- Title
-
Shroom3 is required downstream of FGF signalling to mediate proneuromast assembly in zebrafish
- Authors
- Ernst, S., Liu, K., Agarwala, S., Moratscheck, N., Avci, M.E., Nogare, D.D., Chitnis, A.B., Ronneberger, O., and Lecaudey, V.
- Source
- Full text @ Development
|
Non-muscle myosin is required for apical constriction-driven proneuromast assembly. (A-D′′) Live images of the primordium of a 32 hpf zebrafish embryo co-expressing gfp-myh9 fusion (green) and lynTdTomato (red) mRNAs. B-D′′ are close-up views of the three boxed areas in A. (E-G′) Side view of the primordium of a 32 hpf cldnb:gfp embryo immunostained with Phospho-Myosin Light Chain 2 (ser19) (PMLC2) and GFP antibodies. F-G′ are close-up views of the two boxed areas in G. (H,I) Live images of the primordia of 32 hpf embryos treated with DMSO (H) or with 50 μM blebbistatin. Scale bars: 20 μm in A; 10 μm in B-D′′. EXPRESSION / LABELING:
|
|
shroom3 is expressed in the pLLP and localizes at the apical part of pLLP cells. (A) Schematic of Shroom3 showing different conserved domains of the protein and some of its main known interactors. (B) Schematic of the exon-intron composition of the two zebrafish shroom3 splice variants. (C-N) Lateral views of embryos stained by ISH with a shroom3 probe at 24 hpf (C-F), 36 hpf (G-J) and 48 hpf (K-N). shroom3 is expressed in the central domain of the migrating pLLP at all three stages (C-D′,G-H′,K-L′). It is also expressed in the ventral part of the otic vesicle (E,I,M) as well as in the distal part of the pronephros (F,J,N). (O-R′′) Lateral views of the pLLP (O-Q′′) or a recently deposited neuromast (P-R′′) in heat-shocked hsp70l:shr3v1-tagRFP (O-P′′) or hsp70l:shr3v2-tagRFP (Q-R′′) transgenic embryos. P′,P′′ and R′,R′′ are close-up views of the deposited neuromasts in P and R, respectively. Scale bars: 200 μm in C,G,K; 20 μm in D-F,H-J,L-N,O-O′′,Q-Q′&prime& 10 &m u;m in P,R; 5 μm in P′,P′′,R′,R′′. |
|
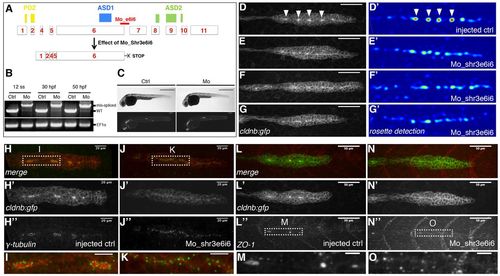
Shroom3 knockdown blocks apical constriction and rosette assembly in the migrating pLLP. (A) Schematic showing the position of Mo_Shr3e6i6. (B) RT-PCR with shr3- (top) and EF1α-(bottom) specific primers on zebrafish embryos injected with Mo_Shr3e6i6 or with water (ctrl) showing that Mo_Shr3e6i6 efficiently generates an alternative splice variant (mis-spliced) by blocking the excision of intron 6, potentially leading to a truncated protein lacking the poly-proline and ASD2 domain (A). (C) transmitted light and fluorescence pictures of embryos injected either with water (left) of with Mo_Shr3e6i6 (right). (D-G′) Live images of the primordium of cldnb:gfp embryos injected with water (D,D′) or with Mo_Shr3e6i6 (E-G′) showing that knocking down Shroom3 function blocks the assembly of apically constricted rosettes (arrowheads in D). D′-G′ are the corresponding detection response images (using Jet, a blue-to-red colour map) obtained from our rosette detector (see Materials and methods). (H-O) Side views of the primordium of 36 hpf cldnb:gfp embryos immunostained for the centrosomal marker γ-tubulin (H-K) or the tight-junction protein ZO1 (L-O). I, K, M and O are close-up views of the boxed areas in H, J, L′′ and N′′, as indicated. Scale bars: 50 μm in D-G′ ;10 μm in I,K,M,O. EXPRESSION / LABELING:
PHENOTYPE:
|
|
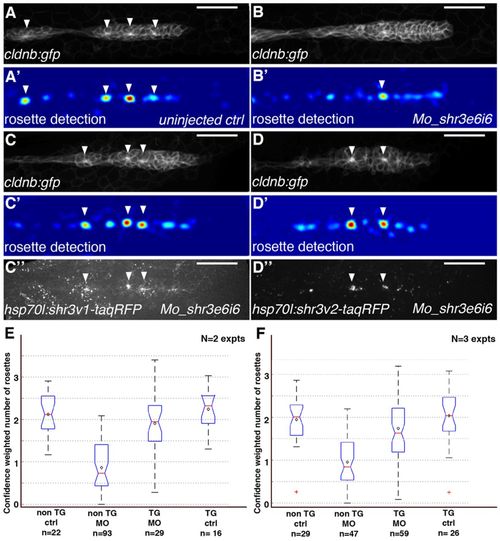
Induction of shroom3 expression rescues rosette assembly in Mo_Shr3e6i6-injected zebrafish embryos. (A-D′′) Images of the pLLP in heat-shocked non-transgenic siblings (A-B′), hsp70l:shr3v1-tagRFP (C-C′′) or hsp70l:shr3v2-tagRFP (D-D′′) embryos injected with water (A,A′) or Mo_Shr3e6i6 (B-D′′). A′-D′ show the corresponding detection response images. Scale bars: 50 μm. (E,F) Boxplots displaying the results and statistics of confidence-weighted rosette numbers from the trained rosette detector (see Materials and methods). The red line and black diamond represent the median and mean value of each group, respectively. The edges of the box are the 25th and 75th percentiles. The black dashed line extends to the most extreme data points not considered to be outliers, and outliers are plotted individually as red plus signs. The medians of two groups are significantly different at the 5% significance level if their respective notch intervals do not overlap (interval end points represent the extremes of the notches). N is the number of independent experiments and n is the number of embryos per condition. EXPRESSION / LABELING:
|
|
Rho kinases are also required for the assembly of pLLP in apically constricted rosettes. (A-F) Live images of the pLLP of zebrafish embryos treated with DMSO (A), or 50 μM (B), 100 μM (C,D) or 200 μM (E,F) of the Rho kinase inhibitor Rockout. (G) Boxplot of the quantification of the Rockout treatment experiments. Scale bars: 50 μm. EXPRESSION / LABELING:
|
|
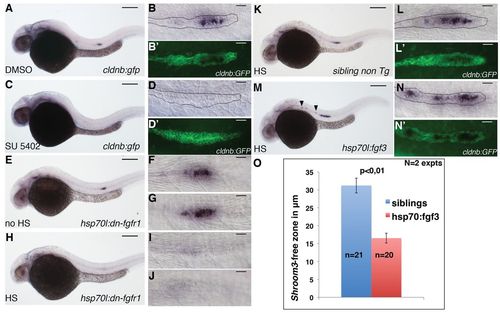
FGF signalling is necessary and sufficient for activating shroom3 expression in the pLLP. (A-N′) Lateral views of zebrafish embryos stained by ISH with a shroom3 probe. Treatment of embryos with 10 μM of SU5402 (C-D2), or overexpression of dn-fgfr1 (H-J) totally inhibits shroom3 expression in the pLLP (compare C-D2 with A-B′, and H-J with E-G). Overexpression of fgf3 after heat-shock of Tg(hsp70l:fgf3-Myc)zf115 embryos leads to an increase and expansion of shroom3 expression (compare M-N′ with K-L′). (O) Quantification of the length of the ′shroom3-free′ zone in the leading region in heat-shocked Tg(hsp70l:fgf3-Myc)zf115 (red) and non-transgenic siblings (blue). Statistical analyses were performed using the t-test. Error bars represent s.e.m. Scale bars: 200 μm in A,C,E,H,K,M; 20 μm in B,B′,D,D′,F,G,I,J,L,L′,N,N′. EXPRESSION / LABELING:
|
|
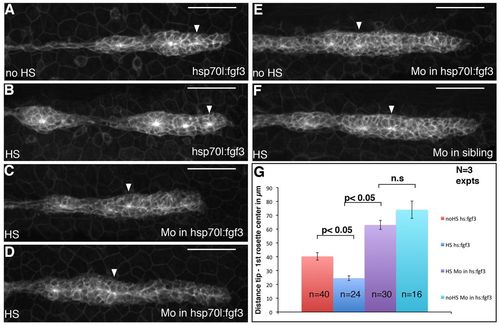
Shroom3 mediates FGF activity during assembly of apically constricted rosettes. (A-F) Pictures of the primordium of cldnb:gfp zebrafish embryos carrying a hsp70l:fgf3-Myc transgene (A-E) or of their non-carrier siblings (F). Embryos have been heat-shocked for 15 minutes (B-D,F) or not heat-shocked (A,E) and either injected with Mo_Shr3e6i6 (C-F) or injected with water (A,B). All embryos were kept at 25°C before and after heat-shock. Seven hours after HS, an ectopic rosette has formed in the leading region of Tg(hsp70l:fgf3-Myc)zf115 (B), but not in Mo_Shr3e6i6-injected siblings (C,D). (G) Quantification of the distance between the tip of the pLLP and the first rosette centre. Statistical analyses were performed using the t-test and error bars represent s.e.m. Scale bars: 50 μm. EXPRESSION / LABELING:
|
|
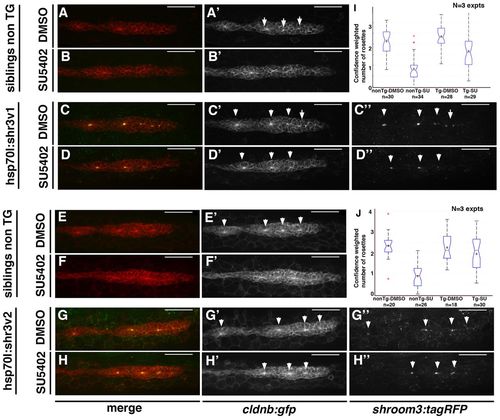
Shroom3 overexpression counteracts the loss of rosettes induced by FGFR inhibitor treatment. (A-H′′) Images of the pLLP in heat-shocked non-transgenic siblings (A-B′,E-F′), hsp70l:shr3v1-tagRFP (C-D′′) or hsp70l:shr3v2-tagRFP (G-H′′) zebrafish embryos treated with DMSO (A,C,E,G) or with 5 μM SU5402 for 4 hours (B,D,F,H). Treatment with 5 μM of SU5402 for 4 hours leads to a loss of apically constricted rosettes (compare B,F with A,E). Induction of shr3v1 (C,D) or shr3v2 (G,H) expression upon heat-shock blocks the loss of rosettes induced by SU5402 treatment (D,H). (I,J) Boxplots displaying the quantification of this experiment by the rosette detector for shr3v1 (I) and shr3v2 (J). Arrowheads indicate centres of apically constricted rosettes. Scale bars: 50 μm. EXPRESSION / LABELING:
|