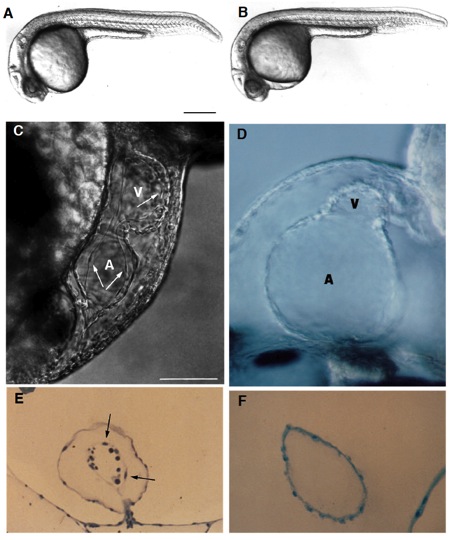
Fig. 1 Live wild-type (A) and homozygous clom39 mutant (B) zebrafish embryos at 24 hours (all times are in hours postfertilization at 28.5°C; clo indicates the locus of the mutation, and m39 is the allele designation). At this time and at this magnification, a slight edema is noticeable in the pericardial region of the mutant embryo but no other abnormalities are apparent. Indeed, development appears normal until the onset of the heart beat by the 26-somite stage (22 hours). From that time onwards, the heart exhibits a weak beat and becomes abnormally enlarged, especially the atrium. Edema becomes very pronounced by day 2. There is no circulation, although cells are present in the trunk vessels (see Fig. 3). (C) In a normal heart at around 40 hours, the myocardium and endocardium (arrows) are clearly distinct. Endocardial cells are the thin elongated cells that form the inner layer of the heart. The clo mutant heart only has a single layer of cells (as shown in a medial view of the heart (D), see also F), determined to be the myocardium by its spontaneous contractility, and also morphologically, ultrastructurally, and biochemically, using monoclonal antibodies (mAbs) against myosin heavy chain and tropomyosin (Stainier and Fishman, 1992) (see also Figs 2 and 4). The main chambers (atrium (A) and ventricle (V)) are distinct morphologically and biochemically (using chamber specific mAbs against myosin heavy chain; data not included) (Stainier and Fishman, 1992) and contract sequentially. In the mutant heart, the chambers are dysmorphic, the atrium is abnormally enlarged and the ventricle is collapsed. Some mutant hearts undergo normal looping morphogenesis. (E) Transverse sections of the atrium of a normal heart at 3 days showing the myocardium and endocardium (arrows) as well as some blood cells (dark round cells) and of a clo mutant heart at 3 days (F) showing the presence of a single layer of cells, previously determined to be the myocardium. Scale bars, 200 mm (A,B); 50 mm (C-F). [In Fig. 1 C and E, a silent heart (sih) mutant embryo is shown as the wild-type control. Because there is no circulation in the clo mutant embryo, we wanted to distinguish the specific effects of the clo mutation from the indirect effects resulting from a lack of circulation. For this purpose, we examined the phenotype of another mutant, the sih mutant (isolated in Eugene, Oregon by C. Walker and C. Kimmel), which, as the name indicates, exhibits no heart beat, and thus no blood flow. We previously determined at the molecular level that the sih mutation is cardiac specific and is thus a good model to examine the indirect effects resulting from a lack of circulation (Stainier et al., in preparation). The sih mutant heart is morphologically similar to the wild-type heart.]
Image
Figure Caption
Figure Data
Acknowledgments
This image is the copyrighted work of the attributed author or publisher, and
ZFIN has permission only to display this image to its users.
Additional permissions should be obtained from the applicable author or publisher of the image.
Full text @ Development