- Title
-
A zebrafish model for FHL1-opathy reveals loss-of-function effects of human FHL1 mutations
- Authors
- Keßler, M., Kieltsch, A., Kayvanpour, E., Katus, H.A., Schoser, B., Schessl, J., Just, S., Rottbauer, W.
- Source
- Full text @ Neuromuscul. Disord.
|
(A) Box shade diagram of amino acid sequence of human FHL1 and both zebrafish orthologs fhl1a and Fhl1b. Amino acid sequences of zebrafish Fhl1a and Fhl1b are highly conserved from zebrafish to humans, showing a 67% for Fhl1a and 60% for Fhl1b overall amino acid identity with human FHL1, and a 100% conservation within the essential Zinc-binding LIM domains (light blue boxes). (B) Whole-mount RNA in situ hybridization using a fhl1a (left) and fhl1b (right) antisense probe. Both, fhl1a and fhl1b, localize to skeletal muscle. fhl1a is detectable abundantly already at the 18-somite stage of embryonic development in the skeletal muscle, whereas fhl1b localizes to slow muscle cells and the pectoral fin muscles at 72 hpf. (C) Immunhistochemistry of zebrafish skeletal muscle. Merge of FHL1 and Nexilin immunohistochemistry and DAPI staining. FHL1 localizes to z-discs and nucleus. Nexilin localizes to z-discs. DAPI staining of the nucleus. (D) Whole-mount RNA in situ hybridization using a fhl1a (left) and fhl1b (right) antisense probe. Both, fhl1a and fhl1b, localize to the heart. EXPRESSION / LABELING:
|
|
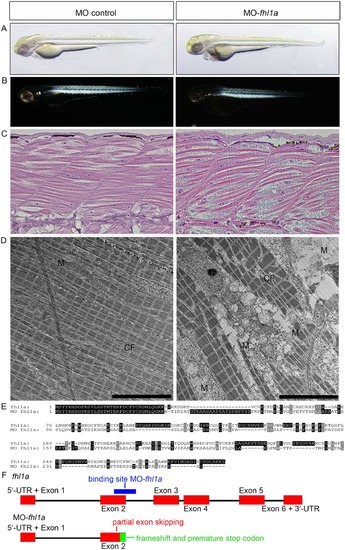
(A) Phenotype of fhl1a-morphants. fhl1a-morphants (right) display skeletal muscle myopathy and cardiomyopathy in 73% of injected zebrafish embryos (n = 160 embryos in total, three independent experiments) at 72 hpf compared to control-injected zebrafish embryos (left). In fhl1a-morphants skeletal muscle is reduced and of irregular morphology in light microscopy. Regarding cardiomyopathy, cardiac contractility is impaired and a pericardial edema as well as blood congestion is present in fhl1a-morphants. (B) In the birefringence assay of wild-type zebrafish embryos at 72 hpf (left) the well-organized skeletal muscle appears bright. In contrast, skeletal muscle of fhl1a-morphants (right) displays a general reduction of birefringence consistent with myopathic muscle. (C) H&E staining of skeletal muscle of control-injected and fhl1a-depleted zebrafish embryos at 72 hpf. Myofibrillar disarray and a reduced content of contractile fibers in the skeletal muscle of fhl1a-morphants compared to wild-type zebrafish embryos. (D) Ultrastructural analysis by transmission electron microscopy of control-injected zebrafish and fhl1a-morphants. The content of contractile fibers (CF) is reduced in fhl1a-morphants compared to control zebrafish embryos at 72 hpf. Intermyofibrillar degeneration including enlargement of the sacroplasmic reticulum in fhl1a-morphants and subsarcolemmal and intermyofibrillar accumulation of dysmorphic mitochondria (M). (E, F) Amino acid sequence alignment and schematic illustration of Fhl1a and MO fhl1a. Morpholino-modified antisense oligonucleotide-mediated knockdown (binding site, blue) leads to a partial exon skipping of 103 nucleotides of exon 2 (red), causing a frameshift and predicted to cause an early truncation after exon 2 of Fhl1a (green region in Fig. 2F). PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
(A) Schematic structure of FHL1. One Zinc finger domain (ZF) and four LIM domains (double Zinc finger motif, LIM1-LIM4). Three common human mutations are marked in red (H123Y, C132F and C224W). (B) Western blot of zebrafish embryos at 24 hpf (left) and in-vitro-translatate (IVT, right) staining, Pan Cadherin C as control and myc-expressing FHL1 with myc-antibody. mRNA of wild-type FHL1 and its mutated forms is translated into stable protein in zebrafish embryos and therefore can be detected in FHL1-mRNA injected zebrafish embryos but not in control embryos. (C) Phenotype of either control-injected or fhl1a-depleted zebrafish embryos. The lower four embryos are fhl1a-depleted and either injected with wild-type FHL1-mRNA or mutated FHL1-mRNA. When MO-fhl1a and wild-type (wt) FHL1-mRNA are co-injected, the phenotype resembles wild-type zebrafish. The skeletal muscle (SM) of MO-fhl1a and wild-type FHL1-mRNA co-injected embryos displays regular structural organization. Heart function is normalized (see Fig. 4E). In contrast to human wild-type FHL1, the mutated human FHL1-proteins (FHL1-H123Y, FHL1-C132F and FHL1-C224W) are not capable of suppressing the FHL1-opathic phenotype in fhl1a–depleted zebrafish embryos. These embryos exhibit severe myopathy with severe functional impairment and morphological disruption, compromised heart function (see Fig. 4E) and pericardial edema (PE). (D) Quantification of the FHL1-opathic phenotype of the injected embryos. The majority of fhl1a-depleted zebrafish embryos expressing mutated FHL1 exhibit FHL1-opathic phenotype. Only human wild-type FHL1 protein is capable of suppressing the FHL1-opathic phenotype. Rate of FHL1-opathic phenotype is 78% ± 1% (n = 130 embryos in total, three independent experiments, p = 0.4457) in MO-fhl1a + FHL1-H123Y, 76% ± 2% (n = 130 embryos in total, three independent experiments, p = 0.6967) in MO-fhl1a + FHL1-C132F and 67% ± 1% (n = 130 embryos in total, three independent experiments, p = 0.4993) in MO-fhl1a + FHL1-C224W, compared to 73% ± 4% (n = 130 embryos in total, three independent experiments) of FHL1-opathic embryos in the fhl1a-morphants and only 34% ± 8% (n = 230 embryos in total, three independent experiments, p = 0.0232) in MO-fhl1a + wild-type FHL1. (E) Cardiac function could not be rescued and heart rate was not normalized in fhl1a-morphants, when mutated human FHL1 is expressed in these morphants. In contrast, heart rate of fhl1a-morphants expressing human wild-type FHL1 normalized. Heart rate remained decreased to 74 ± 8 bpm (n = 20 embryos in total, three independent experiments, p = 0.5918) in MO-fhl1a + FHL1-H123Y, 84 ± 14 bpm (n = 20 embryos in total, three independent experiments, 0.3142) in MO-fhl1a + FHL1-C132F and 70 ± 13 bpm (n = 20 embryos in total, three independent experiments, p = 0.6933) in MO-fhl1a + FHL1-C224W, compared to 89 ± 8 bpm (n = 20 embryos in total, three independent experiments) in fhl1a-morphants and 151 ± 1 bpm (n = 20 embryos in total, three independent experiments) in wild-type control embryos. In contrast, the heart rate of fhl1a-morphants could be normalized to 130 ± 3 bpm (n = 20 embryos in total, three independent experiments; p < 0.0001) by overexpressing human wild-type FHL1. |
|
Phenotype of overexpression of human FHL1-H123Y, FHL1-C132F and FHL1-C224W in wild-type zebrafish embryos. (A) Overexpression of human wild-type FHL1 did not lead to a specific, FHL1-opathic phenotype in wild-type zebrafish embryos compared to control-injected zebrafish embryos at 72 hpf. (B) Heart rate of zebrafish embryos overexpressing either mutated or wild-type human FHL1 did not significantly differ from control zebrafish embryos. |
|
Phenotype of mismatch Morpholino oligonucleotide (MO-fhl1a MM)-control-injected zebrafish embryos at 72 hpf. Absence of heart and skeletal muscle abnormalities in the control-injected zebrafish embryos (B) compared to wild-type embryos (A) confirms specificity of the MO-oligonucleotide mediated knockdown of fhl1a. |
|
Spontaneous movement assay with false-colored superimposed overviews of 24 hpf control of control-injected embryos and of the fhl1a-morphants. 85% ± 3% (n = 20 embryos per experiment, three independent experiments) of wild-type embryos moved spontaneously within a timeframe of 10 seconds. In contrast, only 22% ± 2% (n = 20 embryos per experiment, three independent experiments, p < 0.0001) of the fhl1a-morphants exhibited spontaneous movement. PHENOTYPE:
|