- Title
-
Ciliopathy genes are required for apical secretion of Cochlin, an otolith crystallization factor
- Authors
- Leventea, E., Zhu, Z., Fang, X., Nikolaeva, Y., Markham, E., Hirst, R.A., van Eeden, F.J.M., Malicki, J.J.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
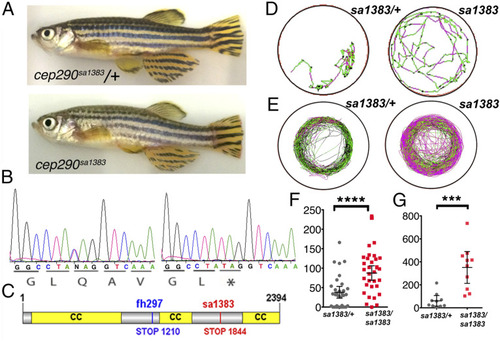
cep290 mutant phenotype. (A) Phenotype of homozygous cep290sa1383 adult fish. (B) Sequence of the sa1383 mutant allele used in this study. (C) Cep290 domain structure showing mutations used in this study. (D and E) Example swimming tracks of heterozygote control (Left) and homozygous mutant (Right) larvae after osmotic shock (D) and adults (E); black represents slow movement or static, green represents medium, and red represents fast movement. (F and G) Total distance traveled for swimming trajectories (mm) such as ones shown in (D and E) for larvae after osmotic shock (F) and adults (G). ****P < 0.0001; ***P < 0.001; Student’s t test; 95% CI. |
|
Otolith crystallization defects in cep290 mutant homozygotes. (A) Example scanning electron microscopy images of wild-type and cep290sa1383 otoliths at 18 mo (Scale bar, 300 μm). Enlargements in Bottom panels (Scale bar, 150 μm) (n = 3). (B) Side views of otic vesicles in live wild-type and cep290sa1383 homozygous embryos at 36 hpf. In the posterior ear, in the mutant example, two closely apposed otoliths are seen, where one is expected. Anterior is Left, dorsal Up, arrowheads indicate otoliths. (C) Frequency of otolith defects at 36 hpf. P < 0.001, χ2 test. (D) Typical example of cobalt nitrate staining of utricular otoliths in wild-type and cep290sa1383 mutants (n = 20). |
|
Otolithic Cochlin deficiency in mutants of cilia genes and cochlin mutant phenotype. (A) Otolithic proteins from wild-type and cep290sa1383 homozygotes separated on a polyacrylamide gel and visualized with Coomassie blue staining. The arrow indicates Cochlin band. (B) Domain structure of the cochlin gene. LCCL, Limulus factor C, Cochlin, Lgl1 domain; vWF, von Willebrand Factor domains. Deletions in CRISPR-induced mutants are indicated. The antiserum against Cochlin was made against the C terminus (amino acids 390 to 553) (C) Otolith morphology in morphant and cochsh536 mutant embryos as indicated. ATG-targeted morpholino (ATG) was used. Shown are side views of otic vesicles in live embryos, the arrows point at otoliths that are abnormal. (D) Scanning electron microscopy images of typical otoliths from wild-type and coch sh536 mutant adult at 7 mo of age (Scale bars, overview 300 μm, blow up 150 μm) (n = 3). (E) Example of cobalt nitrate staining of utricular otoliths from wild-type and coch sh536 mutant at 7 mo (n = 3). |
|
Otolith phenotypes of BBS mutants and genetic interactions between cep290 and bbs loci at 36 hpf. (A and D) Schematic representation of Bbs2 and Bbs9 protein domain structure. Sites of mutations in bbs2sa2952 and bbs9sa14425 alleles used in this study are indicated. (B), bbs2−/− mutant phenotype. Lateral views of otic vesicles from (i) bbs2+/− outcross to a wild-type strain and (ii) bbs2+/− incross. In (ii) an individual with abnormal otolith morphology is shown. (iii) Frequency of otolith defects in embryos originating from bbs2+/− outcross and incross. (P < 0.0001, χ2 test) (C) Phenotype of embryos originating from bbs2+/− outcrosses to a wild-type and a cep290−/− strain. Lateral views of otic vesicles from (i) bbs2+/− outcross to a wild-type stain and (ii) bbs2+/− outcross to cep290sa1383 homozygous strain. (iii) Frequency of otolith defects in bbs2+/− outcrosses. (P < 0.0001, χ 2 test). (E) Phenotype of bbs9−/− mutant homozygotes. Lateral views of the otic vesicle from (i) bbs9+/− heterozygote and (ii) bbs9−/− mutant homozygote with abnormal otolith number (3 instead of 2). (iii) Frequency of otolith defects in bbs9−/− homozygotes, compared to bbs9+/− heterozygotes (P < 0.0001, x2 test). (F) Phenotype of embryos originating from bbs9+/− outcrosses to a wild-type and a cep290−/− strain. Lateral views of otic vesicles from (i) bbs9+/− outcross to a wild-type and (ii) bbs9+/− outcross to cep290−/− homozygous strain. (iii) Frequency of otolith defects in bbs9+/− outcrosses as above (P < 0.0001, χ 2 test). The arrows indicate abnormally forming otoliths. (G) Examples of cobalt nitrate staining of utricular otoliths from bbs2−/− (H) and bbs9−/− (i) mutants at 23 and 26 mo of age, respectively (n = 3). |
|
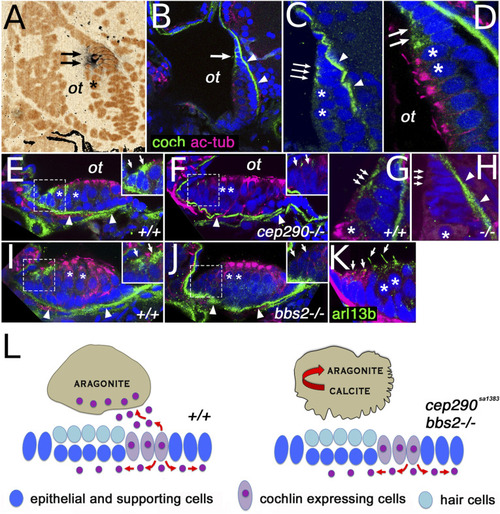
Cochlin expression pattern. (A) cochlin transcript expression (arrows) in the posterior macula of the zebrafish otic vesicle at 5 dpf. (B and C) Cochlin protein expression (in green) in the posterior macula at 5 dpf. (D) Cochlin expression (in green, arrows) at the dorsal rim of the posterior macula in cells immediately adjacent to hair cells (asterisks). (E–H) Cochlin protein localization in anterior (E and F) and posterior (G and H) maculae of wild-type and cep290sa1383 homozygotes as indicated at 5 dpf. (I and J) Cochlin expression in the anterior macula of wild-type (I) and bbs2−/− mutants (J). (K) Cilia visualized with Arl13b-GFP transgene (in green, arrows), Cochlin in red. (L) Model of cep290 and bbs2 function in Cochlin secretion. Loss of cep290, bbs2, and most likely other cilia-related genes leads to defects apical Cochlin secretion into the ear lumen and causes abnormal crystallization of otolithic calcium carbonate. In (D–K), sections are double stained for acetylated tubulin (in magenta), which accumulates apically in hair cells. All sections are counterstained with DAPI in blue (pseudocolored in orange in A). Insets in (E, F, I, and J) show enlargements of Cochlin-secreting cells (enclosed in dashed line boxes). In (B–J), the arrows indicate Cochlin-secreting cells and the arrowheads point to Cochlin presence in the basal lamina. “ot” otolith position, asterisks indicate hair cells. CaCO3 crystal structure after ref. 36. |