- Title
-
Tribulus terrestris L. Extract Protects against Lipopolysaccharide-Induced Inflammation in RAW 264.7 Macrophage and Zebrafish via Inhibition of Akt/MAPKs and NF-κB/iNOS-NO Signaling Pathways
- Authors
- Zhao, W.R., Shi, W.T., Zhang, J., Zhang, K.Y., Qing, Y., Tang, J.Y., Chen, X.L., Zhou, Z.Y.
- Source
- Full text @ Evid. Based Complement. Alternat. Med.
|
The cytotoxicity of BJL and BJL-decreased LPS-induced NO release in RAW 264.7 cells. (a) RAW 264.7 cells were treated LPS (0.3 μg/ml) with or without various concentrations of BJL (3, 10, and 30 μg/ml) for 24 h and then followed by NO release assay. (b) RAW 264.7 cells were treated with various concentrations of BJL (3, 10, 30, 50, 80, and 100 μg/ml) for 24 h and the cell viability was measured by MTT assay. (c) RAW 264.7 cells were treated LPS (0.3 μg/ml) with or without various concentrations of BJL (3, 10, and 30 μg/ml) for 24 h and the cell viability was measured by MTT assay. Data were presented as the percentages of the control group. Results were means ± S.E.M. of more than 3 independent experiments. p<0.001 versus the control group. *p<0.05, **p<0.01, and ***p<0.001 versus the LPS-treated group. |
|
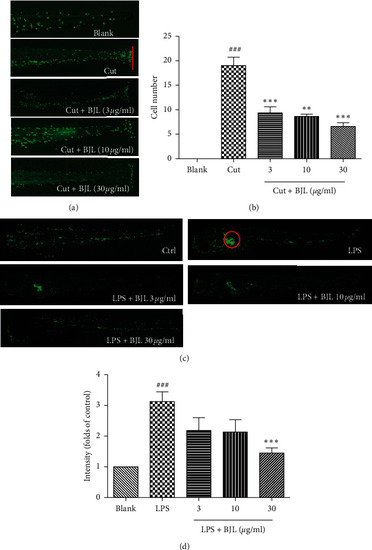
BJL ameliorated fin transection or LPS-induced migration and aggregation of the neutrophils in zebrafish. (a-b) The transgenic zebrafish line Tg(MPO:GFP) which expresses green fluorescence protein in neutrophils was employed in this experiment. Three DPF zebrafish embryos were treated with various concentrations of BJL (3, 10, and 30 μg/ml) for 2 h and then zebrafish embryos were cut a part of the fin using a sharp needle and treated with BJL for another 2 h The photos were taken under a fluorescent stereoscopic microscope. The red line indicates the amputation site. The migrated cell numbers of neutrophils were calculated in each zebrafish embryo. (c-d) Three DPF zebrafish embryos were injected with LPS (0.3 μg/ml) and then treated with various concentrations (3, 10, and 30 μg/ml) of BJL for 24 h. The red circle indicates the injection site. The fluorescence intensity of the injection site was calculated by Image J data which were presented as the folds of the control group. Results were the means ± S.E.M. of more than 3 independent experiments. p<0.05 and p<0.001 versus the blank control group. **p<0.01 and ***p<0.001 versus the LPS-treated group. |
|
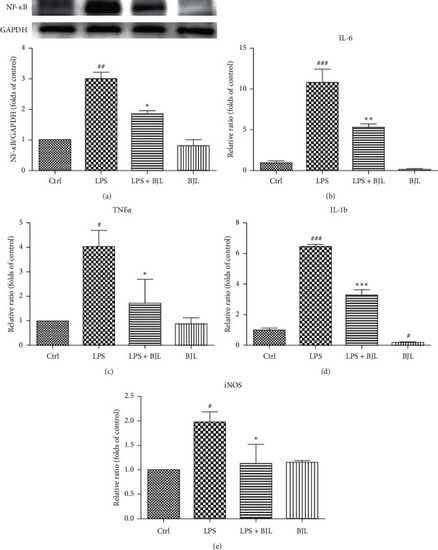
BJL partially inhibited LPS-induced upregulations of NF-κB, iNOS, and proinflammatory cytokines in RAW 264.7 cells. RAW 264.7 cells were treated LPS (0.3 μg/ml) with or without BJL (30 μg/ml) for 24 h. (a) The intracellular protein level of NF-κB was tested by western blotting using the specific primary antibody. (b–e) The mRNA expression levels of TNF-α, IL-6, IL-1β, and iNOS were detected by real-time PCR. Data were presented as the folds of the control group. Results were means ± S.E.M. of more than 3 independent experiments. p<0.055, p<0.01, and p<0.001 versus the control group. *p<0.05, **p<0.01, and ***p<0.001 versus the LPS-treated group. |
|
BJL suppressed LPS-stimulated activation of Akt/MAPK signaling cascades in RAW 264.7 cells. RAW 264.7 cells were treated LPS (0.3 μg/ml) with or without BJL (30 μg/ml) for 24 h and then followed by western blot analysis. Data were presented as the folds of the control group. Results were means ± S.E.M. of more than 3 experiments. p<0.05 and p<0.01 versus the control group. p<0.05 and p<0.01 versus the LPS-treated group. |
|
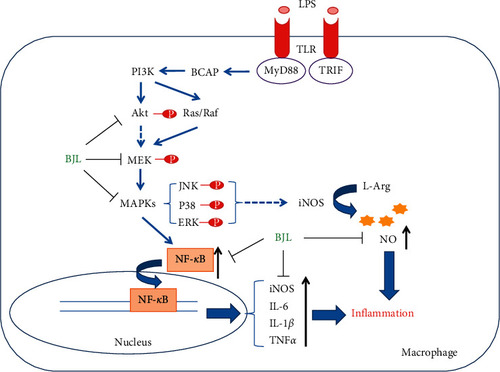
Schematic overview of the mechanism underlying the anti-inflammatory effect of BJL. |