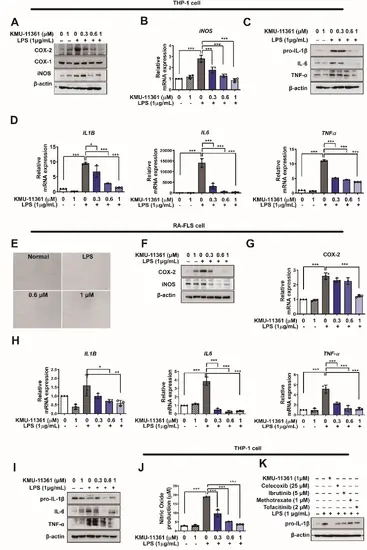
Fig. 5
|
KMU-11361 suppresses COX-2, NO, and pro-inflammatory mediator activity in human macrophages and RA-FLS cells. THP-1 cells were differentiated into macrophages by treatment with PMA (100 nM) for 24 h. THP-1 and RA-FLS cells were pretreated with varying concentrations of KMU-11361, then stimulated with LPS (1 µg/mL) for the designated time points. A, C Whole-cell lysates of THP-1 cells were isolated and used to measure the protein expression levels of COX-1, COX-2, iNOS, IL-1β, IL-6, and TNF-α by western blotting. B, D In THP-1 cells, total RNA was extracted and used to evaluate the mRNA expression levels of pro-inflammatory cytokine (IL1B, IL6, TNFa and iNOS, respectively. *p < 0.05, ***p < 0.001. E The effect of KMU-11361 on cell proliferation and the morphological changes in human RA-FLS cells were visualized under a microscope (×100). F, I RA-FLS cell lysates were isolated and used to measure the protein expression levels of COX-2, iNOS, IL-1β, IL-6, and TNFα by western blotting. G, H In human RA-FLS, total RNA was extracted and used to evaluate the mRNA expression levels of pro-inflammatory cytokine (IL1B, IL6, TNFa and COX-2, respectively, *p < 0.05, **p < 0.01, ***p < 0.001. J RAW 264.7 cells were pretreated with the indicated concentrations of KMU-11361 for 1 h, followed by stimulation with LPS (1 µg/mL) for 24 h. Nitric oxide production was measured using Griess reagent. ***p < 0.001. K Cells were then pretreated with KMU-11361 (1 µM), celecoxib (25 µM), ibrutinib (5 µM), methotrexate (1 µM), or tofacitinib (2 µM), followed by stimulation with LPS (1 µg/mL) for 6 h. Whole-cell lysates were collected after 1 h, and the protein expression levels of pro-IL-1β were analyzed by western blotting. Values in the graph indicate the mean ± SD of three independent experiments (n = 3). The expression level of β-actin protein was used as the loading control |