- Title
-
RhoC maintains vascular homeostasis by regulating VEGF-induced signaling in endothelial cells
- Authors
- Hoeppner, L.H., Sinha, S., Wang, Y., Bhattacharya, R., Dutta, S., Gong, X., Bedell, V.M., Suresh, S., Chun, C.Z., Ramchandran, R., Ekker, S.C., Mukhopadhyay, D.
- Source
- Full text @ J. Cell Sci.
|
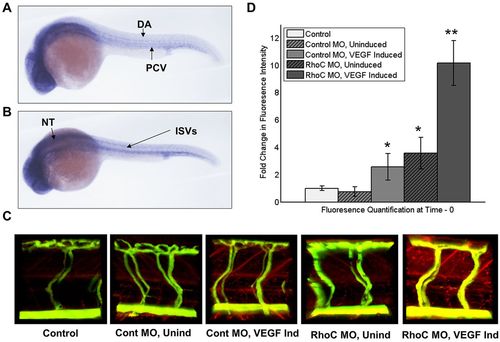
RhoC negatively regulates VEGF-induced vascular permeability in zebrafish. (A,B) Using zebrafish cDNA, a probe to zebrafish RhoC (Rhoad) was created and in situ hybridization was performed on 24hpf zebrafish embryos. Multiple images were captured using a Zeiss Axioplan 2 microscope and overlayed in Photoshop, such that areas of focus were unmasked, to generate a composite image. Lateral (A) and superior (B) views are shown. DA, dorsal aorta; PCV, posterior cardinal vein; ISVs, anterior intersomitic vessels; NT, neural tubes. (C) Microangiography was performed on anesthetized 3dpf zebrafish embryos by injecting FITC–dextran (2000kDa) and Texas-Red–dextran (70kDa), VEGF was induced through heat exposure (when applicable), and extravasation of red tracer as a measure of zebrafish vascular permeability was live imaged using a ZEISS LSM 780 confocal microscope. Control, no MO injection and no VEGF induction; Cont MO, control MO injection; RhoC MO, Rhoad and Rhoae MO injection; Unind, no VEGF induction; VEGF, heat induction of VEGF transgene. (D) Quantification of extravasated red tracer. **P<0.05 (RhoC MO, VEGF Induced versus Control MO, VEGF Induced); *P<0.05 (RhoC MO, Uninduced versus Control; Control MO, VEGF Induced versus Control) (paired two-tailed Student′s t-test). Results are mean±s.d. (n=??). EXPRESSION / LABELING:
|
|
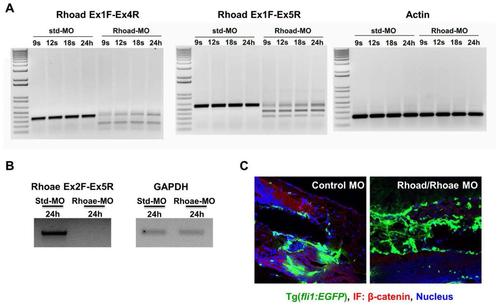
MO-mediated RhoC ablation decreases β-catenin in the vessels of zebrafish. (A-B) RhoC (Rhoad/Rhoae) or control (std) MOs were injected into 1-2 cell stage Tg(Fli:GFP) zebrafish embryos. Embryos were collected at the indicated times, RNA was collected using an RNeasy Plus Mini Kit (Qiagen) and converted to cDNA, PCR was performed using the indicated oligonucleotide pairs, and visualized after running on an agarose gel. (A) Oligonucleotides were used to amplify Rhoad Exon 1-4 (A, left), Rhoad Exon 1-5 (A, middle) and loading control β-actin (A, right). (B) Oligonucleotides were used to amplify Rhoae Exon 2-5 (B, left) and loading control GAPDH (B, right). (C) RhoC (Rhoad/Rhoae) or control MOs were injected into 1-2 cell stage Tg(Fli:GFP) zebrafish embryos. At 3 dpf, the zebrafish were formalin-fixed, placed in OCT, frozen, sectioned with a microtome onto slide, stained with a primary β-catenin antibody followed by a corresponding secondary Alexa-Fluor 568 (red) antibody, and visualized using a Zeiss LSM 780 confocal microscope. Green: Tg(Fli:GFP); Red: β-catenin; Blue: DAPI-stained nuclei. |