- Title
-
Local angiogenic interplay of Vegfc/d and Vegfa controls brain region-specific emergence of fenestrated capillaries
- Authors
- Parab, S., Card, O.A., Chen, Q., America, M., Buck, L.D., Quick, R.E., Horrigan, W.F., Levkowitz, G., Vanhollebeke, B., Matsuoka, R.L.
- Source
- Full text @ Elife
|
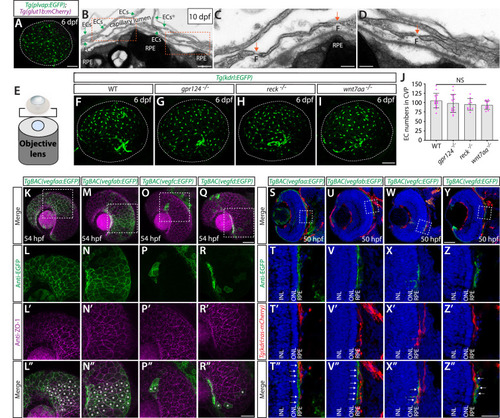
Characterization of endothelial marker and β-catenin reporter expression in zebrafish larval brain vasculature. ( |
|
Characterization of endothelial marker and β-catenin reporter expression in zebrafish larval brain vasculature. ( |
|
Characterization of the ( |
|
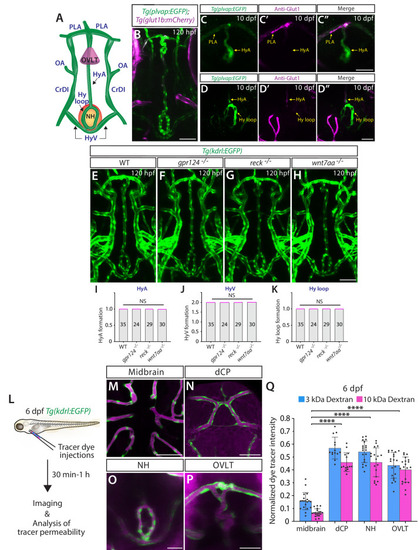
Wnt/β-catenin signaling deficiency does not cause any apparent defect in fenestrated capillary formation in the neurohypophysis (NH) and organum vasculosum of the lamina terminalis (OVLT). ( |
|
Developmental angiogenesis at the interface of the diencephalic choroid plexus (dCP) and pineal gland (PG), and ( |
|
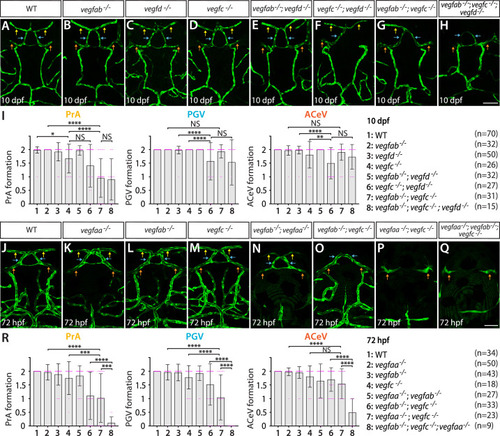
Heterogeneous endothelial requirements for Vegfs-dependent angiogenesis at the diencephalic choroid plexus (dCP)/pineal gland (PG) interface. ( |
|
Endothelial cell-autonomous and cell non-autonomous requirements of Vegfc for vascularization of the diencephalic choroid plexus (dCP)/pineal gland (PG) interface. ( |
|
Co-localization of the pituicyte marker ( |
|
Co-localization of the pituicyte marker ( |
|
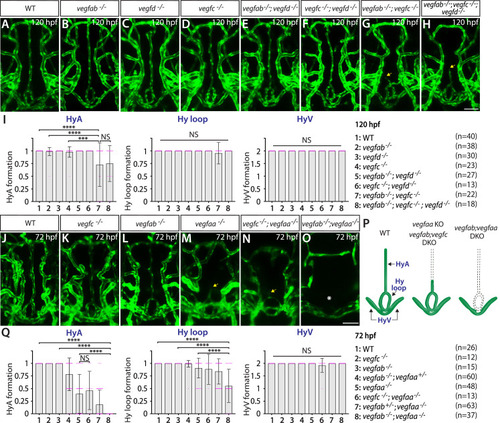
Heterogeneous endothelial requirements for Vegfs-dependent angiogenesis in the ventral brain around the neurohypophysis (NH)/organum vasculosum of the lamina terminalis (OVLT). ( |
|
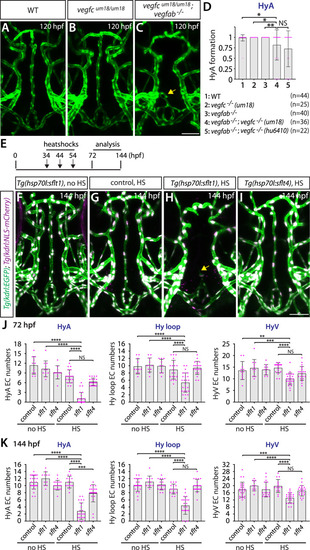
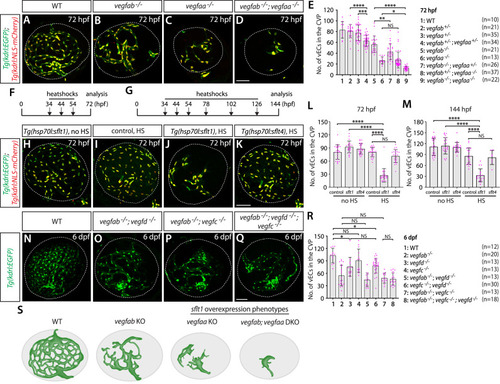
Temporal inhibition of Vegfa signaling by sFlt1 overexpression is sufficient to cause impaired formation of the hypophyseal artery (HyA) and Hy loop. ( |
|
Normal choriocapillaris formation in zebrafish deficient for Wnt/β-catenin signaling, and conserved expression of Vegfa paralogs in retinal pigment epithelium (RPE). ( |
|
Zebrafish Vegfa paralogs are redundantly required for fenestrated choroidal vascular plexus (CVP) formation. ( |
|
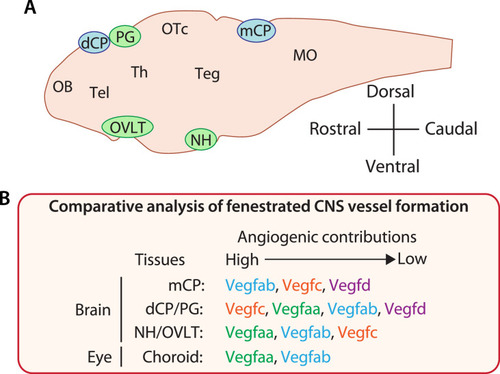
Comparative analysis of vascularization across fenestrated central nervous system (CNS) vascular beds reveals crucial angiogenic interplay of Vegfc/d and Vegfa in choroid plexus (CP) and circumventricular organ (CVO) vascularization. ( |