- Title
-
The Regenerating Adult Zebrafish Retina Recapitulates Developmental Fate Specification Programs
- Authors
- Lahne, M., Brecker, M., Jones, S.E., Hyde, D.R.
- Source
- Full text @ Front Cell Dev Biol
|
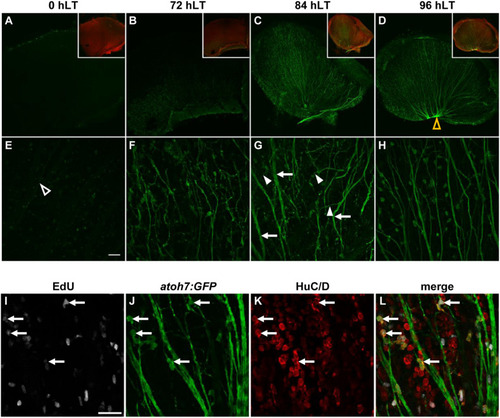
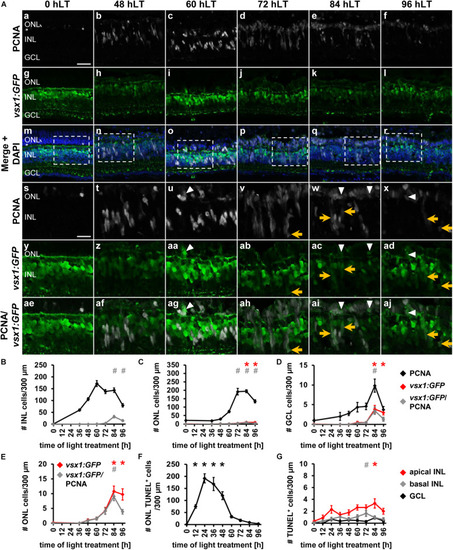
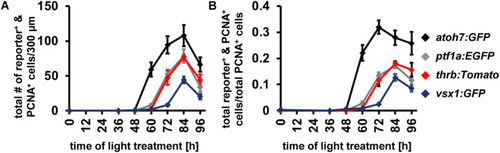
Generation of all neuronal cell types and expression of cell type specific developmental competence factors in the light-damaged retina. |
|
Ganglion cell competence factor |
|
Newly generated ganglion cells extend axons. |
|
Amacrine and horizontal cell competence factor |
|
Competence factor of red cone precursor cells, |
|
Bipolar cell competence factor |
|
Comparison of the temporal expression patterns of neuronal competence factors in the light-damaged retina. |
|
Expression of developmental competence factors and generation of all neuronal cell types following rod photoreceptor cell death in a genetic ablation model. |
|
Comparison of the temporal expression patterns of neuronal competence factors in the NMDA-damaged retina. |