- Title
-
HOXC9 Regulates Formation of Parachordal Lymphangioplasts and the Thoracic Duct in Zebrafish via Stabilin 2
- Authors
- Stoll, S.J., Bartsch, S., and Kroll, J.
- Source
- Full text @ PLoS One
|
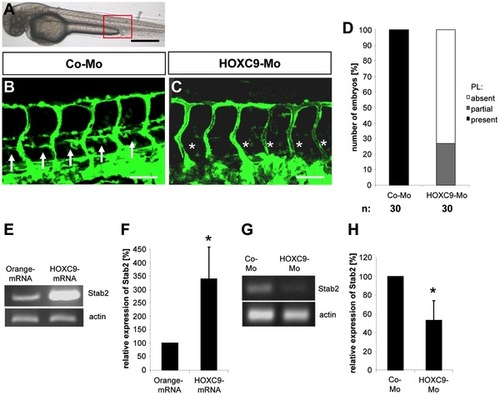
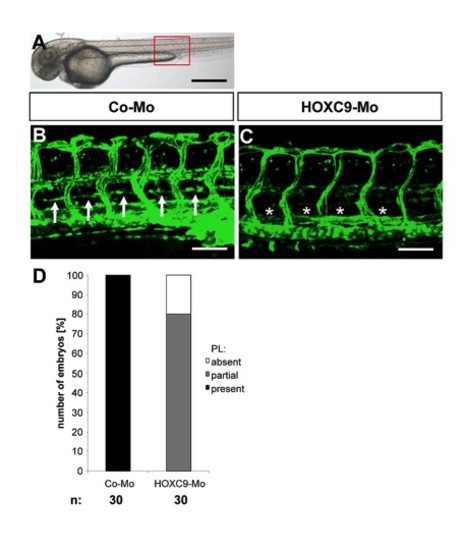
Silencing of HOXC9 expression in zebrafish inhibits assembly of parachordal lymphangioplasts (PLs). |
|
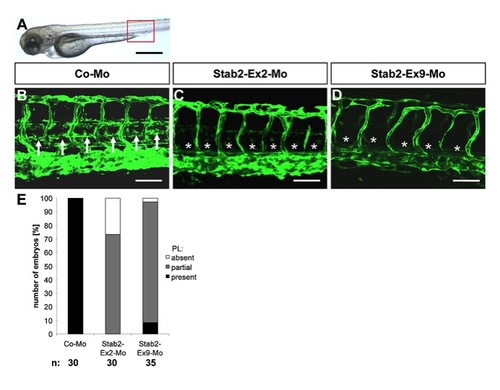
Silencing of Stab2 expression in zebrafish inhibits assembly of parachordal lymphangioplasts (PLs). |
|
HOXC9 overexpression rescues the defects in parachordal lymphangioplast (PL) formation in Stab2 morphants. |
|
Silencing of Stab1 expression in zebrafish inhibits assembly of parachordal lymphangioplasts (PLs). |
|
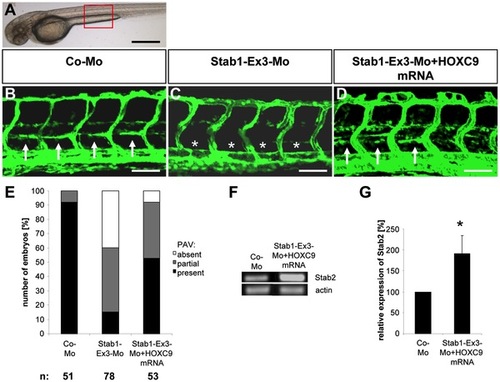
HOXC9 overexpression rescues the defects in parachordal lymphangioplast (PL) formation in Stab1 morphants. |
|
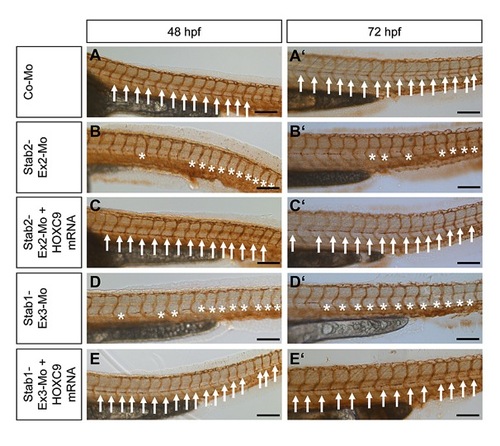
Silencing of HOXC9, Stab1 and Stab2 expression in zebrafish inhibits formation of the thoracic duct (TD). |
|
Silencing of HOXC9, Stab2 and Stab1 expression in zebrafish inhibits assembly of parachordal lymphangioplasts (PLs). |
|
Silencing of HOXC9 expression in zebrafish inhibits assembly of parachordal lymphangioplasts (PLs) at 72 hpf. |
|
Silencing of Stab2 expression in zebrafish inhibits assembly of parachordal lymphangioplasts (PLs) at 72 hpf. (A) Overall morphology of 72 hpf zebrafish embryo after control morpholino injection. Red box shows region displayed in (B–D). (B) Normal formation of the PLs (arrows) in 72 hpf tg(fli1:EGFP) zebrafish embryo after injection of 4 ng control morpholino. (C,D) Silencing of Stab2 expression using 4 ng splice-blocking morpholino targeting exon 2 (C) or 2 ng splice-blocking morpholino targeting exon 9 (D) disrupted the formation of the PLs (asterisks) in 72 hpf tg(fli1:EGFP) zebrafish embryos. (E) Quantification of 72 hpf tg(fli1:EGFP) zebrafish embryos showing a disturbed PL formation. Embryos were divided in three groups depending on the PL appearance being completely absent, partially formed or completely present. Black scale bar: 500 μm. White scale bar: 50 μm. |
|
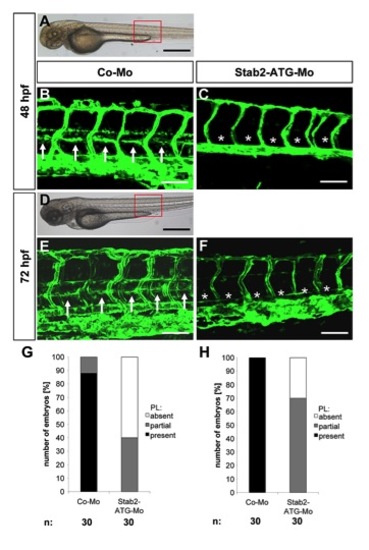
Silencing of Stab2 expression using an ATG-morpholino in zebrafish inhibits assembly of parachordal lymphangioplasts (PLs). (A;D) Overall morphology of 48 hpf (A) and 72 hpf (D) zebrafish embryos after injection of control morpholino. Red box shows region displayed below in (B,C,E,F). (B,E) Normal formation of the PLs (arrows) in 48 hpf (B) and 72 hpf (E) tg(fli1:EGFP) zebrafish embryos after injection of 4 ng control morpholino. (C,F) Silencing of Stab2 expression using 2 ng translational-blocking morpholino disrupted the formation of the PLs (asterisks) in 48 hpf (C) and 72 hpf (F) tg(fli1:EGFP) zebrafish embryos. (G,H) Quantification of 48 hpf (G) and 72 hpf (H) tg(fli1:EGFP) zebrafish embryos showing a disturbed PL formation. Embryos were divided in three groups depending on the PL appearance being completely absent, partially formed or completely present. Black scale bar: 500 μm. White scale bar: 50 μm. |
|
HOXC9 overexpression rescues defects in parachordal lymphangioplast (PL) formation in Stab2 and Stab1 morphants. |
|
HOXC9 overexpression rescues the defects in parachordal lymphangioplast (PL) formation in Stab2 morphants at 72 hpf. |
|
Low dose injection (50 pg) of HOXC9 mRNA shows no effect on zebrafish vascular morphology. |
|
Silencing of Stab1 expression in zebrafish inhibits assembly of parachordal lymphangioplasts (PLs) at 72 hpf. |
|
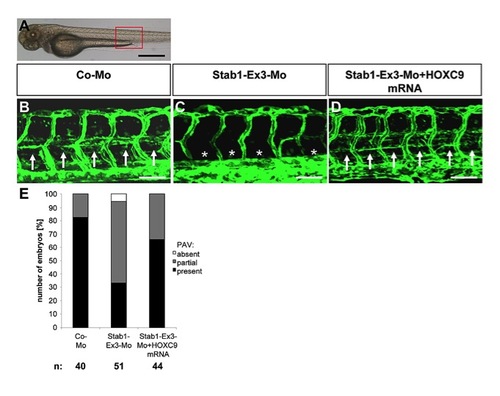
HOXC9 overexpression rescues the defects in parachordal lymphangioplast (PL) formation in Stab1 morphants. (A) Overall morphology of 72 hpf zebrafish embryo after control morpholino injection. Red box shows region displayed in (B–D). (B) Normal formation of the PLs (arrows) in 72 hpf tg(fli1:EGFP) fish embryo after injection of 4 ng control morpholino. (C) Silencing of Stab1 expression using 4 ng splice-blocking morpholino disrupted the formation of the PLs (asterisks) in 72 hpf tg(fli1:EGFP) zebrafish embryo. (D) Injection of HOXC9 mRNA (50 pg) rescued the Stab1 loss-of-function phenotype in 72 hpf tg(fli1:EGFP) zebrafish embryo. (E) Quantification of 72 hpf tg(fli1:EGFP) zebrafish embryos showing a disturbed PL formation including rescue experiments using HOXC9 mRNA (50 pg). Embryos were divided in three groups depending on the PL appearance being completely absent, partially formed or completely present. Black scale bar: 500 μm. White scale bar: 50 μm. |
|
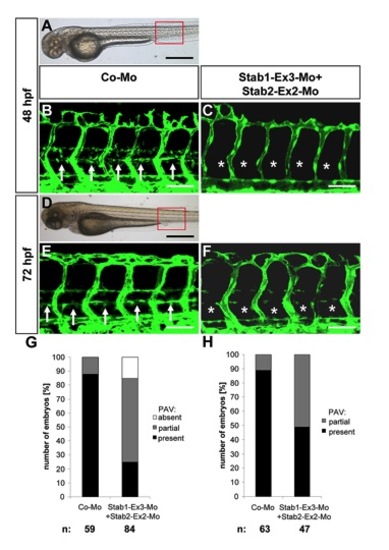
Expression silencing of Stab1 and Stab2 shows no additive effect on parachordal lymphangioplast (PL) assembly. |