- Title
-
Marcksl1 modulates endothelial cell mechanoresponse to haemodynamic forces to control blood vessel shape and size
- Authors
- Kondrychyn, I., Kelly, D.J., Carretero, N.T., Nomori, A., Kato, K., Chong, J., Nakajima, H., Okuda, S., Mochizuki, N., Phng, L.K.
- Source
- Full text @ Nat. Commun.
|
|
|
|
|
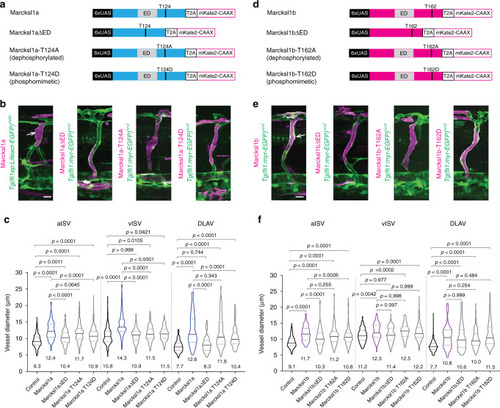
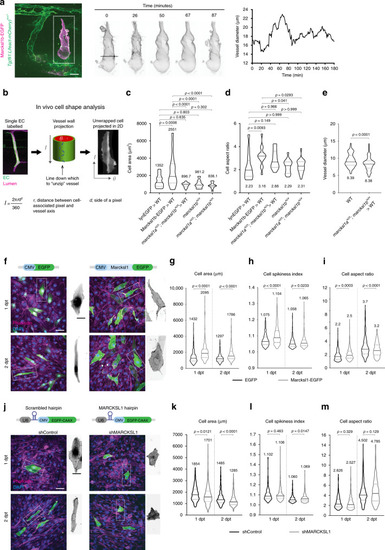
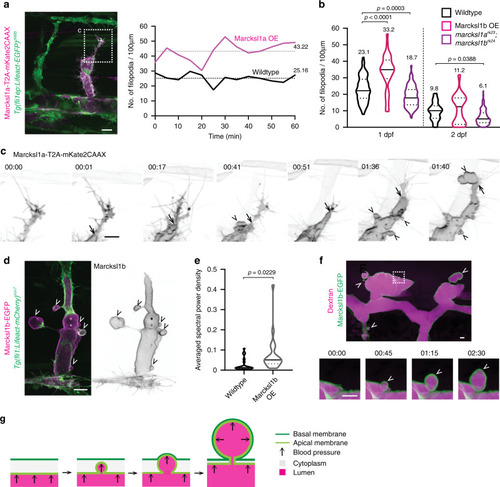
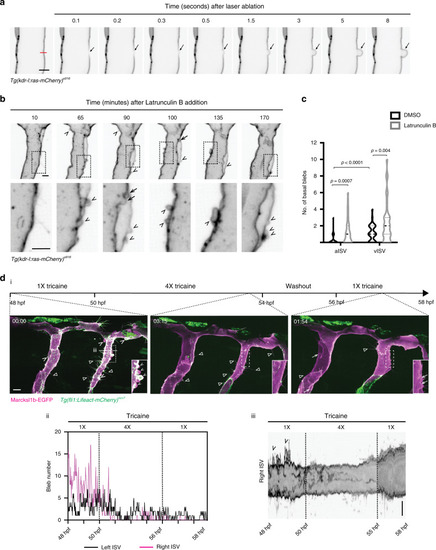
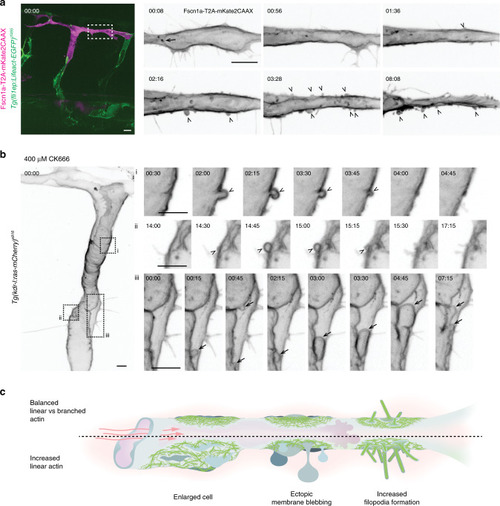
PHENOTYPE:
|
|
|
|
|
|
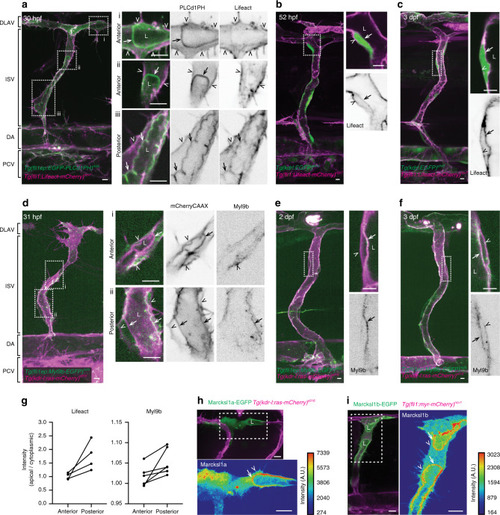
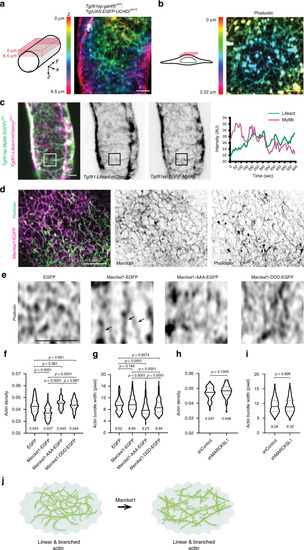
The spatiotemporal dynamics Lifeact (actin, |
|
|
|
|
|
|