Fig. 4
- ID
- ZDB-FIG-221212-4
- Publication
- Lu et al., 2021 - Discovery of a subtype-selective, covalent inhibitor against palmitoylation pocket of TEAD3
- Other Figures
- All Figure Page
- Back to All Figure Page
|
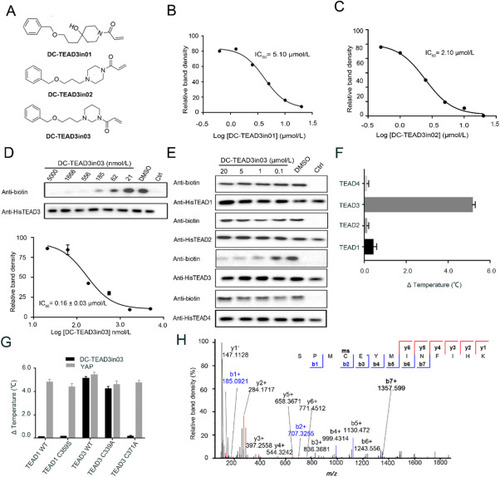
Figure 4. The biophysical and biochemical characterization of DC-TEAD3in03. (A) The chemical structures of DC-TEAD3in01, DC-TEAD3in02 and DC-TEAD3in03. (B)‒(D) The inhibitory activities of DC-TEAD3in01, DC-TEAD3in02 and DC-TEAD3in03 through CuAAC-based click chemistry. The band intensity was quantified in ImageJ (NIH). The experiments were performed in triplicate and the data was shown as mean ± SD, n = 3. (E) DC-TEAD3in03 selectively inhibited TEAD1 and TEAD3 palmitoylation while sparing TEAD2 and TEAD4. (F) and (G) DC-TEAD3in03 significantly improved the thermostability of TEAD3 but had minimal effect on TEAD1 or corresponding C359S mutant. Synthetic YAP peptide was used as the positive control with the ratio of 1:5. The experiments were performed in triplicate. (H) The modification of transfected-TEAD3 upon 50 μmol/L DC-TEAD3in03 treatment for 6 h was detected by MS experiments. |