Fig. 3
- ID
- ZDB-FIG-150616-35
- Publication
- Torraca et al., 2015 - The CXCR3/CXCL11 signaling axis mediates macrophage recruitment and dissemination of mycobacterial infection
- Other Figures
- All Figure Page
- Back to All Figure Page
|
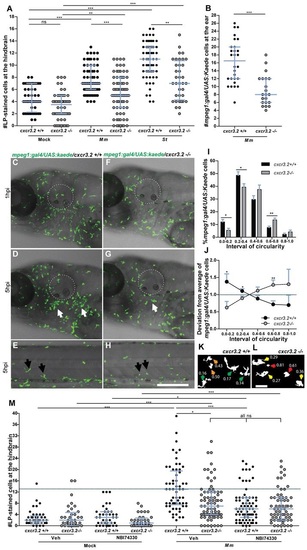
Cxcr3.2-dependent macrophage recruitment to localized bacterial infections. (A) M. marinum and S. typhimurium infection in the hindbrain ventricle. cxcr3.2+/+ and cxcr3.2−/− embryos were injected at 30 hpf with 200 CFU of M. marinum (Mm) or S. typhimurium (St), or mock injected with 2% polyvinylpyrrolidone-40 in PBS. Lp-stained cells accumulated in 3 hours within the hindbrain limits were counted as macrophages, because neutrophils do not significantly contribute to the total number of leukocytes recruited to the hindbrain at this developmental stage (supplementary material Fig. S3). Significant reduction of macrophage chemotaxis to infection is determined by cxcr3.2 mutation. Data were accumulated from two (St) or three (Mm) independent experiments. Sample size (n): 52, 52, 63, 75, 60, 36. Error bars: median and interquartile range. (B–H) Macrophage recruitment and systemic activation following M. marinum infection in the otic vesicle. Tg(mpeg1:gal4/UAS:kaede) cxcr3.2+/+ and Tg(mpeg1:gal4/UAS:kaede) cxcr3.2−/− larvae were injected with 200 CFU of M. marinum into the otic vesicle (dotted line) at 3 (B) or 4 (C–H) dpf. At 4 hpi of 3 dpf larvae, the accumulation of mpeg1:gal4/UAS:kaede-positive cells within the perimeter of the otic vesicle was reduced in cxcr3.2−/− larvae (B). Following injection at 4 dpf, macrophages are less able to penetrate the otic vesicle, but accumulate in the surrounding area (white arrows in D and G), as shown in representative time course movies (supplementary material Movies 3, 4) and stills from these movies at 1 hpi (C,F) and 5 hpi (D,G). A reduced accumulation was observed in the cxcr3.2 mutant. At a distal location in the trunk (E,H), macrophages seemed to show more frequently a branched morphology in cxcr3.2+/+ and a round morphology in cxcr3.2−/− (black arrows in E and H). Sample size (n) in B: 28, 21. Error bars: median and interquartile range. Scale bar in C–H: 250 μm. (I–L) Quantification of distal macrophage activation following local M. marinum infection. In order to quantify the distal activation of macrophages upon M. marinum infection, 200 CFU of bacteria were injected in the otic vesicle of 3 dpf mpeg1:gal4/UAS:kaede larvae. Images of the macrophages were acquired from the trunk of the infected larvae at 4 hpi and the circularity index (CI) of the distal macrophages was calculated. The graph in I represents the percentage of macrophages residing in the different intervals of CI, whereas the graph in J represents the divergence in distribution of the cxcr3.2+/+ and cxcr3.2−/− macrophages in the different classes of CI, calculated as the ratio between the percentage of cxcr3.2+/+ or cxcr3.2−/− macrophages in a certain CI interval and the overall mean percentage [(mutant + wt)/2] of macrophages in that interval (see Materials and Methods). Macrophages of cxcr3.2+/+ and cxcr3.2−/− larvae were distributed in a different fashion along the different classes, with the classes of high circularity more populated in the mutants and the classes of reduced circularity more populated in the wild type (wt). K and L show representative macrophages analyzed in I and J and their corresponding CI. Green arrows: 0.0≤CI<0.2; yellow arrows: 0.2≤CI<0.4; orange arrows: 0.4≤CI<0.6; red arrows: 0.6≤CI≤1.0. Sample size (n) in I–L: 390 cxcr3.2+/+ and 293 cxcr3.2−/− cells from 14 and 16 embryos, respectively. Error bars: mean±s.e.m. Scale bar in K,L: 40 μm. (M) Attenuation of macrophage recruitment to M. marinum via treatment with the CXCR3 antagonist NBI74330. cxcr3.2+/+ and cxcr3.2−/− embryos were bath-exposed to 50 μM NBI74330 or to vehicle only (Veh; 0.5% DMSO in medium) for 3 hours and then injected in the hindbrain ventricle at 30 hpf with mock or 200 CFU of Mm. Embryos were kept in NBI74330 or vehicle medium for an additional 3 hours and then collected for Lp immunostaining. Treatment with NBI74330 reduced the macrophage chemotaxis to infection in cxcr3.2+/+ embryos to similar levels as the vehicle-treated cxcr3.2−/− embryos, and no significant additive effect of cxcr3.2 mutation and NBI74330 treatment was observed. Data were accumulated from two independent experiments. Sample size (n): 41, 36, 39, 36, 61, 79, 73, 67. Error bars: median and interquartile range. ns, non-significant; *P<0.05; **P<0.01; ***P<0.001. |
| Fish: | |
|---|---|
| Condition: | |
| Observed In: | |
| Stage: | Prim-15 |