(fli1:EGFP) background acquired at the confocal microscope by positioning the embryo on a dorsal orientation. CtrlMO injected embryos have fully lumenized ISVs (A) while VE-cadherin severe morphants (High VEcadMO) do not (B). (C–E) Lumenization of ISVs is influenced by blood circulation. Lateral views of trunk vessels of ctrlMO (C), high VEcadMO (D) and sihMO (E) injected embryos at 48 hpf. At this stage, ISVs and the dorsal longitudinal anastomotic vessels (DLAV) are fully lumenized in control embryos. In contrast, VE-cadherin and sih morphants present only a very small lumen in some portions of the ISVs (arrowheads in D and E) and essentially no lumen in DLAV. (F–I) Transplantation experiments. (F) Scheme of the experimental procedure. Donor cells from fli1:EGFP embryos were transplanted into AB wild type embryos at 4 hpf and, after development, embryos with EGFP positive ISVs were selected for analysis. When wild type embryos were used as donor (G), ISVs did form lumen and were integrated into the host vasculature at 48 hpf. Morphant donor cells transplanted into wild type embryos form thin lumens at 48 hpf (H) that fail to correctly integrate in the host vasculature and possibly collapse at 72 hpf (arrowheads in panel I). (J–L) Lumenization of subintestinal vein (SIV) is independent of blood flow as it occurs in the absence of heart beat in sih morphants at 72 hpf (L) but is dramatically affected by the absence of VE-cadherin. High VEcadMO SIVs display only partially lumenized regions which are not fully interconnected (asterisks in panel K). |
|
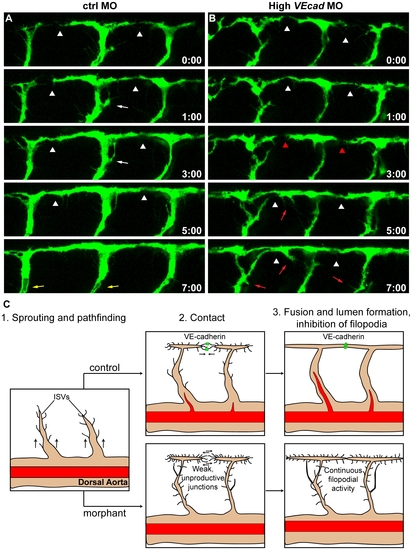
VE-cadherin is required for correct vascular connection and fusion. Time lapse analyses of ISVs of ctrl and high VEcadMO injected embryos from 28 to 36 hpf, performed in a Tg(fli1:EGFP) background. Lateral views of the trunk vessels are shown at the indicated time points (see Movies S7 and S8 for full time lapse). In ctrlMO injected embryos (A), dorsal extensions of ISVs that are about to form the DLAV (arrowheads) get stabilized soon after making contact with neighbouring vessels. In addition, the filopodial activity decreases once the connections are formed and stops before lumenization (white arrows). See lumen in ISVs at time point 7:00 (yellow arrows). In contrast, vessels of VE-cadherin severe morphants (B) show defects in establishing stable connections, as they make contact (arrowheads in B) but detach afterwards (red arrowheads). Moreover, the filopodial activity persists in ISVs even once they already appear dorsally interconnected (red arrows). (C) Model of the filopodial sprouting behaviour of control and VE-cadherin severe morphant ISVs. |
|
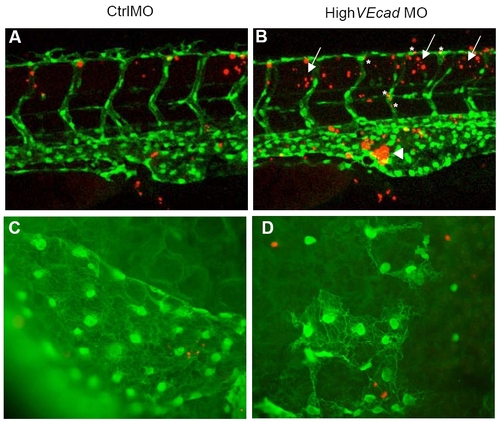
Endothelial cell death is not increased in VE-cadherin endothelial cells. (A–D) TUNEL assay of 48 hpf embryos in a fli1:EGFP background allows the colocalization analysis of apoptotic cells (in red) and endothelial cells (in green). (A–B) Confocal images of the trunk region at the level of the urogenital opening are shown. VE-cadherin severe morphants (B) exhibit increased apoptosis in the blood island (arrowhead) where the blood cells and hematopoietic precursors (some of them fli1:EGFP positive) accumulate in the absence of circulation (arrowhead). In addition, increase in the number of apoptotic cells is observed in the tissues surrounding the ISVs and DLAV (arrows). Few vascular endothelial cells (asterisks) show increase in TUNEL staining. (C–D) Fluorescence microscopy images of the endothelial cells in the sinus venosus. |
|
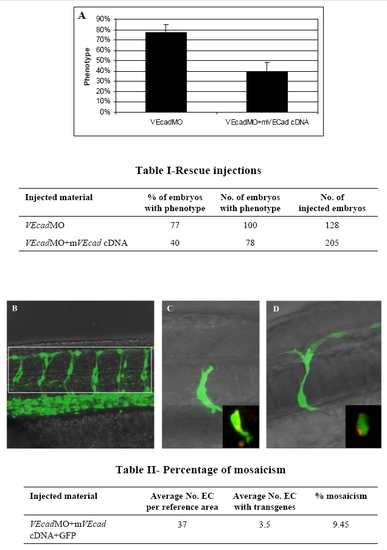
Endothelial specific expression of murine VE-cadherin rescues the phenotype of VE-cadherin morphants. A plasmidic DNA construct containing murine VE-cadherin cDNA under the zebrafish fli1 enhancer/promoter was coinjected with 0.4 ng of MO2 in order to rescue the hemorrhagic phenotype observed in the mild morphants. Three independent experiments were performed and the phenotype of morphant and rescued embryos was examined under the microscope at 54 hpf. The graph in panel A shows the mean percentage of embryos showing a morphant phenotype when injected with morpholino (VEcadMO) or morpholino plus plasmid (VEcadMO+mVEcad cDNA). The number of embryos analyzed is shown in table I. (B–D) Confocal images. Percentage of mosacism in 6 pairs of intersegmental vessels (ISV) located in the area of the trunk shown in figure B is displayed in table II. Average number of endothelial cells (EC) in 12 ISV+DLAV per embryo (at 48 hpf, n = 12, range 34 to 40) was counted in the Tg(fli1:nEGFP)y7 transgenic line, which shows EGFP-positive EC nuclei. Average number of EC expressing the transgenes was counted in AB embryos injected with a mixture of VEcadMO and cDNAs as indicated in the table. ECs expressing EGFP were counted in the same reference area at 48 hpf (n = 40, range 1 to 10). Percentage of mosaicism indicates the average proportion of EC cells expressing the transgene in 12 ISV+ DLAV. Figures C and D show mosaic expression of the EGFP transgene in ECs. Insets show co-expression of EGFP and murine VEcadherin:mCherry in the same ECs. |
|
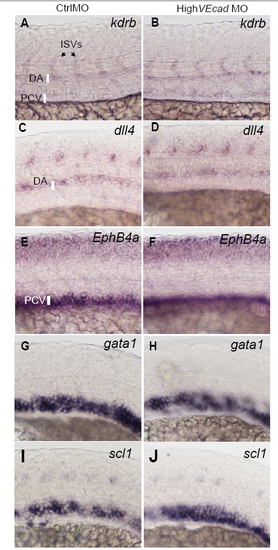
VE-cadherin knockdown does not affect vascular gene expression. In situ hybridization with pan-endothelial (A–B), arterial (C–D), venous markers (E–F) and hematopoietic precursor markers (G–J). Lateral views of the trunk region of 20 hpf embryos are shown, except A, B that report 24 hpf. Embryos injected with high dose (8 ng) of control or VE-cadherin morpholinos (high VEcadMO) were hybridized with the indicated probes (kdrb, dll4, ephB4a, gata1, scl1). High VEcadMO injected embryos (B, D, F, H, J) did not present differences in the expression of these markers when compared to control injected embryos (A, C, E, G). |