- Title
-
αE-catenin-dependent mechanotransduction is essential for proper convergent extension in zebrafish
- Authors
- Han, M.K., Hoijman, E., Nöel, E., Garric, L., Bakkers, J., de Rooij, J.
- Source
- Full text @ Biol. Open
|
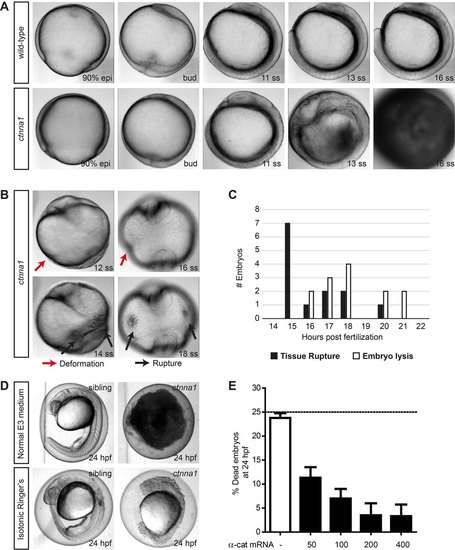
Zygotic ctnna1 mutants show defects in epithelial barrier functioning. (A,B) DIC images from a time-lapse series of zygotic ctnna1 mutants and wild-type siblings imaged from 90% epiboly until 21 somite stage. Lateral view in (A) shows embryo lysis in zygotic ctnna1 mutants during somitogenesis. Anterior views of zygotic ctnna1 mutants in (B) show deformation (red arrows) and tissue rupture (black arrows) occurring before embryo lysis. (C) Quantification of the time of rupture and subsequent embryo lysis in zygotic ctnna1 mutants (n=13 embryos from three independent experiments). (D) Phenotype at 24 hpf of zygotic ctnna1 mutants and siblings grown in regular E3 medium, and embryos transferred to isotonic Ringer's buffer at 50% epiboly. (E) Rescue of embryo lysis in ctnna1 mutants using increasing concentrations of α-catenin-GFP mRNA. The dashed line indicates the expected mortality of non-injected incrossed heterozygous ctnna1 mutants based on Mendelian genetics. Data represent three independent experiments, n>120 embryos per condition. Data are represented as the mean±s.e.m. PHENOTYPE:
|
|
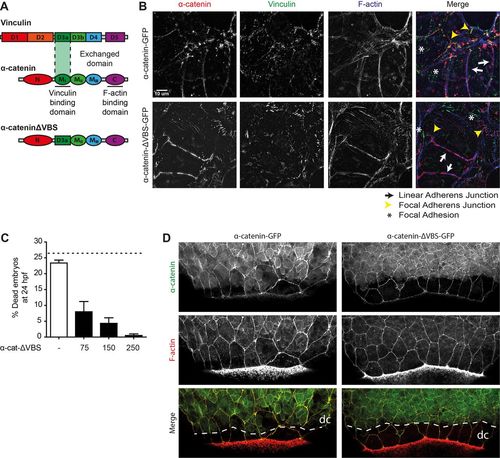
The vinculin-binding domain in αE-catenin is not needed for barrier function. (A) Schematic representation of the structures of α-catenin, vinculin and the exchanged domain in α-catenin-ΔVBS. The domain containing the vinculin binding site (VBS) was replaced by a homologous domain from vinculin itself, which does not bind vinculin. (B) Images of fixed α-catenin-deficient DLD1 R2/7 cells expressing zebrafish α-catenin-GFP (top) or α-catenin-ΔVBS-GFP (bottom) (both depicted in red), and stained for vinculin (green) and F-actin (blue). Linear adherens junction structures are highlighted by the white arrows, focal adherens junctions with the yellow arrowheads, and focal adhesions with white asterisks. (C) Rescue of embryo lysis in ctnna1 mutants using increasing concentrations of α-catenin-ΔVBS-GFP mRNA. The dashed line indicates the expected mortality of non-injected incrossed heterozygous ctnna1 mutants based on Mendelian genetics. Data represents three independent experiments, n>120 embryos per condition. Data is represented as the mean±s.e.m. (D) Whole-mount immunostaining of zebrafish embryos at 85% epiboly expressing either α-catenin-GFP (left) or α-catenin-ΔVBS-GFP (right) (both depicted in green) and stained for F-actin (red). The dashed white line marks the deep cell margin (dc) while the actin ring marks the EVL margin. |
|
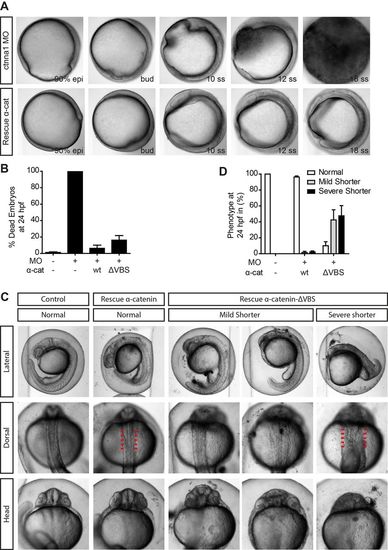
The vinculin-binding domain is essential for proper body axis elongation. (A) DIC images from a time-lapse image series of embryos injected with 0.84 ng ctnna1 MO (top) rescued with 250 pg α-catenin-GFP (bottom). Embryos were imaged from 80% epiboly until 21 somite stage. (B,D) Quantification of the mortality (B) and the body axis phenotype (D) of non-injected embryos, embryos injected with 0.56 ng ctnna1 MO or ctnna1 MO-injected embryos rescued with either 250 pg α-catenin-GFP or 250 pg α-catenin-ΔVBS-GFP. Data represents three independent experiments, n>70 embryos per condition. Data is represented as the mean±s.e.m. (C) Representative images of non-injected control embryos (left) and ctnna1 MO-injected embryos rescued with α-catenin-GFP or α-catenin-ΔVBS-GFP at 24 hpf. Clear morphological differences between α-catenin-GFP and α-catenin-ΔVBS-GFP-rescued embryos were apparent in anterior-posterior axis extension (top) as well as in the mediolateral dorsal convergence (middle and bottom). Red lines indicate the width of the dorsal body embryonic structure. PHENOTYPE:
|
|
Blocking vinculin recruitment to α-catenin induces convergent extension defects. (A) DIC images of non-injected embryos, embryos injected with 0.84 ng ctnna1 MO or ctnna1 MO-injected embryos rescued with either 250 pg α-catenin-GFP or 250 pg α-catenin-ΔVBS-GFP at the 5-7 somite stage (12 hpf). (B) Quantification of the angle between the anterior-most point in the head, and the posterior-most point in the tail of the embryos (shown in red in A). Datapoints represent single embryos from three independent experiments (n>25 embryos per condition). Data is represented as mean±s.e.m. One-way ANOVA following Tukey's multiple comparison test was used for comparisons among multiple groups, with ***P<0.0001. (C) Dorsal views of whole-mount in situ hybridization at 9-11 somite stage of non-injected embryos and ctnna1 MO-injected embryos rescued with either 250 pg α-catenin-GFP or 250 pg α-catenin-ΔVBS-GFP. MyoD marks the muscle precursors to visualize the somites. EXPRESSION / LABELING:
|
|
Altered lammelipodial polarity of the lateral mesoderm after blocking cadherin mechanotransduction. (A) Representative image sequence of a functional lamellipodial protrusion (red arrows) coupled to cell body displacement. The red asterisk marks the cell making protrusions, with the dashed lines indicating the displacement of the cell body. Dorsal to the right. (B) Percentage of lamellipodia coupled to cell body displacement in the direction of the protrusion by at least half the diameter of the cell. The number of cells analyzed (n) for each case is indicated below the x-axis. These were obtained from 4 embryos from 3 independent experiments and each datapoint in the graph represents the average value for one embryo analyzed. |