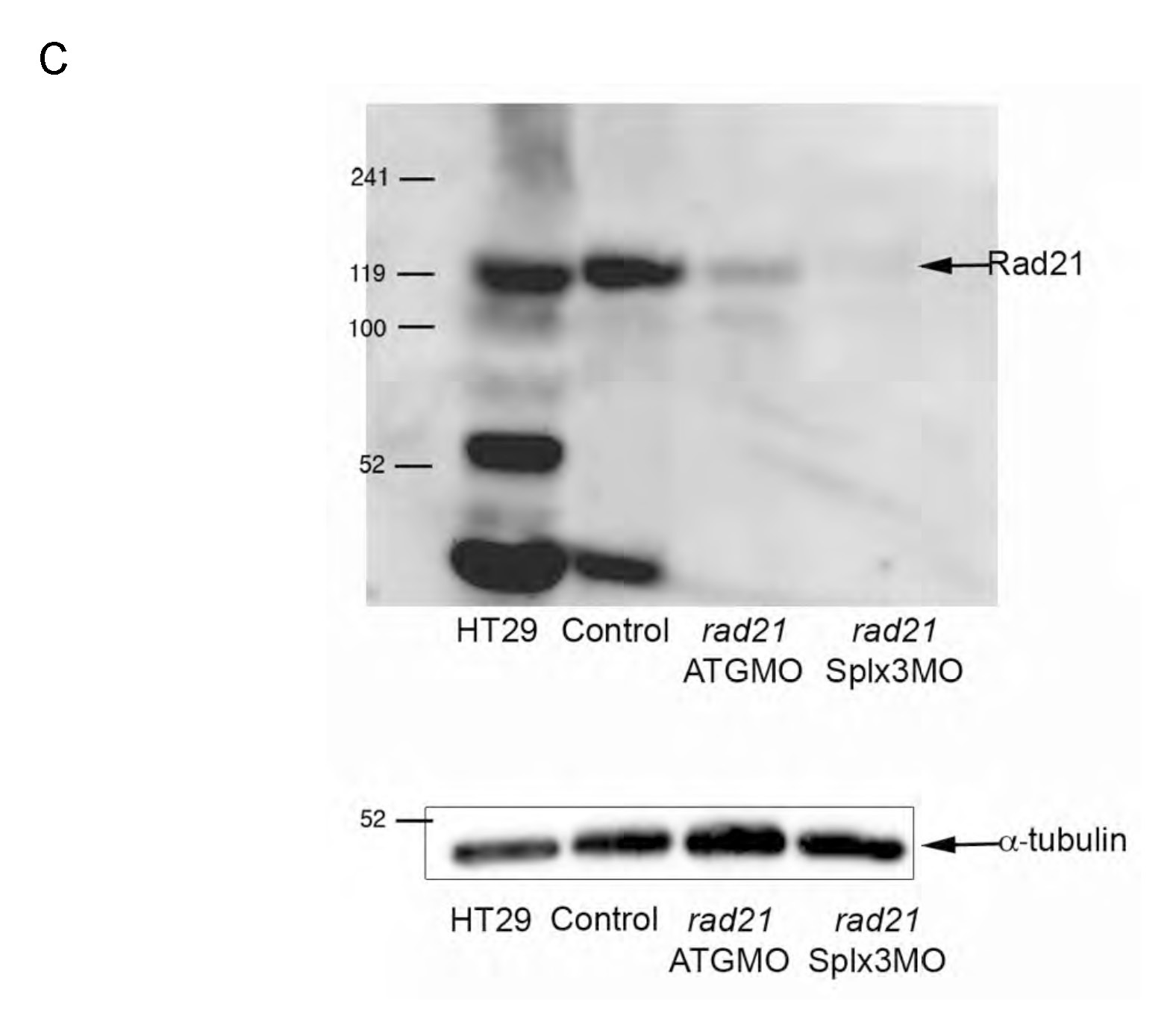
Fig. S2 cDNA and protein sequence of Rad21nz171 and previously characterized alleles. The rad21 transcript contains 2615 nt and encodes a protein of 643 residues (NCBI). There are two chromosome 16- associated rad21 sequences available at NCBI: NM_199595/BC045311 and AY423040/Ensembl Transcript ENSDART00000005927. An alignment of proteins encoded by these two transcripts revealed a polymorphism at position 592, where glycine is substituted for serine in NM_199595/BC045311. (A) Annotated sequence of the cDNA synthesized from rad21nz171 homozygotes. The sequence is 2428 bp and incorporates the entire open reading frame. We designed antisense morpholino oligonucleotides directed against the rad21 transcript: two targeting the start and 5′ regions, and one targeting the 5′ donor of exon 3. Morpholino binding sites are indicated by highlighting: UTR_MO, yellow; ATG_MO, green; SPLX3_MO, pink. The start codon is boxed and the stop codon is indicated in bold. The G->T mutation in the rad21nz171 allele is boxed and highlighted in red. (B) Comparison of the protein sequence of two Rad21 sequences deposited in GenBank, NM_199595 and AY423040, with the Rad21nz171 protein sequence (Rad21 17.1). The stop codon at position 277 created by the mutation is indicated by an asterisk. The 276 amino acid truncated protein resulting from this mutation is predicted to be non-functional, as the C-terminal winged helix domain important for contacting the Smc1 head (Nasmyth and Haering, 2005) is deleted. Rad21nz171 and AY423040 both have serine at position 592, whereas NM_199595 has glycine. The hydrophilic nature of this region is conserved. (C) Top, immunoblot showing lack of Rad21 protein in rad21 morphants, indicating correct targeting of rad21 mRNA by morpholinos. The Rad21 antibody (AB3233, Chemicon) is directed to the C-terminus of the protein and is not expected to recognize truncated products. HT29 is a human colon carcinoma cell line; 5 μg total protein was loaded as a positive control for the anti-Rad21 antibody. ‘Control’ refers to non-injected wild-type embryos. Embryos were harvested at 48 hpf, and 20 μg total protein was loaded per lane. Bottom, anti-α-tubulin immunoblot as a loading control.
Image
Figure Caption
Figure Data
Acknowledgments
This image is the copyrighted work of the attributed author or publisher, and
ZFIN has permission only to display this image to its users.
Additional permissions should be obtained from the applicable author or publisher of the image.
Full text @ Development