- Title
-
Bi-allelic variants in FLII cause pediatric cardiomyopathy by disrupting cardiomyocyte cell adhesion and myofibril organization
- Authors
- Ruijmbeek, C.W., Housley, F., Idrees, H., Housley, M.P., Pestel, J., Keller, L., Lai, J.K.H., van der Linde, H.C., Willemsen, R., Piesker, J., Al-Hassnan, Z.N., Almesned, A., Dalinghaus, M., van den Bersselaar, L.M., van Slegtenhorst, M.A., Tessadori, F., Bakkers, J., van Ham, T.J., Stainier, D.Y., Verhagen, J.M., Reischauer, S.
- Source
- Full text @ JCI Insight
|
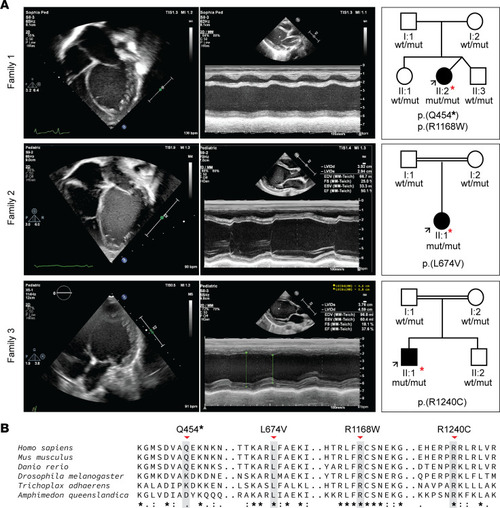
Clinical manifestation of early-onset DCM and family pedigree of affected individuals included in this study. ( |
|
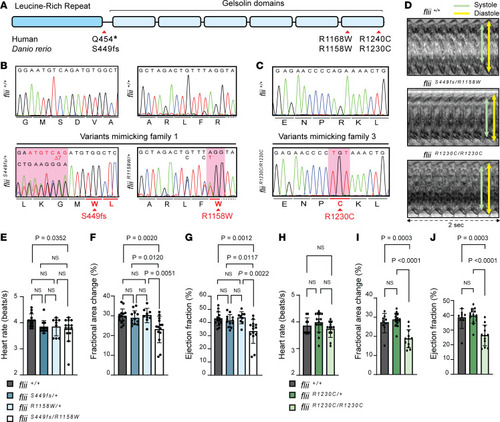
CRISPR/Cas9-mediated genome editing of patient-specific biallelic variants in ( |
|
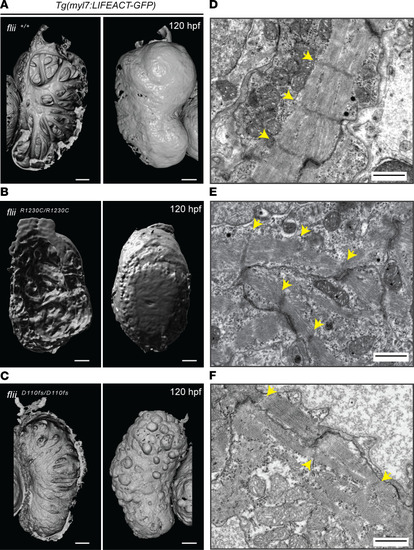
Flii dysfunction results in myofibrillar architectural abnormalities of the ventricular myocardium. ( |
|
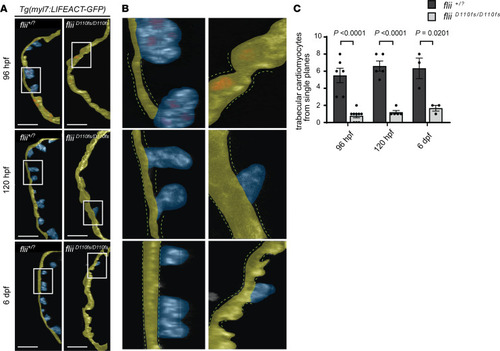
Loss of Flii results in a reduced number of cardiac trabeculae. ( |
|
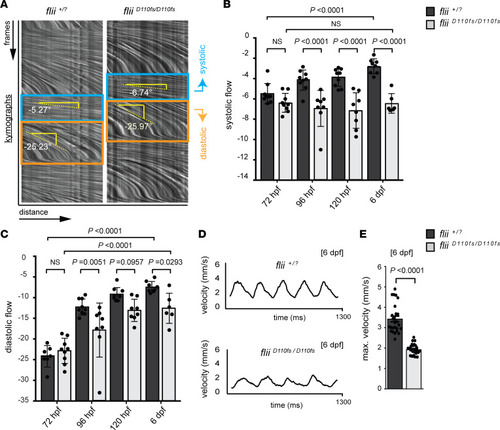
Blood flow analysis reveals reduced cardiac performance upon Flii deficiency, including a developmental arrest in the systolic hemodynamic force. ( |
|
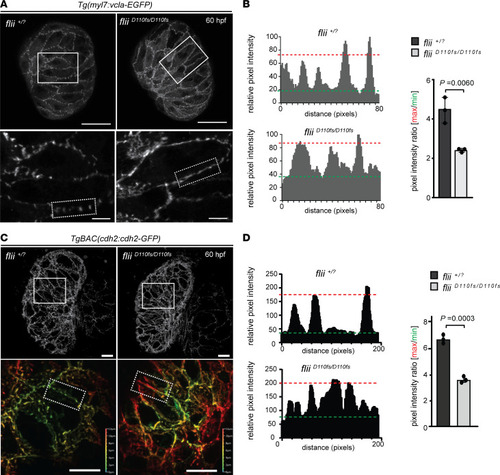
Flii-deficient zebrafish exhibit defects in vinculin-EGFP and cadherin2-EGFP localization. ( |
|
Aberrant activation of the Notch and Hippo signaling pathways in Flii-deficient ventricles. ( |