- Title
-
Different Fgfs have distinct roles in regulating neurogenesis after spinal cord injury in zebrafish
- Authors
- Goldshmit, Y., Tang, J.K.K.Y., Siegel, A.L., Nguyen, P.D., Kaslin, J., Currie, P.D., Jusuf, P.R.
- Source
- Full text @ Neural Dev.
|
Fgf signalling increases neurogenesis after spinal cord injury. a Micrographs through intact non-injured adult zebrafish spinal cord show weak p-MAPK expression. b Micrographs through adult zebrafish spinal cord two weeks post injury (wpi) shows p-MAPK levels upregulated particularly in non-radial glia GFAP negative neurons at the central canal at the lesion site (arrows, B) (n = 5) some of which are Islet1 positive (c). d, e While Fgf signalling gain (spry4−/−) or loss (Tg(hsp70l:dn-fgfr1-EGFP) has no effect on NeuN+ neurons in intact spinal cord (SC), two weeks after injury, the significant increase in NeuN+ neurons in WT can be further increase with Fgf signaling gain and abolished with Fgf signaling loss. Graphs shows mean ± SEM, (n = 6 fish /group) ** p < 0.01. Scale bars in A, B and C are 25 μm, scale bar in D is 50 μm |
|
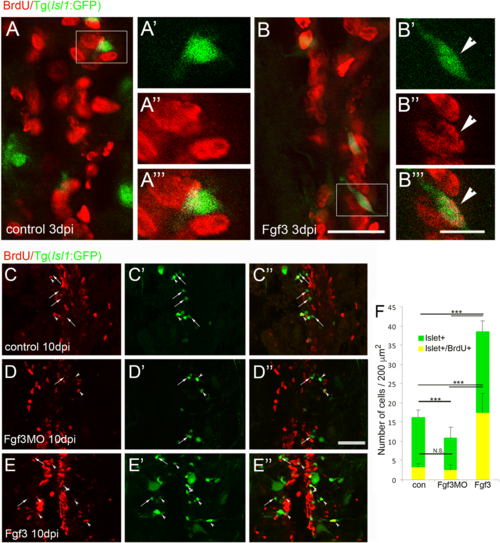
Fgf3 facilitates proliferation and neurogenesis of Islet1 motor neurons after spinal cord injury. a, b Three days post spinal cord injury (dpi), very few newly generated BrdU+ (red) cells express Islet1+ (green) motor neuron marker (a), unless treated with Fgf3 for three days (B, arrowhead). White box indicates region shown at higher magnification with individual and merged channels (A’ - A”’ and B’ – B”’). (C – F) Analysis of controls at 10 dpi shows that usually only a small proportion of newly generated BrdU+ cells usually become Islet1+ motor neurons (c, f). However, treatment with Fgf3 for three days facilitates both overall proliferation (increased number of BrdU+ cells) and specifically the proportion of newly generated cells that are becoming Islet1+ motor neurons (e, f), while overall Islet+ numbers, but not the newly generated BrdU+ cohort is significantly reduced when Fgf3 signalling is inhibited (d, f). Results in C show mean ± SEM, (n = 5 fish /group) *** p < 0.001; N.S.: not significant. Scale bar in B (for A and B) is 50 μm, scale bar in B”’ (for A’ – A”’ and B’ – B”’) is 10 μm and scale bar in F” (for D - F is 100 μm) |
|
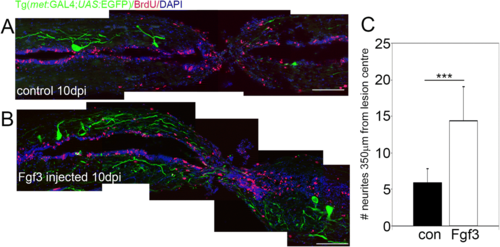
Fgf3 facilitates neurite sprouting of C-Met motor neurons after spinal cord injury. a, b Longitudinal sections through the spinal cord lesion site reveal that ten days post injury (dpi) Fgf3 treatment resulted in significantly more neurites at the lesion. Scale bars in A and B are 200 μm. c Quantitation of neurites up to 350 μm from lesion centre from both sides. Results are presented in C as mean ± SEM, (n = 7 fish /group) *** p < 0.001 |
|
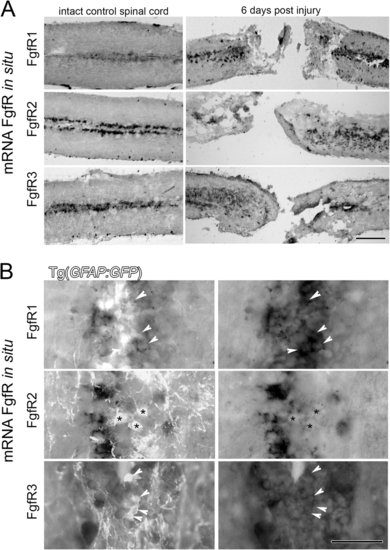
FgfRs expression around the central canal of the spinal cord on GFAP expressing radial glia. a Longitudinal sections show FgfR1–3 mRNA expression in cells at the central canal in intact uninjured spinal cord. At 6 days post injury (dpi) in situ hybridization shows an increase of the mRNA of all three FgfRs in these central canal cells particularly around the lesion site. b Sections from spinal cords in Tg(gfap:EGFP) transgenic fish show the location of GFAP+ ependymal radial glia cells and in situ hybridisation mRNA signal for FgfRs 1–3. At least some of the glia cells express varying levels of particularly FgfR1 and FgfR3 (arrowheads) with little overlap observed for FgfR2 (asterisks). Scale bar in A is 200 μm, Scale bar in B is 50 μm |
|
FgfRs expression in cells around the central canal and Islet1 and c-Met expressing motor neurons 6 days post spinal cord injury. a Sections from Tg(Isl1:GFP) transgenic fish showing the location of Islet1+ motor neurons compared to in situ hybridisation for FgfRs 1–3 mRNA. The middle and left panels are higher magnification insets of the boxes indicated in the left panels showing either FgfR signal alone (middle) or merged channels (right). At least some of the Islet1+ motor neurons express FgfR1 and 3 (arrowheads), but not FgfR2 (asterisks). b Sections from Tg(met:GAL4; UAS:EGFP) transgenic fish showing the location of c-Met+ motor neurons compared to in situ hybridisation for FgfRs 1–3 mRNA. Similarly as above, C-met neurons co-labelled with FgfR1 and 3, but not FgfR2 mRNA. The right panels show the merged C-met only (green - left panels) and in situ FgfR mRNA only (red - middle panels). Scale bar in A for left panels is 50 μm, and for middle and right panels is 10 μm. Scale bar in B is 50 μm |
|
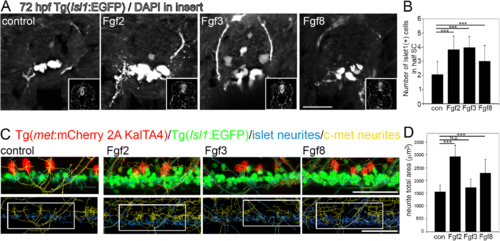
Fgfs mediate neurogenesis and neurite outgrowth during zebrafish development at three days postfertilisation. a Tranverse sections through Tg(Isl1:GFP) spinal cords after 48 h incubation in Fgf2, 3 or 8, showing Islet1+ motor neurons. Insets show DAPI nuclear labelling in lower right corner for each image. b Quantitation of half spinal cord in the sections at the level of the back-fin shows a significant increase in Islet1+ motor neurons following incubation in Fgf2, 3 or 8. Results are presented in B as mean ± SEM, (n = 10 fish/group)*** p < 0.001. c Representative images of longitudinal spinal cord images of double transgenic Tg(Isl1:GFP)/ Tg(c-met:mCherry) fish incubated for 48 h in Fgf2, 3 or 8. Upper panel shows Islet1+ (green) and c-Met+ (red) transgenic label with Islet+ neuritis computationally annotated by Imaris software traced in blue and c-Met neurites computationally annotated by Imaris software traced in yellow. Lower panel shows an example of region of interest taken for analysis. d Quantitation of neurite total area of Islet1+ GFP and c-Met+ mCherry neurites reveals a significant increase in neurite outgrowth following Fgf2 and to a lesser extent Fgf8, but not Fgf3 incubation. Results are presented in D as mean ± SEM, (n = 8 fish/group) *** p < 0.001, N.S. = not significant. Scale bar in A is 50 μm, scale bar in C is 50 μm |