- Title
-
Novel cardiovascular gene functions revealed via systematic phenotype prediction in zebrafish
- Authors
- Musso, G., Tasan, M., Mosimann, C., Beaver, J.E., Plovie, E., Carr, L.A., Chua, H.N., Dunham, J., Zuberi, K., Rodriguez, H., Morris, Q., Zon, L., Roth, F.P., and MacRae, C.A.
- Source
- Full text @ Development
|
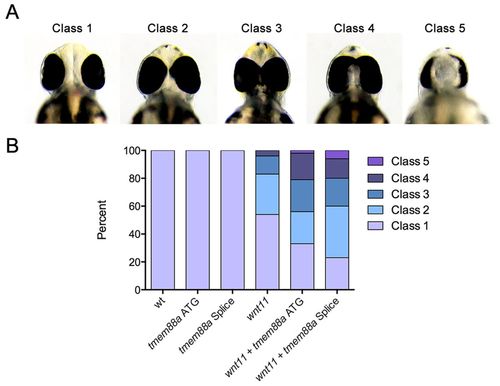
tmem88a sensitizes the wnt11 cyclopia phenotype. Embryonic zebrafish were injected with low doses of morpholinos targeting either wnt11 or tmem88a (both ATG targeting and splice blocking) and morphants scored for cyclopia at 48 hpf. Cyclopia classes (A) are as previously defined (Marlow et al., 1998), such that in class 1 eye spacing is comparable to that of wild type, class 2 the spacing is decreased, class 3 eyes are marginally fused, class 4 eyes are completely fused, and class 5 have one eye. The proportion by class (B) is significantly different (P<0.05, Chi-squared test) for both wnt11 + tmem88a ATG and wnt11 + tmem88a splice morpholino co-injections as compared with wnt11 morpholino alone. PHENOTYPE:
|
|
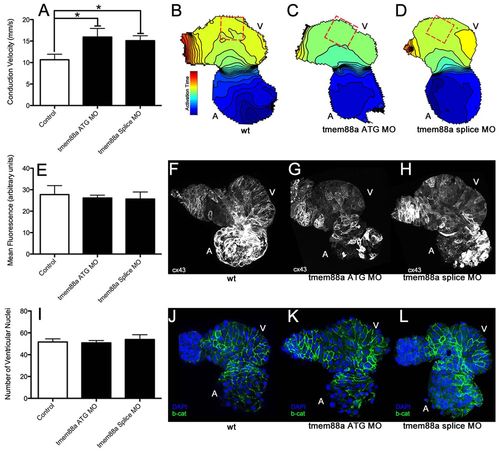
tmem88a inhibition alters cardiac ventricular polarity. Following knockdown of tmem88a by ATG-targeting or splice-blocking morpholinos, conduction velocity as measured in isolated hearts following staining with Di-8-ANEPPS was significantly increased along the outer curvature of the ventricle (A). This difference is also illustrated through isochronal maps showing voltage propagation along a control heart (B) versus hearts from embryos injected with the ATG (C) or splice-blocking (D) morpholinos (isochrons are 5 mseconds apart, conduction goes from blue to red). There was no apparent change in connexin 43 expression or cell number accompanying this alteration in cell coupling. Expression of connexin 43 (E) was strong in the atria, but weak in the ventricles in control hearts (F), and did not appear to change with inhibition of tmem88a (ATG-blocking morpholino in G, splice-blocking morpholino in H). Cell number as measured by manual counting of ventricular nuclei (I) revealed no obvious differences between control hearts (J) and hearts from embryos injected with the ATG (K) or splice (L) morpholinos (β-catenin in green, DAPI in blue). *P<0.05 (Student’s t-test, n>4 for each condition). Error bars indicate standard error. EXPRESSION / LABELING:
PHENOTYPE:
|
|
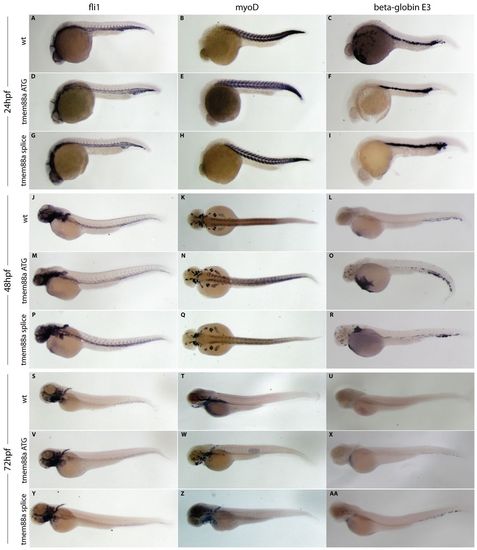
Expression analysis confirms the lack of a systematic developmental delay following tmem88a knockdown. Markers specific to the endothelium (fli1), developing muscle (myoD) and hematopoiesis (beta-globin E3) were visualized using in situ hybridization at 24 (A-I), 48 (J-R) and 72 (S-AA) hpf in wild-type embryos (A-C,J-L,S-U) and following injection of either the ATG (D-F,M-O,V-X) or splice-blocking (G-I,P-R,Y-AA) tmem88a morpholinos. Whereas fli1 and myoD expression appears unaltered, beta-globin E3 shows a slight elevation in expression following tmem88a knockdown at 48 hpf, which is largely alleviated by 72 hpf. High-resolution images of any panel are available upon request. EXPRESSION / LABELING:
|
|
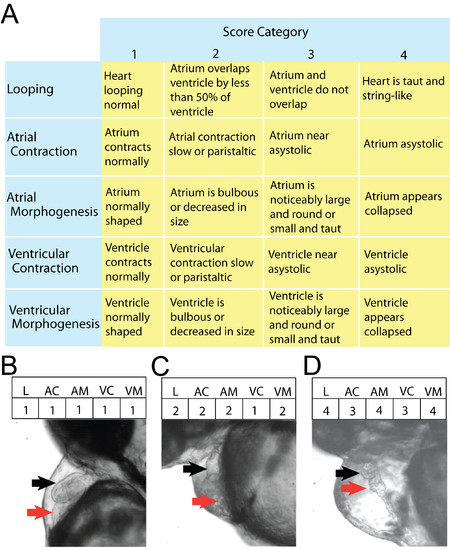
The five-category score of heart function used to identify differences in morphology and heart conduction. Numerical scores ranging from 1-4 (with 1 being nearest wild type) were assigned to 5 parameters of cardiovascular function from anonymized heart videos. Criteria for assigning these scores are outlined in A. Frames from analyzed video files depict hearts scoring progressively higher for all 5 parameters (B-D). Specifically: B is phenotypically normal, while C shows abnormal looping and a minor defect in both atrial and ventricular morphogenesis, and D depicts severe abnormalities in looping, conduction, and morphology. Red and black arrows show atria and ventricles, respectively. |
|
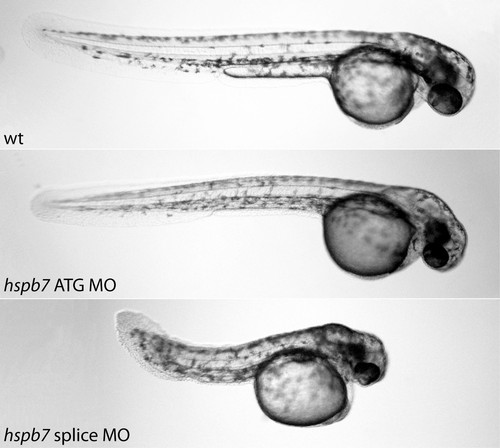
hspb7 morpholino injection causes cardiac and tail truncation phenotypes. Compared to wild-type controls at 48hpf (top panel), hpsb7 ATG (middle panel) and splice blocking (lower panel) morpholinos caused a reduction in cardiac output. In addition to the cardiac phenotype, the ATG morpholino caused an apparent thinning of the musculature in the tail, while the splice morpholino caused tail truncation. PHENOTYPE:
|