- Title
-
Mir-126 is a conserved modulator of lymphatic development
- Authors
- Kontarakis, Z., Rossi, A., Ramas, S., Dellinger, M.T., Stainier, D.Y.R.
- Source
- Full text @ Dev. Biol.
|
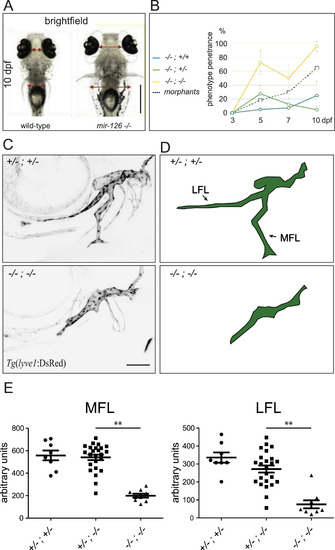
mir-126 mutant zebrafish develop edema and exhibit lymphatic hypoplasia. (A) Ventral view of wild-type and mir-126 mutant (mir-126a-/-; mir-126b-/-) siblings at 10 dpf. The strong periorbital and trunk edema is observed in almost all mutant larvae at this stage. (B) Penetrance of the edema phenotype in mutant and morphant larvae over time. Combined results from mir-126a-/-; mir-126b+/- and mir-126a+/-; mir-126b-/- incrosses. All animals appear unaffected at 3 dpf. The phenotype starts to become evident at 5 dpf and affects close to 100% of the mir-126a-/-; mir-126b-/- larvae by 10 dpf. A low penetrance of the edema can be seen in the other genotypes. mir-126 MO injected larvae exhibit a similar penetrance pattern. Error bars represent 95% confidence interval. 78 larvae analysed for each time-point (minimum 17 per genotype). (C,D) Representative confocal images and cartoons (side views, anterior to the left, dorsal up) of mir-126 heterozygous (mir-126a+/-; mir-126b+/-) and mir-126 mutant (mir-126a-/-; mir-126b-/-) facial lymphatic network at 5 dpf, highlighted using the Tg(lyve1:DsRed2) line. Lateral Facial Lymphatic (LFL) and Medial Facial Lymphatic (MFL) vessel development is affected in mutant larvae. (E) Quantification of LFL and MFL vessel length shows a significant reduction in mir-126 mutants. Single mutants do not show consistent reduction in vessel length. n mutants. Single mutan. Error bars represent Standard Deviation. Unpaired Student's t-test was performed to test for statistical significance. |
|
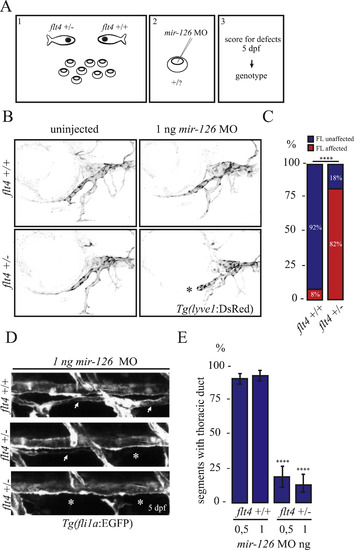
flt4+/- larvae are sensitive to mild miR-126 knockdown. (A) Design of the experiment: embryos from a flt4+/- outcross were injected with a low dose of mir-126 MO, scored for lymphatic defects at 5 dpf, and then genotyped. (B, C) Facial lymphatic analysis of flt4+/- and wild-type siblings injected with 1 ng of mir-126 MO. Uninjected larvae of both genotypes develop a functional facial lymphatic network. However, unlike their wild-type siblings, flt4+/- larvae display facial lymphatic defects (asterisk) when injected with 1 ng of mir-126 MO. Graph shows data from 3 independent experiments. Total number of larvae analysed: 73 (35 flt4+/- and 38 flt4+/+). (D,E) Thoracic duct (TD) analysis. Representative side views for normal, partial, and no TD development; asterisks indicate lack of TD formation; two suboptimal doses of mir-126 MO (0,5 ng and 1 ng) were used. Wild-type siblings display a largely complete TD; flt4+/- injected with mir-126 MO resemble mir-126 mutants. Results from 4 independent experiments; 16 larvae analysed per experiment. Unpaired Student's t-test was performed to test for statistical significance. PHENOTYPE:
|
|
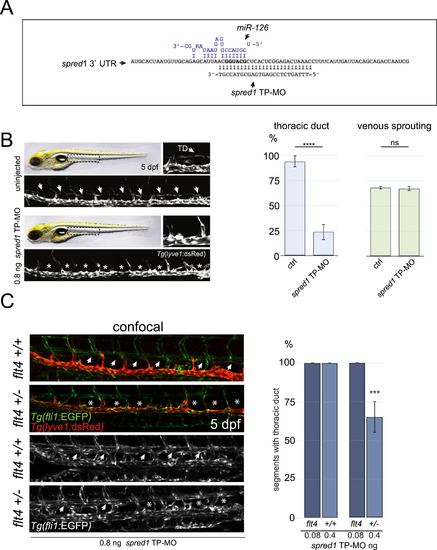
Blocking miR-126 dependent spred1 suppression phenocopies miR-126 loss. (A) miR-126 binding site in the spred1 3′UTR. “Seed” sequence in bold, miR-126 in blue. The target protector morpholino (TP-MO) was designed to mask the “seed” sequence with its 3′ end to minimise binding to other miR-126 targets. (B) Injection of spred1 TP-MO phenocopies miR-126 loss. The formed TD can be visualized in uninjected control Tg(lyve1:DsRed2) larvae at 5 dpf. Injection of 800 pg of spred1 TP-MO reduced TD formation without affecting vISV number. At least 24 injected embryos analysed per experiment; 3 independent experiments. (C) flt4+/- larvae are more sensitive to spred1 TP-MO injections. A significant reduction of TD formation can be observed in flt4+/- but not in wild-type siblings injected with 400 pg of spred1 TP-MO. Injection of 80 pg of TP-MO did not affect TD development in either genotype. Minimum 8 embryos analysed per genotype per treatment group; 2 independent experiments. Error bars represent SEM. Unpaired Student's t-test was performed to test for statistical significance. PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
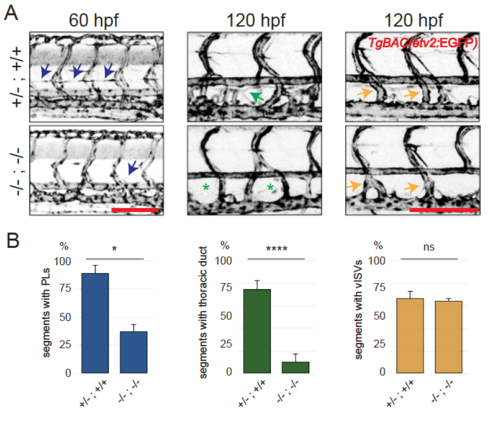
mir-126 mutants and morphants develop lymphatic hypoplasia. mir-126 mutant larvae display severe edema around the eyes and in the trunk starting at 5 dpf. (A) The lymphatic defects precede the onset of edema. mutant animals exhibit reduced parachordal lymphangioblast (PL) number at 60 hpf and thoracic duct (TD) hypoplasia at 120 hpf. Angiogenic secondary sprouting (vISV number) is not significantly affected. All images are maximal projections of 20–40 confocal sections, lateral views, anterior to the left. Arrows and asterisks indicate presence or absence of the trait of interest, respectively. (B) Quantification of segments with PL at 60 hpf, TD at 120 hpf and vISVs at 120 hpf. Minimum 5 animals per genotype were scored in each experiment per cross; data from> 2 independent crosses. Error bars represent SEM. Unpaired Student’s |
|
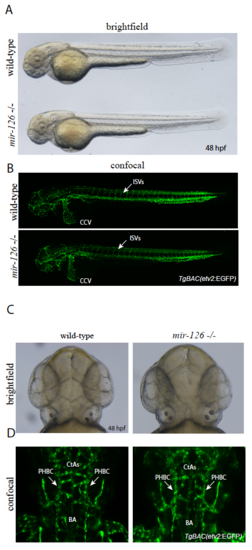
mir-126 mutants do not exhibit an obvious angiogenic phenotype. (A) Brightfield micrograph of 48 hpf wild-type sibling and mir-126-/- embryos in. lateral view. B) Confocal micrographs of 48 hpf TgBAC(etv2: EGFP) wild-type sibling and mir-126-/- embryos in lateral views; arrows point to ISVs in the trunk. (C) Brightfield micrographs of 48 hpf wild-type sibling and mir-126-/- embryos in dorsal views. D) Confocal micrographs of 48 hpf TgBAC(etv2: EGFP) wild-type sibling and mir-126-/- embryos in dorsal views; arrows point to PHBCs (primordial hindbrain channels). BA (basal artery), CtAs (central arteries). PHENOTYPE:
|
|
mir-126 MO injections cause lymphatic defects. (A) mir-126 MO injection inhibits TD formation; top, uninjected sibling; bottom, larva injected with 16 ng of mir-126 MO (brightfield and confocal images; 5 dpf animals shown). The TD is mostly missing in mir-126 morphants. (B) Representative quantification of TD formation (as percentage of segments with formed TD) and vISVs (venous sprouting). At least 16 injected larvae analysed per experiment; 4 independent experiments. PHENOTYPE:
|
|
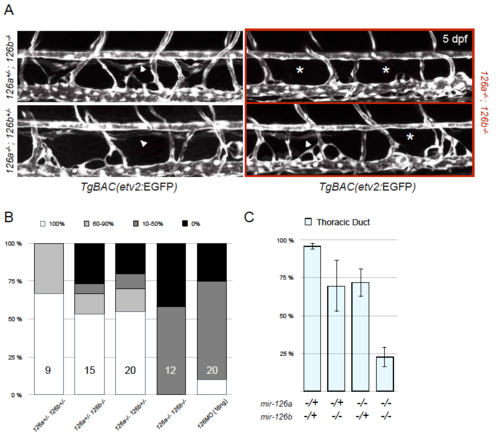
Penetrance of the thoracic duct phenotype in mir-126a and mir-126b mutants. (A) Confocal images of TD formation in mir-126a-/-; mir-126b+/-, mir-126a+/-; mir-126b-/-, and mir-126a-/-; mir-126b-/- larvae at 5 dpf. (B) Larvae were put into 4 categories according to TD formation (100%, 60–90%, 10–50%, and 0%). Double heterozygous larvae (mir-126a+/-; mir-126b+/-) show minimal TD defects. As more mir-126 alleles are “lost”, the penetrance of the phenotype increases. mir-126 morphants (16 ng) exhibit a similar penetrance as mir-126 mutants do. n, number of larvae for this particular experiment are indicated in the bars. (C) Plotting the average TD formation per genotype group shows that the phenotype is strong in the mir-126 population. Error bars are showing SEM. Graph shows data from one representative experiment. Analyses were done> 3 times and were consistent across all. PHENOTYPE:
|
|
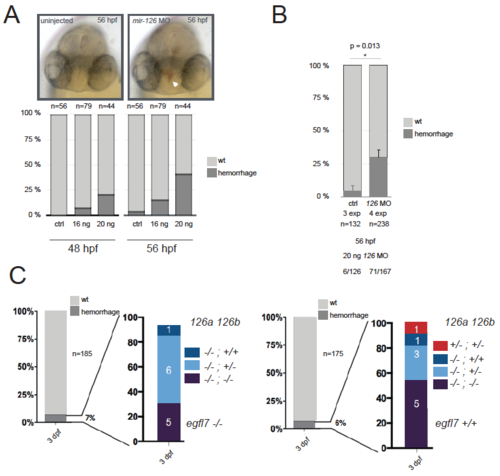
Vascular integrity is not obviously affected in mir-126 mutant embryos or mir-126; egfl7 compound mutants. (A,B) Injection of 16 or 20 ng of mir-126 MO results in brain haemorrhage by 48 hpf. (C) On the contrary, analysis of a population of mir-126 mutant larvae reveals a small fraction of larvae (not exclusively double mutant) exhibiting brain haemorrhage. Left and right graphs are for egfl7 mutant and wild-type backgrounds, respectively. PHENOTYPE:
|
|
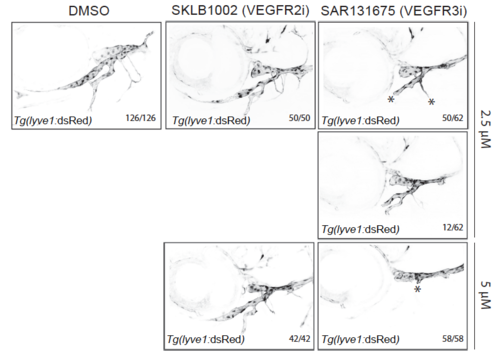
Flt4 but not Vegfr2 inhibition disrupts facial lymphatic development in zebrafish. Tg(lyve1:DsRed2) animals were treated with inhibitors for Vegfr2 (SKLB1002), Flt4 (SAR131675) or DMSO (equivalent to the highest inhibitor concentration) from 48 hpf until 5 dpf (as in Supplementary Figure 8) and facial lymphatic formation was scored at 5 dpf. DMSO controls and Vegfr2 inhibitor treated larvae (2.5 μM or 5 μM) showed no defects. SAR131675 treatment inhibited facial lymphatic formation at both concentrations tested; two severity categories for 2,5 μM are shown, all 5 μM SAR131675 treated larvae completely lacked facial lymphatic outgrowth. Data from 3 independent experiments. |
|
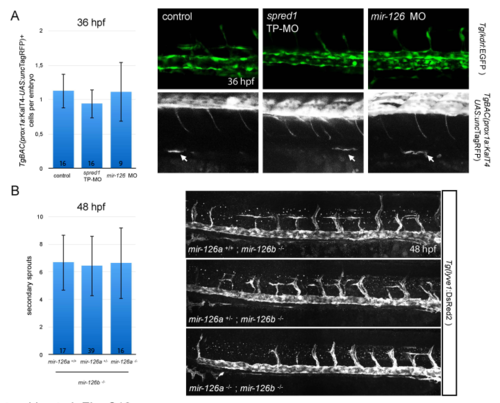
Early events of venous and lymphatic development in mir-126 and spred1 TP morphants. (A) Emergence of prox1+ cells in mir-126 (16 ng) and spred1 TP (800 pg) morphants at 36 hpf in the TgBAC(prox1a:KalT4-UAS: uncTagRFP) line. No observable reduction in TgBAC(prox1a:KalT4-UAS: uncTagRFP)+ cells was found between treatments; confocal pictures of live imaging; zero to three cells were found in the ventral part of the PCV in each embryo analysed; Error bars are SEM. (B) Secondary sprouting analysis in a Tg(lyve1:DsRed2) mir-126a+/-; mir-126b-/- incross at 48 hpf, after immunostaining for DsRed. Similar to our observations at 5 dpf, mir-126 mutants display secondary sprouts from the PCV; two independent crosses. Error bars are SEM. |
Reprinted from Developmental Biology, 437(2), Kontarakis, Z., Rossi, A., Ramas, S., Dellinger, M.T., Stainier, D.Y.R., Mir-126 is a conserved modulator of lymphatic development, 120-130, Copyright (2018) with permission from Elsevier. Full text @ Dev. Biol.