- Title
-
Spinal Projection Neurons Control Turning Behaviors in Zebrafish
- Authors
- Huang, K.H., Ahrens, M.B., Dunn, T.W., and Engert, F.
- Source
- Full text @ Curr. Biol.
|
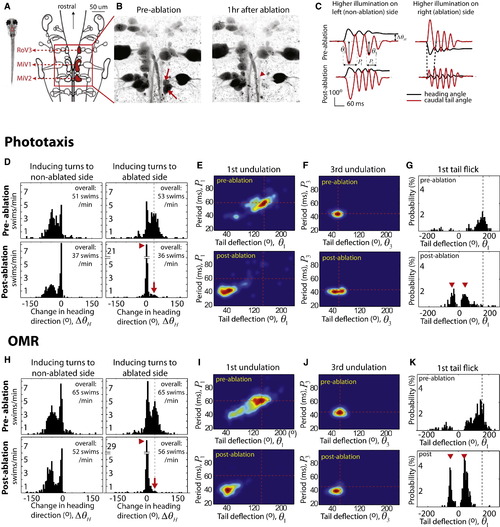
Laser Ablation of vSPNs Specifically Affects the First Undulation Cycle and Promotes Forward Swims during Phototaxis and the OMR (A) Schematic of hindbrain spinal projection neurons (SPNs) of larval zebrafish (image modified from [11]). (B) Two right MiV2 cells before and after laser ablation (arrows). The nearby ventral branch of the medial longitudinal fascicle (arrowhead) remains intact. (C) Example of ablation phenotypes. Visually induced right turns are replaced by forward swims after ablation of the vSPNs on the right (right panels). The amplitude of the first tail bend (Θ1) is weaker, and the period (P1) of the first undulation is reduced. Turning to the nonlesion side is unaffected (left panels). (D) Histograms of the change in heading direction (ΘΔH) before and after vSPN ablation. The unilateral ablation abolishes turning to the lesioned side (red arrow) while drastically increasing the occurrence of forward swims (red arrowhead). Data were collected from the same eight fish to plot the histograms in (D)–(K). (E and F) Analysis of tail movements during phototaxis, represented by a 2D histogram with tail deflection (Θ) plotted on the x axis and cycle period (P) plotted on the y axis. The ablation affected the first undulation cycle (E), but not the later cycles (F). Dotted red line indicates the position of the preablation maximum. (G) Histograms of the angle of the initial tail bend. The amplitude of bends toward the lesioned side is greatly reduced after the ablation. Instead, small-angle bends on either side of the body are performed (red arrowheads). (H–K) During the OMR, the ablation also specifically affects the first undulation cycle of tail movements and promotes forward swims. See also Figure S2. |
|
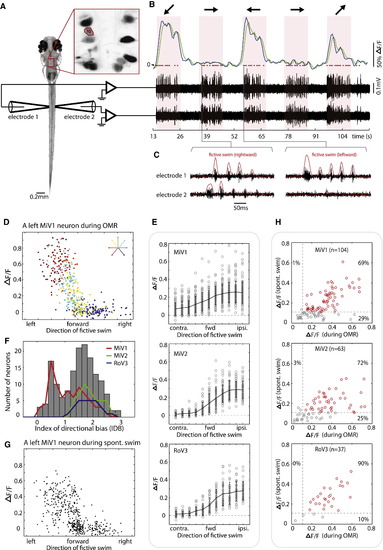
vSPNs Show Graded Responses with Respect to Turn Angle and Show Correlated Activity for Visually Evoked and Spontaneously Occurring Swim Events (A) Simultaneous recordings of motor nerve signals and hindbrain neuron activity reported by calcium imaging. (B) A left MiV1 cell (marked “10” in inset of A) backfilled with calcium green dextran responds strongly to left and backward left gratings and weakly to a forward right grating. Region-of-interest averaged fluorescence time series is shown in green, and the deconvolved trace is shown in blue. Motor nerve activity (black traces) was recorded bilaterally to identify fictive swims (red dots). (C) Motor nerve signals shown at higher resolution. Examples of a right turn (left panel) and a left turn (right panel) are shown. (D) Fluorescent calcium response (ΔF/F) of a MiV1 neuron as a function of swimming direction. Each dot represents a discrete swimming event, and the color indicates the visual stimulus used to elicit the swim. The cell exhibits a rectifying-shaped activation profile. (E) Activation profile of the three nuclei (83, 47, and 29 neurons in MiV1, MiV2, and RoV3 nuclei, respectively). Swim direction was divided into 11 bins, and each circle indicates the average ΔF/F of a neuron in the swim direction. Error bars represent SD. Only responsive neurons are analyzed (ΔF/F > 0.12; MiV1, 83 of 104 cells; MiV2, 47 of 63 cells; RoV3, 29 of 37 cells). (F) Analysis of the directional bias of all vSPNs reveals two functional groups. (G) Calcium responses of the same neuron shown in (D) during spontaneous fictive swimming. The same rectifying shape is apparent. (H) The majority of the vSPNs that are active during the OMR are also active during spontaneous swimming. Each circle represents a neuron. A threshold of ΔF/F > 0.12 (dashed lines) is used to define active cells. Neurons above thresholds are shown in red; neurons below thresholds are shown in gray. See also Figures S3 and S4. |