|
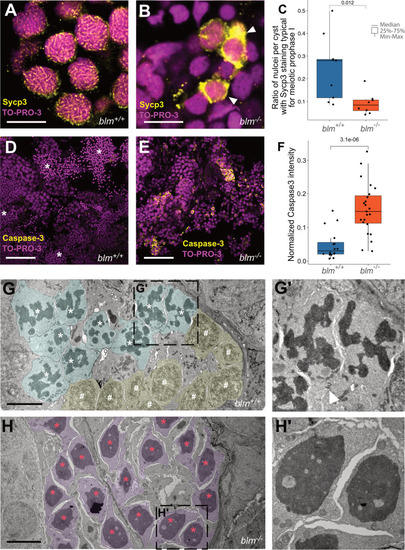
Blm loss-of-function results in meiotic defects during spermatogenesis in zebrafish males.A, B Representative Sycp3 immunostainings of wild-type and blm−/− spermatogonial cysts. In wild-type cysts (A) patterns typical for meiotic prophase I can be detected, whereas in mutant cysts (B) aberrant Sycp3 patterns (white arrowheads) can be observed (n = 8 different wild-type and blm−/− samples were tested each). TO-PRO-3 staining (red) denotes nuclei (scale bar: 8 μm). C Ratio of nuclei showing Sycp3 immunostaining patterns characteristic for pachytene. A comparison of wild-type and mutant testes suggests that blm−/− are defective in entering pachytene. D, E Cells undergoing programmed cell death as shown by Caspase-3 (green) staining in wild-type (D) and blm−/− testes (E). TO-PRO-3 staining (red) denotes nuclei (n = 6 different wild-type and blm−/− testis lobes were tested each). Asterisks denote spermatozoa clusters (Scale bar: 50 μm). F Normalized Caspase-3 stainings of wild-type and mutant testes suggest increased apoptosis in the absence of Blm. G, G′ Electron microscopic images of representative spermatogenic cysts with spermatocytes in meiotic prometaphase (cyan) and other early stages of meiosis I (yellow) from wild-type males. White asterisks denote condensed chromosomal structures, white hash symbols indicate nuclei in early meiotic prophase I (see Zhang et al., 2014). The white arrowhead in G′ indicates a cytoplasmic bridge between spermatocytes (scale bar: 5 μm). H, H′ Electron micrographs of spermatogenic cysts of blm−/− mutant males with spermatocytes (purple) showing aberrantly condensed chromatin (red asterisk), which was found to be the characteristic feature of mutant testes. Note that there are no nuclear envelopes observed around chromatin condensates, indicating that this phenomenon is related to cell division (see also Supplementary Fig. 5) (scale bar: 5 μm).
|