Fig. 3
|
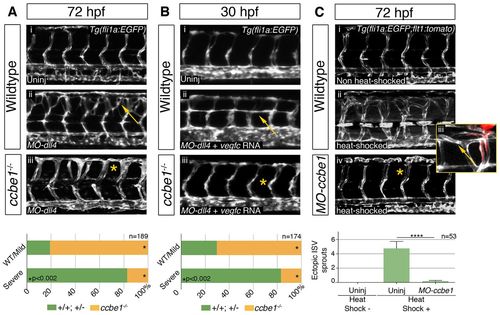
Phenotypes driven by ectopic Vegfc/Vegfr3 signaling are suppressed in ccbe1-deficient embryos. (A) At 72 hpf, dll4 morphants display an arterial hyperbranching phenotype (arrow) driven by increased Vegfc/Vegfr3 signaling in the transgenic Tg(fli1a:EGFP) line. This phenotype was suppressed in ccbe1hu3613 mutants. Eighty-one per cent of MO-dll4 injected embryos displaying wild-type or mild phenotypes were ccbe1 mutants (n=17/21), whereas the population displaying the most severe phenotype was mainly composed of wild-type or heterozygous siblings (83%; n=139/168). (B) In dll4 morphants, arteries are sensitized to increased vegfc expression during primary sprouting. Arteries in MO-dll4, vegfc mRNA-injected embryos display aberrant, ectopic turning (arrow) as early as 30 hpf. Embryos from ccbe1 carrier incrosses, injected with 100 ng vegfc mRNA and 5 ng MO-dll4, were sorted into the phenotypic categories ‘wild type’ and ‘severe’. Genotyping revealed that 70% of the embryos displaying wild-type morphology were ccbe1 mutants (n=19/27). By contrast, the population affected by the most severe phenotype was composed of 83% wild-type or heterozygous siblings (n=122/147). (C) Confocal projections of Tg(fli1a:EGFP; flt1:tomato; hsp70l:Gal;4XUAS:vegfc) embryos show that endothelial cells in heat-shocked embryos display aberrant ectopic branching at 72 hpf. The ectopic endothelial cells are venous derived (flt1:tomato negative, arrow in Ciii). Heat-shocked embryos that were injected with 2.5 ng of MO-ccbe1 do not show this phenotype (asterisks). Scoring of the number of aberrant vISVs per heat-shocked embryo showed a significant rescue (0.12 in MO-ccbe1 injected n=22, versus 4.76 in uninjected controls n=25; P<0.0001) of the phenotype. |
| Fish: | |
|---|---|
| Condition: | |
| Knockdown Reagents: | |
| Observed In: | |
| Stage: | Protruding-mouth |