- Title
-
Zebrafish model for human long QT syndrome
- Authors
- Arnaout, R., Ferrer, T., Huisken, J., Spitzer, K., Stainier, D.Y., Tristani-Firouzi, M., and Chi, N.C.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
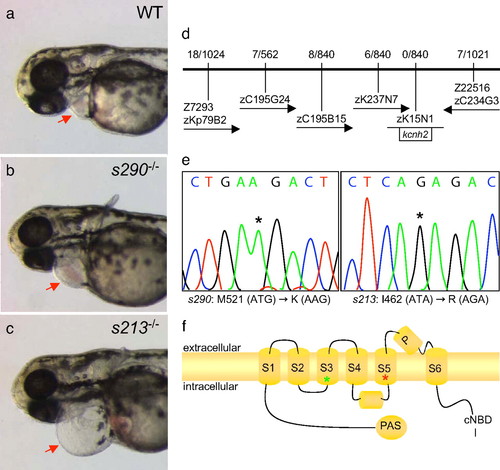
Molecular analysis of two kcnh2 mutant alleles. (a–c) Wild-type and mutant embryos at 48 hpf, lateral oblique views, anterior to the left. Compared with wild type (WT) (a), s213 (b), and s290 (c), mutants exhibit a silent ventricle and, therefore, absent cardiac output and pronounced pericardial edema (red arrows). (d) The mutations map to linkage group 3. Flanking CA repeat markers and markers from selected BACs are shown, with the number of recombinants for the given number of meioses. The s213 and s290 mutations map to the potassium channel gene kcnh2. (e) Sequence analysis of kcnh2 in s213 mutants reveals a T–G transition at codon 462, resulting in an Ile–Arg substitution. For s290, a T–A transversion at codon 521 results in a Met–Lys substitution. (f) Schematic diagram showing the modular structure of Kcnh2. A Kcnh2 subunit consists of six transmembrane domains: the S1–S4 domains sense membrane potential, whereas the S5–S6 domains form the K-selective pore. Green and red stars represent the s213 and s290 mutation sites, respectively. PHENOTYPE:
|
|
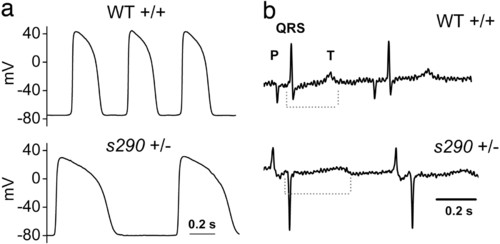
APs recorded from explanted embryonic zebrafish hearts. (a) (Upper) Representative spontaneous APs recorded from wild-type (WT) atrium (Left) and ventricle (Right) at 48 hpf. (Lower) Spontaneous APs recorded from kcnh2s290 mutant atrium (Left). Recordings of transmembrane voltage (V m) from mutant ventricle (Right) revealed marked membrane depolarization and the absence of action potentials. (b) V m recorded from kcnh2s290 mutant ventricle. Intracellular injection of hyperpolarizing current (-100 pA) caused membrane hyperpolarization and allowed for the generation of spontaneous APs. PHENOTYPE:
|
|
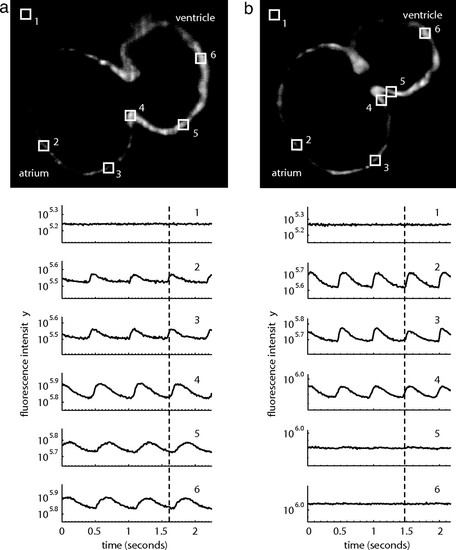
Wild-type embryonic hearts in the Tg(cmlc2:gCaMP)s878 background exhibit atrial and ventricular conduction waves, whereas mutant hearts exhibit atrial, but no ventricular, conduction waves. SPIM videos of live 48 hpf hearts in the Tg(cmlc2:gCaMP)s878 background (see SI Movies 3 and 4) were processed to determine the fluorescence intensity of selected regions of the heart over time. Each selected region has a corresponding number plotted below. The dotted lines mark an arbitrary point in time to facilitate comparison across the different plots. (a) In a wild-type heart, fluorescence intensity varies with time in atrial and ventricular regions of the heart as the wave of depolarization propagates through the heart. This wave represents a wild-type heart rhythm. (b) In a kcnh2s290 mutant heart, fluorescence intensity varies in the atrium, but the ventricle maintains a constant, low-level intensity. The mutant heart lacks a ventricular conduction wave. PHENOTYPE:
|
|
Heterozygous kcnh2 zebrafish manifest delayed ventricular repolarization. (a) APs recorded from 48 hpf heterozygous ventricle show increased AP duration compared with wild-type (see Results). (b) Representative electrocardiograms recorded from anesthetized, paralyzed wild-type and heterozygous adult zebrafish. Dashed line indicates duration of QT interval. QT corrected for heart rate (see Materials and Methods) was 396 and 467 msec for the exemplar wild-type and heterozygote, respectively. |

Unillustrated author statements |