- Title
-
Population-scale organization of cerebellar granule neuron signaling during a visuomotor behavior
- Authors
- Sylvester, S.J.G., Lee, M.M., Ramirez, A.D., Lim, S., Goldman, M.S., Aksay, E.R.F.
- Source
- Full text @ Sci. Rep.
|
Imaging the signaling properties of transgenically identified granule cells during oculomotor control. (a) Current understanding of the signal flow underlying the control of optokinetic behavior. The core sensorimotor elements of the circuit are the pretectum (PT), second-order vestibular nuclei (V), the velocity-storage neural integrator (VSNI), and the velocity-to-position neural integrator (VPNI). Adaptations in oculomotor behavior are mediated by the Purkinje (PJ) and granule (Gr) cells of the cerebellum. In the mammal, pretectal signals and stimulus information processed in a cortical path are combined at the dorsolateral pontine nucleus before being sent to the cerebellum22. Filled circle - inhibitory connections; arrows – excitatory connections; for simplicity, callosal projections not explicitly indicated. (b) Experimental setup. Two-photon excitation with a Ti-Sapphire laser tuned to the infrared frequency range enables simultaneous fluorescence microscopy of granule cell function, behavioral stimulation, and eye-movement tracking. Abbreviations OBJ: objective, L: lens, F: filter, PMT: photomultiplier tube, PD: photodetector, DM: dichroic mirror, IR LED: infrared light emitting diode. (c) Spectral finger-printing procedure allowing identification of GFP-positive granule neurons. (Top) A histogram of the pixel-by-pixel differences in intensity (uint16) between two excitation wavelengths (, black) was compared to the histogram of differences expected from noise (grey) (fit of noise histogram as dashed orange line). Pixels with difference values one noise standard deviation or more from zero were considered GFP-positive candidates (green box). (Bottom) Candidate GFP-positive pixels (green), at one image plane, identified in the inner granule layer (IGL) of the right lobe of the transgenic zebrafish cerebellum. Also visible is an electrode for glutamate iontophoresis (red). The location of the center of the midbrain-hindbrain border (*) is visible at the upper left. |
|
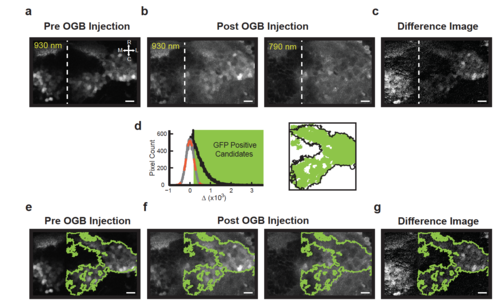
Spectral fingerprinting procedure for identifying the inner granule layer (IGL) after OGB dye loading. (a) Before OGB loading, GFP-positive granule cells in the inner granule layer (IGL) are easily identified with 930 nm excitation in Tg(gata1:GFP) zebrafish. The shown image plane contains large portions of the right cerebellar lobe and parts of the left lobe (dashed: midline); granule cell axons are located rostrally and medially. The scale bar, here and below, is 10 μm. (b) After OGB injection into the right lobe, the IGL has similar intensity to the rest of the cerebellum, both at 930 nm (left) and 790 nm (right) excitation. Note, however, the slightly elevated intensity in the IGL at 930 nm. (c) A pixel-by-pixel difference (Methods) of the 930 nm and 790 nm images highlights the IGL. (d; left) To identify the borders of the IGL, a histogram of pixel intensity differences (black) is compared to that expected from noise; the noise histogram is generated by mirroring negative difference values about zero (grey; gaussian fit shown in dashed orange). Those difference values a standard deviation or greater from zero were considered candidate GFP-positive pixels (green box). (right) Identified regions passing size and contiguity cutoffs (green, Methods) align closely with the outlines of the IGL identified in panel a (black). (e-g) Outlines of the region identified as containing GFP-positive granule neurons (green) overlaid onto the images in panels a-c. |
|
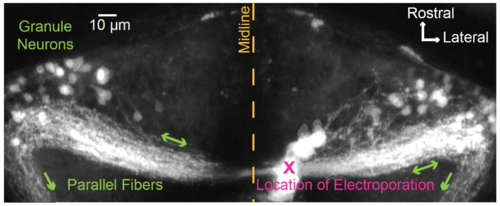
Targeting granule neurons for antidromic activation. To determine how changes in granule cell firing were coupled to changes in the somatic calcium concentration, we sought to identify a location for electrical stimulation of granule cell axons that would enable antidromic activation of granule neurons in the absence of direct depolarization to the soma or secondary input from mossy fibers or other cerebellar neuronal classes. Here we show that targeting of the caudal molecular layer in one half of the cerebellum allows selective access to many of the granule cells with somata in the other half of the cerebellum. To do so, we targeted caudal lobe commissure parallel fibers1 for electroporation with 3 kD Texas Red Dextran (D-3329, Life Technologies; 10 mM in distilled water; 7 dpf nacre). Electroporation via a fine glass capillary (1 second long train of 2 msec 70 V pulses delivered at 200 Hz) resulted in immediate loading of cells and tissue near the poration site (“X”). Over a course of ten minutes, dye diffused in parallel fibers away from the poration site to label granule cell somata located on either side of the midline within the eminentia granularis and lateral portions of the IGL. Some parallel fibers were also seen to exit the cerebellum on both sides (green arrows) and terminate in rhombomeres 1 and 2 (not shown). For the antidromic activation experiments found within the main text, depolarizing electrical pulses were therefore delivered at locations similar to the location of the electroporation shown here, thereby minimizing activation of a larger portion of the cerebellar circuit and ensuring that changes in somatic fluorescence intensity were directly driven by changes in granule cell firing. |