- Title
-
IL4/STAT6 Signaling Activates Neural Stem Cell Proliferation and Neurogenesis upon Amyloid-β42 Aggregation in Adult Zebrafish Brain
- Authors
- Bhattarai, P., Thomas, A.K., Cosacak, M.I., Papadimitriou, C., Mashkaryan, V., Froc, C., Reinhardt, S., Kurth, T., Dahl, A., Zhang, Y., Kizil, C.
- Source
- Full text @ Cell Rep.
|
Aβ42 Injection Paradigm and Aggregation-Dependent Phenotypes in Adult Zebrafish Brain (A) Immunohistochemistry (IHC) for Aβ42 in control brains at 1, 3, and 14 days post-injection (dpi) of PBS. Right panels: green channel. (B-D) IHC for Aβ42 in brains injected with uncoupled Aβ42 (B), R9-Aβ42 (C), or TR-Aβ42 (D). (E) Quantification for green fluorescence intensity as a measure of amyloid deposition. (F) CD spectra for all Aβ42 versions in buffer (PBS). Aβ42 peptides show β sheet formation. (G-H') Electron micrographs of TR-alone- (G) and TR-Aβ42-injected (H) brains. Blue, nucleus; green, amyloid. (G' and H') Higher magnification of perinuclear regions with Aβ42. (I and I') IHC for Aβ42 in control (I) and TR-Aβ42-injected (I’) brains. Green and DAPI are shown separately. (J and J') TUNEL for control (J) and TR-Aβ42-injected (J') brains. Insets: single channels of a close-up region. (K and K') HuC/D and L-Plastin in control (K) and TR-Aβ42-injected (K') brains. (L and L') Synaptophysin in control (L) and TR-Aβ42-injected (L') brains. (M) Quantification for (J-L'). (N) IHC for HuC/D and TR-Aβ42 coupled to TUNEL staining. (O) Categorization of TUNEL-positive cells according to HuC/D expression and presence of Aβ42. Scale bars, 100 μm. Data are represented as mean ± SD; n = 3 fish. ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001. See also Figures S1–S3. |
|
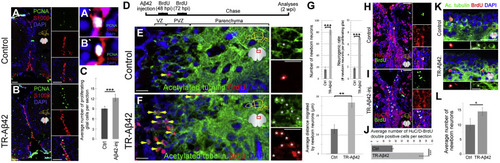
Aβ42 Induces Stem Cell Proliferation and Neurogenesis in Adult Zebrafish Brain (A) PCNA, S100β and DAPI in control brains. Insets: individual channels. (A') High-magnification of a double-positive cell. (B) PCNA, S100β, and DAPI in TR-Aβ42-injected brains. (B') High-magnification of a double positive cell. (C) Quantification for the average number of proliferating glial cells. Ctrl, control; inj, injected. (D) Neurogenesis assay. (E and F) Acetylated tubulin and BrdU in control (E) and TR-Aβ42-injected (F) brains. Insets: single channels. (G) Number and migration distance of newborn neurons. (H and I) HuC/D and BrdU in control (H) and TR-Aβ42-injected brains at 2 weeks post-injection (wpi). (J) Quantification of HuC/D-BrdU double-positive cells. (K) Acetylated tubulin and BrdU in control and TR-Aβ42-injected brains at 3 months after injection. Insets: examples of newborn neurons. (L) Quantification of acetylated tubulin and BrdU double-positive cells per section. Scale bars, 50 μm (A and B) and 20 μm (E, F, H, I, and K). Data are represented as mean ± SD; n = 3 fish. ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001. See also Figures S1 and S3 and Table S2. |
|
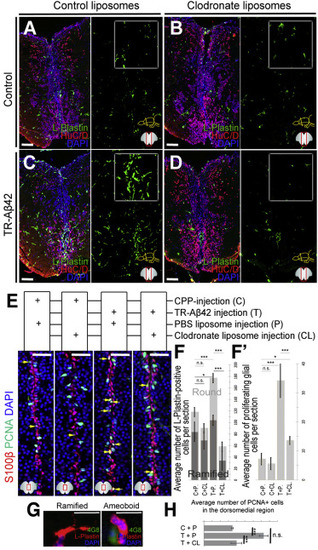
Aβ42-Dependent Stem Cell Proliferation Requires Microglial Activity (A–D) L-plastin and HuC/D in control peptide/control liposome (A), control peptide/clodronate liposome (B), TR-Aβ42/control liposome (C), and TR-Aβ42/clodronate liposome (D) injections. Right panels: green channel. (E) S100β and PCNA in brains injected with the four combinations in (A–D). (F) Quantification of the number of L-plastin-positive cells. (F') Quantification of proliferating NSPCs. (G) Examples of ramified and round/ameoboid L-plastin-positive cells. Round cells are around amyloid (green, 4G8 staining). (H) Quantification for the average number of PCNA-positive cells in clodronate-injected brains. Scale bars, 50 μm. Data are represented as mean ± SD; n = 4 fish. ∗p < 0.05; ∗∗∗p < 0.001; n.s., not significant. See also Tables S1 and S2. |
|
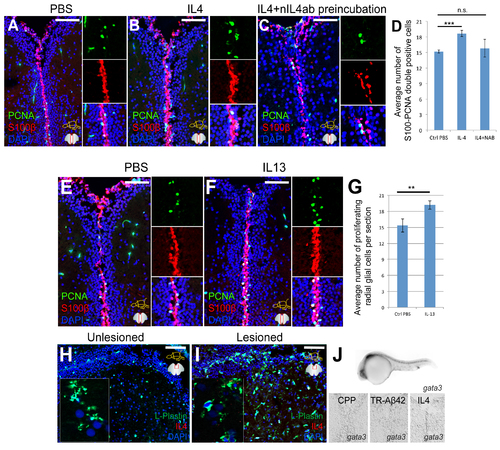
Interleukin-4 Is Necessary and Sufficient for Aβ42-Dependent Stem Cell Proliferation and Neurogenesis in Adult Zebrafish Brain (A–B'') IL4 in control (A–A'') and TR-Aβ42-injected (B-B'') brains. Parenchyma (A' and B') and ventricle (A'' and B'') are shown. (C) IL4, L-plastin, and HuC/D in TR-Aβ42-injected brains. (C') Close-up on a neuron (HuC/D-positive, red) with TR-Aβ42 (green) and IL4 (white). (D–D'') il4r receptor expression in TR-Aβ42 brains at 3 dpi. Sense (D) and antisense (D'). (D'') High magnification. (E and F) S100β and PCNA in control (E) and IL4-injected (F) brains. (G) Quantification graph for (E and F). (H and I) BrdU and HuC/D in control (H) and IL4-injected (I) brains. (J) Quantification graph for (H and I). (K and K') IL4 detection with neutralizing antibody in control (K) and TR-Aβ42-injected (K') brains. (L and L') Secondary antibody staining for the neutralizing IL4 antibody (nIL4ab) in uninjected (L) and injected brains at 1 day after injection (L'). (M and N) PCNA and S100β in TR-Aβ42-injected (M) and in TR-Aβ42 + neutralizing antibody-co-injected (N) brains. Insets: single fluorescence channels. (O) Quantification graph. Control = 1. (P–P''') Phospho-STAT6 in PBS- (P), TR-Aβ42- (P'), IL4- (P”), and TR-Aβ42 + neutralizing IL4-antibody-injected (P''') brains. Red channels shown separately. Insets: close-up image of one exemplary cell. (Q–S) PCNA and S100 in TR-Aβ42 and control morpholino-injected (Q) and in TR-Aβ42 and IL4R morpholino-injected (R) brains. (S) Quantification graph. (T and U) IL4 after TR-Aβ42 in PBS (T) or clodronate-injected (U) brains. (V–Z) PCNA and S100 after TR-Aβ42 in PBS (V), clodronate (X) or clodronate + IL4-injected (Y) brains. (Z) Quantification graph for (V–Y). Scale bars, 50 μm (A, B, E–I, M, N, Q, R,V–Y), 10 μm (A', A'', B', B'', C, P–P''', T, and U), and 20 μm (K–L'). Data are represented as mean ± SD; n = 4 fish. ∗p < 0.05; ∗∗p < 0.01; ∗∗∗p < 0.001; n.s., not significant. See also Figure S4. |
|
Chemical properties and effects of various amyloid beta peptides and expression changes in inflammation-related and synaptic genes. Related to Figure 1, Figure 2. (A) Circular Dichroism (CD) spectra for uncoupled Aβ42, TR-Aβ42, R9-Aβ42 and scrambled Aβ42 in buffer (PBS). Aβ42 peptides show β-sheet formation in buffer. (B) CD for CPPs alone in buffer. They do not show beta structures. (C) Circular Dichroism (CD) spectra for uncoupled Aβ42, TR-Aβ42, R9-Aβ42 and scrambled Aβ42 in water. Aβ42 peptides do not show β-sheet formation in water. (D) CD for CPPs in water. They do not show beta structures. (E) Light scattering analyses to detect aggregation for R9-Aβ42, TR-Aβ42, Aβ42 scrambled and R9-Aβ38 peptides in water and buffer. Aβ42 peptides aggregate in buffer significantly more than Aβ42 scrambled and Aβ38. No peptide aggregates in water. (F) Mass by charge ratio of the native and modified and amyloid beta peptides. (G) Liquid Chromatography - Mass Spectrometry of HPLC-purified native and modified and amyloid beta peptides. (H) Immunostaining for HuC/D and L-Plastin on PBS-injected brains. (H’) Green channel alone. (I) Immunostaining for HuC/D and L-Plastin on TR peptide-injected brains. (I’) Green channel alone. (J) Immunostaining for S100beta and PCNA on PBS-injected brains. (J’) Green channel alone. (K) Immunostaining for S100b and PCNA on TR peptide-injected brains. (K’) Green channel alone. (L) Quantification graph for L-Plastin cells. TR peptide does not alter the number and activation state of the microglia. (M) Quantification graph for radila glial cell proliferation. TR peptide does not alter the proliferation state of the adult zebrafish brain. (N) TUNEL and Aβ42 staining for control (a-c), uncoupled Aβ42 (d-f), R9-Aβ42 (g-i), TR-Aβ42 (j-l) at 1-, 3- and 14 days post injection (dpi). small insets on the right of every panel are individual fluorescence channels and the composite from a close-up region. (O) L-Plastin and HuC/D staining for control (a-c), uncoupled Aβ42 (d-f), R9-Aβ42 (g-i), TR-Aβ42 (j-l) at 1-, 3- and 14 days post injection (dpi). small insets on the right of every panel are individual fluorescence channels and the composite from a close-up region. (P) S100 and PCNA staining for control (a-c), uncoupled Aβ42 (d-f), R9-Aβ42 (g-i), TR-Aβ42 (j-l) at 1-, 3- and 14 days post injection (dpi). small insets on the right of every panel are individual fluorescence channels and the composite from a close-up region. (Q) Coupling reaction for attaching fluorescein to peptides. (R) Fluorescein, TUNEL, S100β and PCNA, L-Plastin and HuC/D, Synptophysin immunostainings for PBSinjected, TR-Aβ38-injected and TR-Aβ42-scrambled peptide injected brains. (S) Quantification graphs for R. (T) Relative fold changes of inflammation-related genes. (U) Relative fold changes of synapse-related genes. Scale bars: 50 μm. Data are represented as mean ± SD. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
Time course analyses of cell death, microglial activation, synaptic degeneration and cell proliferation in adult zebrafish brain after Aβ42 injection. Related to Figure 1, Figure 3. (A) TUNEL staining and immunohistochemistry (IHC) for Aβ42 in PBS-injected controls and TR-Aβ42-injected brains at 1, 3, 7, 14, 30 dpi. (B) Quantification graph for A. (C) HuC/D and L-Plastin IHC in PBS-injected controls and TR-Aβ42-injected brains at 1, 3, 7, 14, 30 dpi. (D) Quantification graph for C. (E) Synpatophysin IHC in PBS-injected controls and TR-Aβ42-injected brains at 1, 3, 7, 14, 30 dpi. (F) Quantification graph for E. (G) S100 and PCNA IHC in PBS-injected controls and TR-Aβ42-injected brains at 1, 3, 7, 14, 30 dpi. (H) Quantification graph for G. (I) Timeline of phenotypic outcomes. (J) Close-up image of Acetylated tubulin (green) and BrdU (red) staining. DAPI (blue) as a counterstain. (K) Red channel of image in J. (L) Green channel of image in J. Scale bars: 50 μm (A, C, E, G), 10 μm (J-L). Data are represented as mean ± SD. |
|
Specificity of the IL4 neutralization, and the effects of IL13 on radial glial cell proliferation. Related to Figure 3, Figure 4. (A-C) Immunohistochemistry (IHC) for S100beta and PCNA on fish brain injected with PBS (A), IL4 (B) and IL4 preincubated with the neutralizing antibody (C). Insets on the right of every panel show individual fluorescence channels and the composite from a close-up region. (D) Quantification graph for A-C. Neutralizing antibody abrogates the inductive effect of IL4 on radial glial cell proliferation. (E,F) IHC for S100beta and OCNA on fish brains injected with PBS (E) or IL13 (F). Insets on the right of every panel show individual fluorescence channels and the composite from a close-up region. (G) Quantification graph for E and F. IL13 does not increase radial glial cell proliferation in adult zebrafish brain. (H) L-Plastin and IL4 staining in unlesioned zebrafish brain. Inset is close-up image. (I) L-Plastin and IL4 staining in zebrafish brain 3 days after lesion. Inset is close-up image. (J) gata3 in situ hybridization in 24 hour post fertilization embryo, and in adult zebrafish brains injected with TR cell penetrating peptide (CPP), TR-Aβ42, and IL4. gata3 is expressed in embryos but not in adult zebrafish brain after TR-Aβ42 or IL4. Scale bars 100 μm. Data are represented as mean ± SD. |