- Title
-
Characterization of zebrafish mutants with defects in embryonic hematopoiesis
- Authors
- Ransom, D.G., Haffter, P., Odenthal, J., Brownlie, A., Vogelsang, E., Kelsh, R.N., Brand, M., van Eeden, F.J., Furutani-Seiki, M., Granato, M., Hammerschmidt, M., Heisenberg, C.P., Jiang, Y.J., Kane, D.A., Mullins, M.C., and N�sslein-Volhard, C.
- Source
- Full text @ Development

Mutations in the gene moonshine (mon) cause early defects in hematopoietic cell development leading to severe anemia. The relative strength of different alleles correlates with expression of GATA-1 mRNA detected by wholemount in situ hybridization. (A-D) Whole-mount in situ hybridization for GATA-1 expression in wildtype and mutant embryos at the 18 somite stage. (A) In wild-type embryos, GATA-1 is expressed in two stripes of hematopoietic cells that converge toward the dorsal midline to form the intermediate cell mass. (B) Embryos with the strongest allele, montb222b, express barely detectable levels of GATA-1. (C) The intermediate allele montc246b, results in a slightly increased GATA-1 expression. (D) The weakest allele, montc239b, has no obvious effect on GATA-1 expression at that stage. All embryos derived from a cross between montc239b heterozygous carriers looked the same as the embryos shown in D. (E-F) Isolated blood cells from wild-type and mutant larvae stained by the Wright-Giemsa method. (E) Blood cells from 5- day old wild-type larvae show the characteristic morphology of differentiated red blood cells. (F) The few blood cells circulating in 5- day old montb222b mutant larvae are large and basophilic. (G-H) Odianisidine staining for hemoglobin (arrow) in 2-day old wild-type and mutant embryos. (G) Hemoglobin in red blood cells is strongly stained in wild-type embryos. (H) In contrast, mon mutant embryos do not express detectable levels of hemoglobin. Scale bar (E,F) 20 �m. EXPRESSION / LABELING:
|
|
Mutations in the genes frascati (frs) and retsina (ret) cause a reduction in the number of circulating blood cells. The remaining blood cells also have an abnormal cell morphology. (A,C,E,G) Isolated blood cells from wild-type and mutant larvae stained by the Wright- Giemsa method. (B,D,F,H) O-dianisidine staining for hemoglobin (arrows) in wild-type and mutant embryos. (A) Blood cells from 2-day old wild-type embryos have the normal morphology of erythroblasts. (B) Hemoglobin in red blood cells is strongly stained in wild-type embryos. (C) The circulating cells of 2-day old frstq223 mutant embryos are larger than normal erythroid cells and many have abnormally fragmented nuclei. (D) The number of blood cells in 2-day old frstq223 mutant embryos is greatly reduced which is reflected in the lower amount of hemoglobin. (E) Blood cells of 3-day old wild-type larva are small erythroblasts. (F) The 3-day old wild-type larva also has a large number of red blood cells producing hemoglobin. (G) The circulating cells of a 3-day old rettr217 mutant larva are larger and more basophilic than normal. (H) The 3-day old rettr217 mutant larva expresses very little hemoglobin, based on o-dianisidine staining. Scale bar (A,C,E,G) is equal to 20 �m. PHENOTYPE:
|
|
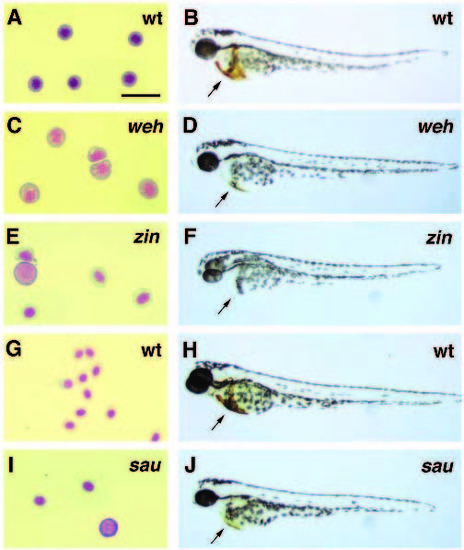
Mutations in the genes wei�herbst (weh), zinfandel (zin) and sauternes (sau) result in embryos with hypochromic blood and decreasing blood cell counts. (A,C,E,G,I) Isolated blood cells from wild-type and mutant larvae stained by the Wright-Giemsa method. (B,D,F,H,J) O-dianisidine staining for hemoglobin (arrows) in wildtype and mutant embryos. (A) Blood cells from 2-day old wild-type embryos have the normal morphology of late erythroblasts. (B) Wild type embryos express large amounts of hemoglobin in their differentiating red blood cells. (C) Blood cells from 2-day old wehtp85c mutant embryos are larger with open nuclei more typical of early proerythroblasts. (D) 2-day old wehtp85c mutant embryos have approximately 50% of the normal number of blood cells which express much lower levels of hemoglobin compared to wild-type blood cells. (E) Blood cells of 2-day old zinte207 mutant embryos are large but are more differentiated than the blood cells of weh mutant embryos. (F) 2-day old zinte207 heterozygotes (shown here) and homozygotes both lack detectable hemoglobin. (G) 3-day old wildtype embryos have mature erythrocytes with condensed nuclei and red staining hemoglobin. (H) A wild-type, 3-day old, embryo has large amounts of blood expressing hemoglobin. (I) Blood cells of 3- day old sauy121a mutant embryos are relatively normal in morphology, but they are reduced in number to between 25% and 50% of normal levels with a few large basophilic cells. (J) 3-day old sauy121a mutant embryos have fewer blood cells and thus a reduced total amount of hemoglobin expression. The scale bar in panel (A) is equal to 20 microns for panels (A,C,E,G,I). PHENOTYPE:
|
|
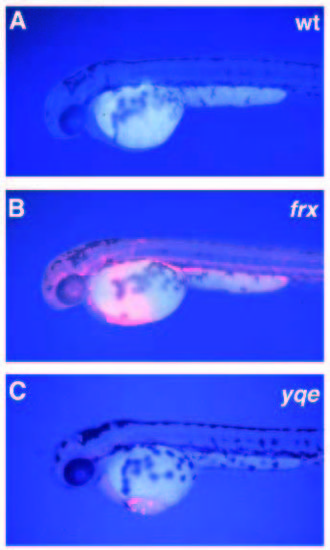
Mutations in the genes freixenet (frx) and yquem (yqe) cause erythropoietic porphyria phenotypes. Mutant and wild-type embryos were examined under 330 to 380 nm UV light for autofluorescence. (A) A wild-type 2- day old embryo does not have autofluorescent blood. (B) The blood of 2-day old frxtu271 homozygotes is brightly autofluorescent under the same conditions. (C) The blood of 2-day old yqetp61 homozygotes is also autofluorescent. In addition, the yqetp61 mutants rapidly develop pericardial edema when exposed to bright light. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|