- Title
-
Zebrafish fat-free is required for intestinal lipid absorption and Golgi apparatus structure
- Authors
- Ho, S.Y., Lorent, K., Pack, M., and Farber, S.A.
- Source
- Full text @ Cell Metab.
|
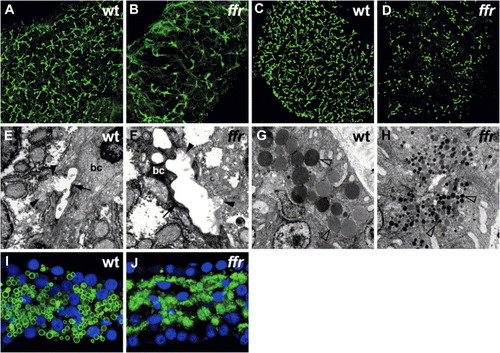
Phenotypic analysis of ffrmutants A and B) Confocal projections through the liver of 5 dpf wild-type and ffr larvae processed for cytokeratin immunohistochemistry. These immunostainings reveal widespread biliary degeneration in ffr mutants (5 dpf). C and D) Similar immunostainings of 5 dpf wild-type and ffr larvae processed for p-glycoprotein immunohistochemistry. These images of the wild-type and ffr liver show that canalicular architecture is also disrupted in the ffr mutants (5 dpf). E and F) Transmission electron micrographs showed degenerating biliary epithelial cells (bc) (indicated by arrow) and canaliculi (indicated by arrowhead) within the liver of ffr mutants (F) compared with wild-type (E). G and H) Electron micrographs of the pancreas reveal small zymogen granules in the ffr mutants (arrowhead; [H]) compared with wild-type (G). I and J) Confocal image from the pancreas of a 5 dpf ffr mutant (J) processed for carboxypeptidase A immunohistochemistry (green) showed altered distribution of this digestive enzyme compared with wild-type (I). Nuclei stained with Dapi (blue). PHENOTYPE:
|
|
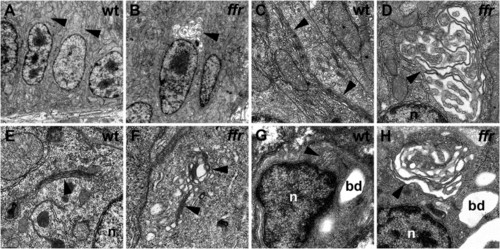
Ultrastructural analyses reveal abnormal Golgi apparatus in the ffr mutants A–D) Transmission electron micrographs show that intestinal Golgi architecture is disrupted in in 5 dpf ffr mutant larvae (B and D) compared to wild-type larvae (A and C). E–H) Golgi morphology is also disrupted in the 5 dpf ffr exocrine pancreas (F) and biliary cells (H), compared with wild-type (E and G) (5 dpf). n, nucleus; bd, bile duct lumen; arrowheads point to Golgi in all panels. PHENOTYPE:
|
|
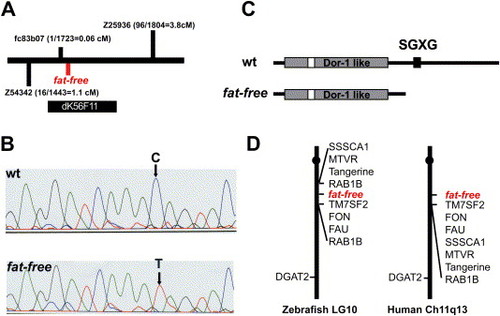
Positional cloning of the zebrafish ffr locus A) Schematic of the ffr locus. Marker fc83b07 is 0.06 cM from the ffr locus (1 recombinant out of 1732 meioses). A radiolabeled probe synthesized from fc83b07 identified five BAC clones. Mapping using the end sequences of these BAC clones indicated that clone dK56F11 spanned the entire ffr locus. B) A single nucleotide mutation (C to T) that results in a premature stop codon was identified in ffr mutants (0 recombinants > 2000 meioses) within a predicted open reading of dK56F11. C) Domain analysis indicates that Ffr has a Dor-1 like domain, a tyrosine phosphorylation site (Tyr-P), and a SGXG motif. The truncated mutant protein is predicted to be approximately half the length of wild-type Ffr. D) ffr is located on zebrafish chromosome 10 in a region syntenic to a region of human chromosome 11q13. |
|
Zebrafish Ffr protein is highly conserved from invertebrate to human A) The zebrafish Ffr protein sequence reveals 75% identity with human ortholog, and 46% identity with fly ortholog. B) Dor-1 like domain (amino acids 41–384) was identified using a prosite motif scan (http://us.expasy.org/prosite/) and returned a hit to Dor-1 (amino acids 41–384, raw-score = -111.2, N-score = 11.150, E-value = 0.00015; pfam Dor1–Dor1-like family). |
|
Phenocopy of the ffr mutation by targeted morpholino antisense oligonucleotides and rescue of the phenotype by ffr mRNA injection A) Genomic structure of the ffr gene. The white blocks represent the exons from 1–11. The morpholino antisense oligonucleotides were designed either targeting the AUG translation initiation codon (MO1) or at mRNA splice junctions (MO2, MO4, and MO5). B and C). ffr mutants (6 dpf) exhibit altered lipid absorption. Lipid processing was visualized by labeling with fluorescent cholesterol (NBD-cholesterol, 2 hr, 3 μg/ml solubilized with fish bile). Gall bladder fluorescence is absent in the ffr larvae. The ffr larva in (B) was indistinguishable from wild-type. D and E) Antisense targeting of ffr phenocopies the mutant phenotype. Embryos were injected with either water (control) or splice junction MOs (morphant) at the 1- to 4-cell stage. Larvae (5 dpf) were labeled with fluorescent cholesterol (NBD-cholesterol, 2 hr, 3 μg/ml solubilized with fish bile). Morphants exhibited decreased fluorescence in the digestive system. F) ffr mRNA injections can restore fluorescent cholesterol labeling of mutant larvae (p < 0.01). The offspring of heterozygous ffr parents were injected with ffr mRNA and phenol red, or with water and phenol red (control) at the 1- to 4-cell stage. Injected larvae (5 dpf) were immersed in the embryo medium containing fluorescent cholesterol (2 hr, 3 μg/ml solubilized with fish bile). Each larva was then photographed prior to molecular genotyping. Fluorescence intensity was digitally quantified as described in the Experimental Procedures. Data represent mean ± SEM. PHENOTYPE:
|
|
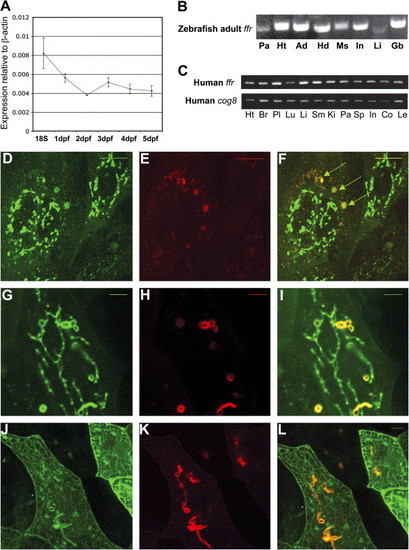
ffr expression and protein localization A) Developmental profile of ffr expression. mRNA levels were determined by a zebrafish oligo microarray. Normalized data are expressed relative to β-actin and from 18 somite to 5 dpf stages. Data represent mean ± SEM, (n = 3–6 at each stage). B) ffr is widely expressed in the adult zebrafish. Expression of ffr was determined by RT-PCR. Tissues: Pa, pancreas; Ht, heart; Ad, adipose tissue; Hd: head; Ms; muscle; In: intestine; Li: liver; Gb, gall bladder. C) Human ffr and COG8 are widely expressed. Expression was assayed by RT-PCR. Tissues: Ht, heart; Br, brain; Pl, placenta; Lu, lung; Li, liver; SM, skeletal muscle; Ki, kidney; Pa, pancreas; Sp, spleen; In, small intestine; Co, colon; Le, leucocyte. D–F) Ffr and COG8 partially colocalize in the Golgi of zebrafish blastomeres. Embryos were coinjected with ffr-gfp mRNA and cog8-mrfp mRNA at the 1- to 4-cell stage. D) Expression of Ffr-GFP in the perinuclear region. E) COG8-mRFP localizes primarily to the peripheral Golgi. F) Merged image of (D) and (E). Arrowheads indicate colocalization of COG8 and Ffr. G–I) Wild-type (in green) and mutant (in red) Ffr partially colocalize in the cells. (G) Expression of ffrWt-GFP. (H) Expression of ffr mut-mRFP. (I) Merged image of (G) and (H). Yellow indicates colocalization. J–L) Mutant Ffr (in red) and clathrin light chain (in green) colocalization. (J) TGN marker (Clathrin, GFP; pClGFP) (Gaidarov et al., 1999) expression in a single enveloping layer cell. (K) ffr mut-mRFP expression in a single cell. (L) Merged image of (J) and (K). Yellow indicates truncated Ffr mutant localized in the TGN. The scale bar represents 5 μm. EXPRESSION / LABELING:
|
|
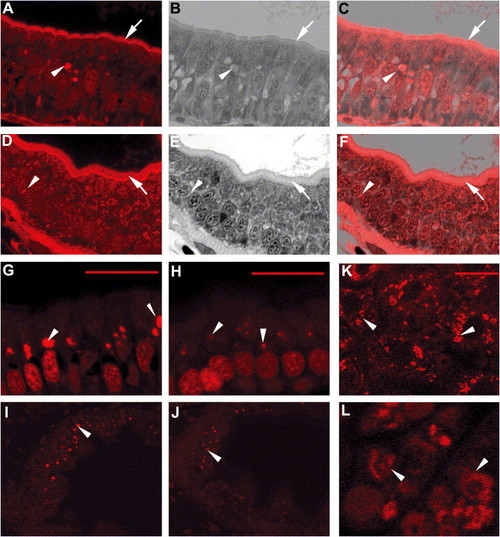
ffr mutants exhibit altered vesicular trafficking and APOA1 targeting A–F) Histological cross-sections through the intestine of 5 dpf ffr (A–C) and wild-type (D–F) larvae. Red fluorescence shows AM1-43 accumulation in plasma membrane (arrows) and intracellular vesicles (arrow head) in ffr mutant and wild-type enterocytes. (B and E) Identical sections in (A) and (D) stained with 1% methylene blue in 1% sodium borate and 1% Azure B (1:1). (C and F) Composites of panels (A) and (B), and (D) and (E), respectively. Note accumulation of AM1-43 in the region of the Golgi of ffr enteroctyes (C). This is best appreciated in histological cross-sections through the intestine of 5 dpf ffr and wild-type larvae following AM1-43 ingestion and cyclodextran wash to remove noninternalized dye within the plasma membrane (G–J). (K–L) Following ffr “knockdown” by MO-1, the apoA1-mRFP fusion protein had a diffuse intracellular distribution (arrowhead in [L]), whereas in control embryos the apoA1-mRFP fusion protein was present within vesicles surrounding the nucleus (arrowhead). The scale bar represents 15 μm. PHENOTYPE:
|
Reprinted from Cell Metabolism, 3(4), Ho, S.Y., Lorent, K., Pack, M., and Farber, S.A., Zebrafish fat-free is required for intestinal lipid absorption and Golgi apparatus structure, 289-300, Copyright (2006) with permission from Elsevier. Full text @ Cell Metab.