- Title
-
Canonical Wnt Signaling Dynamically Controls Multiple Stem Cell Fate Decisions during Vertebrate Body Formation
- Authors
- Martin, B.L., and Kimelman, D.
- Source
- Full text @ Dev. Cell
|
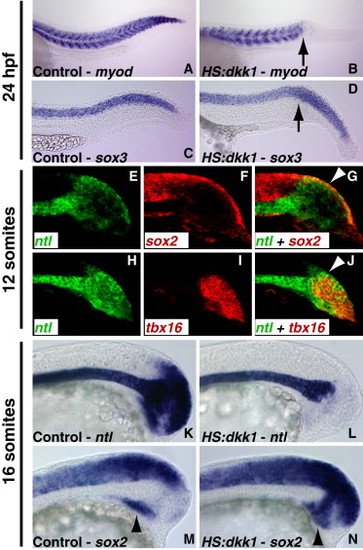
Canonical Wnt Signaling Promotes Mesodermal and Inhibits Neural Development during Postgastrulation Development (A–D) The HS:dkk1 transgenic line was used to inhibit canonical Wnt signaling during postgastrula stages. (A and B) myod expression in wild-type and transgenic embryos that were heat-shocked at bud stage and fixed at 24 hpf. In transgenic embryos, there is a severe truncation of the mesodermally derived somites (B, arrow), despite the overall length of the embryo being similar to wild-type embryos. (C and D) sox3 expression in wild-type and transgenic embryos. An enlarged neural tube is present in the posterior embryo, beginning where somites are absent (D, arrow). (E–G) Cells within the stem zone (SZ; G, arrowhead) express both the mesodermal progenitor marker ntl and the neural progenitor marker sox2. (H–J) The cell population expressing both ntl and sox2 is different than cells that have committed to the mesodermal lineage, as marked by tbx16 expression (J, arrowhead showing nonoverlapping expression of ntl in the SZ). (K–N) Wild-type and HS:dkk1 transgenic embryos were heat-shocked at the 12-somite stage and fixed at the 16-somite stage (3 hr after the heat shock). (K and L) The expression of the mesodermal progenitor marker ntl is completely lost in the SZ of transgenic embryos, (M and N) whereas the expression of the neural progenitor marker sox2 is significantly expanded. The ntl notochord domain is unaffected by Wnt inhibition. Note the unique ventral sox2 domain (arrowheads). See also Figure S1. |
|
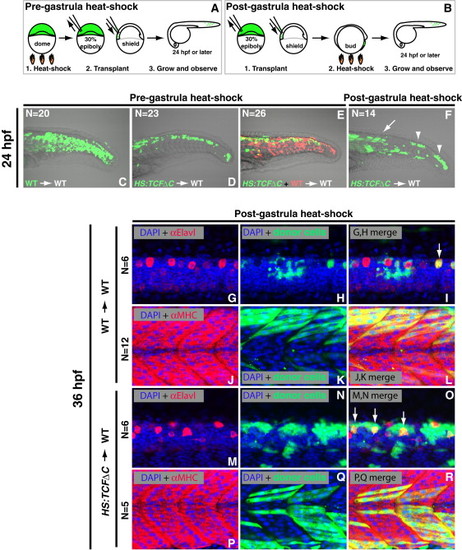
Wnt Signaling Is Cell-Autonomously Required for Tail Bud Stem Cells to Join Somites throughout Body Formation (A and B) Experimental design for cell-transplants examining the pregastrula (A) and postgastrula (B) requirements for Wnt signaling. (C and D) Fluorescein dextran labeled donor cells were transplanted into the ventral margin of shield stage unlabeled wild-type host embryos. (C) Control cells contribute normally to posterior somites. (D) Cells from HS:TCFΔC donor embryos heat-shocked at the pregastrula (dome) stage before transplantation are unable to contribute to somites, and instead reside within the neural tube. (E) Double transplantation of wild-type (red) and HS:TCFΔC (green) cells that were heat-shocked at dome stage and cotransplanted into the ventral margin of wild-type host embryos. Wild-type cells contribute normally to somites whereas cells lacking Wnt function cannot join the posterior somites and instead contribute to the neural tube. (F) Host embryos containing fluorescein dextran labeled HS:TCFΔC cells transplanted into the ventral margin and heat-shocked at the postgastrula (bud) stage have an intermediate phenotype, with anterior cells contributing normally to somites (arrow) and posterior cells transfating to neural tissue (arrowheads). (G–R) The ability of transplanted cells to differentiate into muscle or neurons was verified using antibodies directed against Myosin Heavy Chain or Elavl, respectively. (G–L) Wild-type cells labeled with fluorescein dextran (green) transplanted into the ventral margin of unlabeled wild-type host embryos and heat-shocked at bud stage primarily form muscle (L) with the occasional differentiated neuron (I, arrow), as indicated by the overlap of red fluorescent antibody staining and the green fluorescence of the transplanted donor cells. (M–R) HS:TCFΔC cells labeled with fluorescein dextran were transplanted into unlabeled wild-type host embryos and heat-shocked at bud stage. Transplanted cells in the posterior of the embryo differentiated as neurons (O) and not muscle (not shown), whereas the early differentiating cells in the anterior regions contributed to muscle (R). See also Figures S2 and S3. |
|
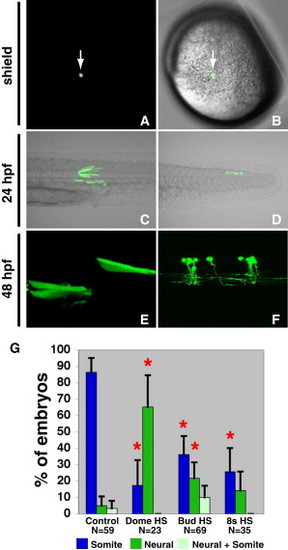
A Single Cell Transplantation Assay Demonstrates that Wnt Signaling Specifies Mesodermal Fate in Bipotential Neural/Mesodermal Stem Cells throughout Body Formation (A and B) Host embryos were visualized under a fluorescent microscope immediately after the single cell transplant to verify that only one cell was transplanted (A, arrow). An overlay with the bright field image illustrates the position of the single cell in the ventral margin of the host embryo (B, arrow, animal pole of the embryo is to the top). (C and D) Single cell transplantations into the ventral margin produce lineage restricted clones, primarily giving rise to clones within the somites (C) with some contributing only to the neural tube (D). (E and F) Single HS:TCFΔC cells transplanted into wild-type host embryos and heat-shocked at bud stage produce morphologically normal muscle fibers (E) and spinal cord neurons (F). (G) Quantification of one-cell transplants of control or HS:TCFΔC indicate that Wnt signaling is required continuously throughout development to specify mesoderm in a bipotential neural/mesodermal stem cell. Inhibiting Wnt signaling before or after gastrulation causes a significant decrease in clones that contribute to the somites and a significant increase in the clones that give rise to spinal cord neurons (p < 0.05 indicated by red asterisk). Nonmuscle mesoderm (see Figure 4) is not included in these graphs so the totals do not add up to 100%. See also Figure S4. |
|
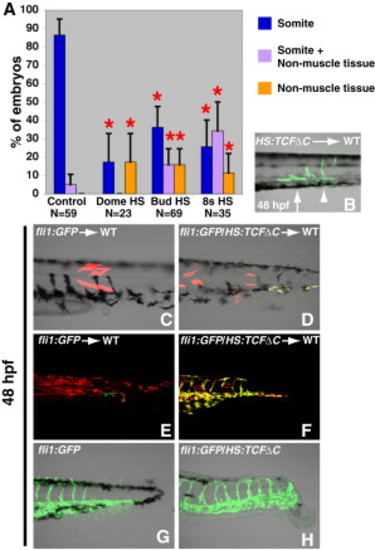
Wnt Signaling Specifies Paraxial Fate in Bipotential Paraxial/Endothelial Mesodermal Progenitors (A) In the absence of Wnt signaling, there is a significant shift in the resident location of cells from the somites to the nonmuscle mesoderm (note that the data for the somite clones is the same as that presented in Figure 3G, data for neural clones can be found in Figure 3). (B) An example of a single cell HS:TCFΔC transplant that was heat-shocked at the 8-somite stage, where several cells in the anterior of the clone have contributed normally to somites (arrow), whereas the cells in the posterior have contributed to a ventral mesoderm fate that appears to be vascular endothelium (arrowhead). (C and D) Rhodamine-labeled single cells were transplanted from the indicated donor and heat shocked at bud stage. (D) Wnt inhibition causes some of the progeny of the single cell to express the vascular marker fli1 (seen as yellow from the overlap of GFP and rhodamine). (E and F) Rhodamine-labeled cells were transplanted from the indicated donor and heat shocked at the 8-somite stage. (F) Wnt inhibition causes many of the transplanted cells to adopt a vascular fate instead of a muscle fate. (G and H) fli1:GFP and fli1:GFP/HS:TCFΔC embryos were heat shocked at the 8-somite stage. (H) Wnt inhibition causes a large increase in the amount of posterior vasculature. |
|
Mesodermal Progenitors of the Tail Bud Normally Contribute to Posterior Endothelial Tissue (A) Double fluorescent in situ staining of gata1 (green, arrow) and sox2 (red, arrowhead) indicates that the sox2 positive cells of the tail bud are distinct from the gata1 expressing intermediate cell mass. (B) Double fluorescent in situ staining of ntl (green) and sox2 (red) indicates that sox2 positive cells coexpress ntl in both the ventral domain (arrow) and in the SZ (arrowhead). (C–J) fli1:gfp transgenic embryos were injected with nuclear localized kaede mRNA (C and D). A small population of cells were photoconverted from green to red in the region of the ventral sox2 expressing cells, (G and H) and in the SZ region of sox2 expressing cells. (E, F, I, and J) In both cases, cells gave rise to both somites and vascular endothelium (arrows indicate red nucleus in a cell with cytoplasmic GFP expression from the fli1:gfp). |
|
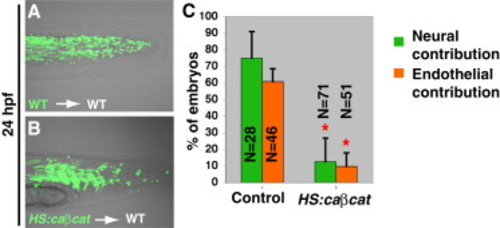
Wnt Signaling Is Sufficient to Specify Paraxial Mesoderm in Multipotent Stem Cells (A and B) Fluorescein labeled wild-type or HS:caβcat cells were heat-shocked and then transplanted into the ventral margin of shield stage unlabeled wild-type embryos. (A) In control transplants, the majority of cells contribute to the posterior somites, but a small number also contribute to the posterior spinal cord. (B) In transplants with activated Wnt signaling, cells contribute to somites but not spinal cord. (C) Host embryos containing transplanted cells with activated Wnt signaling have a significantly reduced likelihood of cells contributing to neural or endothelial fates (as determined by transplanting fli1:GFP or fli1:GFP/HS:caβcat), compared with controls (p < 0.05 indicated by red asterisk). See also Figure S2. |
|
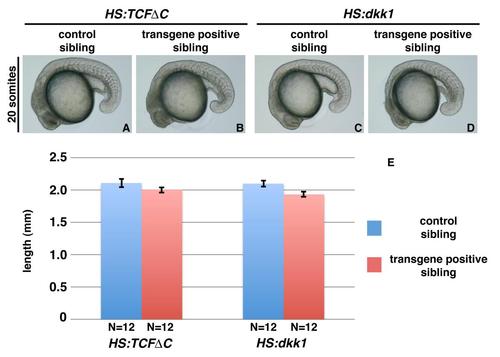
Post-gastrula loss of Wnt signaling has a minor effect on overall body length, related to Figure 1 (A-D) HS:TCFΔC and HS:dkk1 adults were outcrossed and the resulting progeny were heat-shocked at bud stage. Pictures of transgene negative and positive live sibling embryos were taken at the 20-somite stage. (E) Length from the tip of the head to the tip of the tail was measured using ImageJ64 software. Average length comparisons between control transgene negative and transgene positive embryos indicate only a very minor, statistically significant (P<0.01 for both HS:TCFΔC and HS:dkk1) change in overall body length. Error bar values for the graph represent the standard deviation. |
|
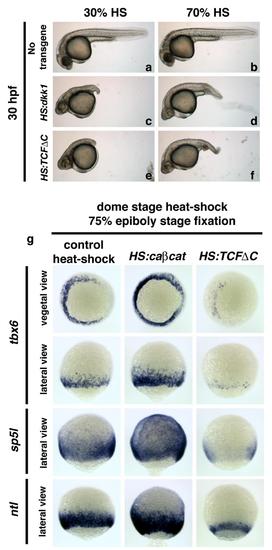
Newly created HS:TCFΔC and HS:caβcat transgenic lines have expected downstream effects on embryonic development, related to Figures 2 and 6 In order to confirm the specificity of the HS:TCFΔC transgenic line, side by side heat-shocks were done with the HS:dkk1 transgenic line and analyzed for gross morphology or gene expression. The HS:TCFΔC embryonic phenotype is exactly the same as HS:dkk embryos (e and f compared to c and d). (g) In order to confirm that the newly created transgenic lines are affecting the expression of downstream Wnt target genes, the expression of several known direct targets of canonical Wnt signaling was examined. Embryos were heat-shocked at dome stage and fixed at 75% epiboly for tbx6, sp5l, and ntl expression (Martin and Kimelman, 2008; Szeto and Kimelman, 2004; Weidinger et al., 2005). In all cases, there was a strong increase in expression of target genes in HS:caβcat embryos, and a strong decrease in expression in HS:TCFΔC embryos. |
|
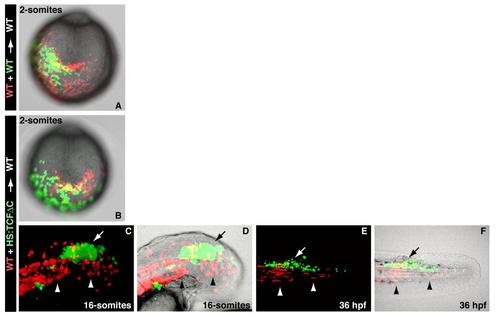
Morphogenetic defects in cells lacking Wnt signaling are not apparent until mid-somitogenesis, related to Figure 2 (A) A control experiment in which wild-type fluorescein and wild-type rhodamine cells were co-transplanted into an unlabeled wild-type host embryo and then examined at the 2-somite stage. (B) Double transplants of wild-type (red) and HS:TCFΔC (green) cells heat-shocked before gastrulation do not display noticeable differences at the 2-somite stage (A and B are posterior end-on views with dorsal to the top). (C, D) A high magnification view of a host embryo at the 16-somite stage in which wild-type (red) and HS:TCFΔC (green) cells heat-shocked before gastrulation were transplanted into the ventral margin. The majority of the wild-type cells have joined the hypoblast (arrowheads), while the HS:TCFΔC cells remain in the epiblast (arrow)(D is the image in C with the brightfield image overlaid). (E and F) At 36 hpf, the embryo from C and D shows that the wild-type cells that joined the hypoblast have given rise mostly to muscle fibers of the somites (arrowheads), while HS:TCFΔC cells that remained in the epiblast have contributed to the spinal cord (arrow) (F is the image in E with the brightfield image overlaid). |
|
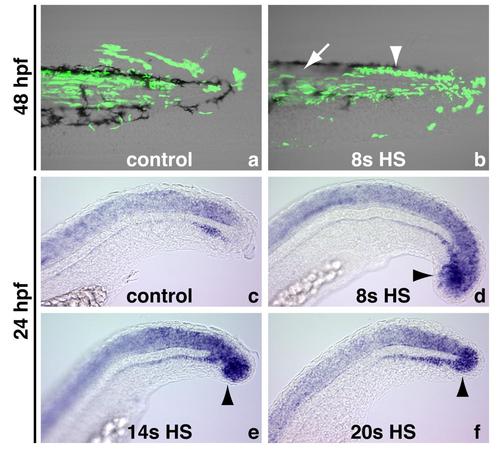
Wnt signaling is continuously required for fate decisions throughout body formation, related to Figure 3 (A) Control or (B) HS:TCFΔC cells were transplanted into the ventral margin of shield stage unlabeled wildtype hosts and heat-shocked at the 8-somite stage. (B) As with earlier stage heat-shocks, the posterior-most cells transfate to spinal cord (arrowhead) while more anterior cells contribute normally to somites (arrow). The ventral posterior-most cells also transfate to vascular endoethelium. (C-F) sox2 expression in 24 hour old control or HS:dkk1-EGP embryos. (C) At 24 hours, sox2 expression is not present in the posterior-most region of the embryo. Heat-shocking embryos at the (D) 8-somite, (E) 14-somite, or (F) 20-somite stage causes an expansion of sox2 positive tissue (arrowheads). |
Reprinted from Developmental Cell, 22(1), Martin, B.L., and Kimelman, D., Canonical Wnt Signaling Dynamically Controls Multiple Stem Cell Fate Decisions during Vertebrate Body Formation, 223-232, Copyright (2012) with permission from Elsevier. Full text @ Dev. Cell