- Title
-
Regulation of Wnt/PCP signaling through p97/VCP-KBTBD7-mediated Vangl ubiquitination and endoplasmic reticulum-associated degradation
- Authors
- Feng, D., Wang, J., Yang, W., Li, J., Lin, X., Zha, F., Wang, X., Ma, L., Choi, N.T., Mii, Y., Takada, S., Huen, M.S.Y., Guo, Y., Zhang, L., Gao, B.
- Source
- Full text @ Sci Adv
|
( |
|
( |
|
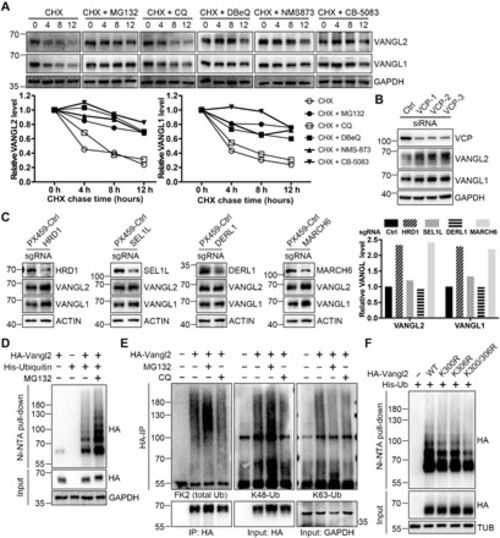
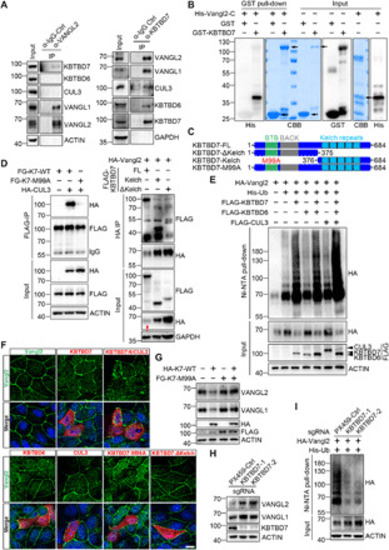
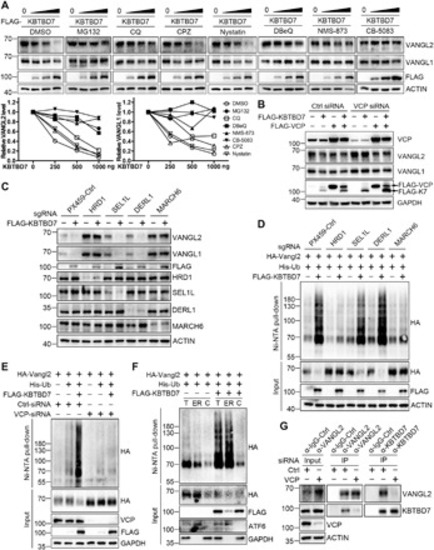
(A) Inhibition of proteasome or p97/VCP attenuated the KBTBD7-induced Vangl degradation. HEK293T cells were transfected with KBTBD7 in an increasing dose (0, 250, 500, and 1000 ng) and treated with dimethyl sulfoxide (DMSO), MG132 (10 μM), CQ (25 μM), clathrin inhibitor CPZ (30 μM), caveolin inhibitor Nystatin (25 μg/ml) or p97/VCP inhibitor DBeQ (10 μM), NMS-873 (2 μM), or CB-5083 (5 μM) for 6 hours (4 hours for DBeQ). The relative Vangl protein levels were quantified below. (B) VCP is required for KBTBD7-induced Vangl degradation. Upon siRNA knockdown of VCP in HEK293T cells, KBTBD7 (K7) failed to induce Vangl degradation, which was restored by reconstitution of VCP. (C) HRD1 and MARCH6 are required for KBTBD7-induced Vangl degradation. sgRNA-mediated silencing of HRD1 or MARCH6, but not SEL1L or DERL1, protected Vangl from KBTBD7-induced degradation. (D) HRD1 and MARCH6 are required for KBTBD7-induced Vangl2 ubiquitination. sgRNA-mediated silencing of HRD1 or MARCH6, but not SEL1L or DERL1, blocked KBTBD7-induced Vangl2 ubiquitination in HEK293T cells. (E) VCP is required for KBTBD7-induced Vangl2 ubiquitination. KBTBD7 was unable to induce Vangl2 ubiquitination upon VCP knockdown by siRNA. (F) KBTBD7-induced Vangl2 ubiquitination mainly occurred in the ER but not in the cytosol. Total lysates, ER, and cytosolic fractions of HEK293T cells were isolated and subjected to ubiquitination assay. Activating transcription factor 6 (ATF6) served as an ER marker. T, total cell lysate; ER, ER fraction; C, cytosolic fraction. (G) The endogenous Vangl2-KBTBD7 interaction depends on VCP. Upon siRNA knockdown of VCP in HEK293T cells, the binding between Vangl2 and KBTBD7 was significantly reduced. |
|
( |
|
( |
|
( |
|
( |
|
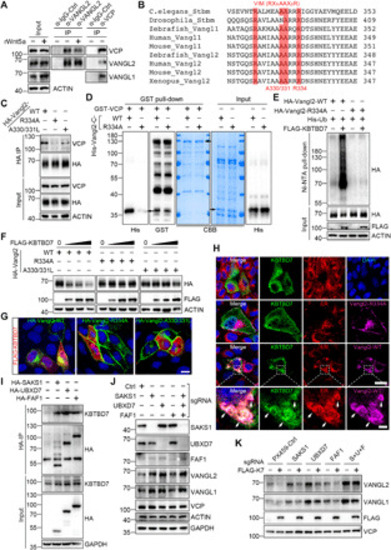
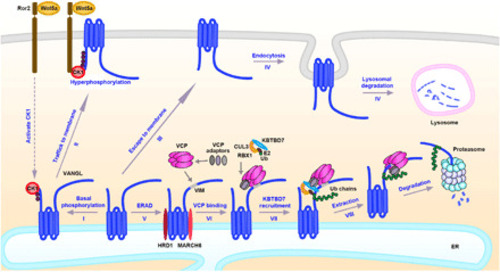
(I) Newly synthesized Vangl proteins are basally phosphorylated by CK1 in the ER; (II) the basal phosphorylated Vangl are transported to the cell surface where they undergo further phosphorylation and become stabilized; (III) some of unmodified Vangl escape ERAD and reach the cell surface; (IV) the unphosphorylated Vangl are not stable and are internalized and degraded via the lysosomal pathway; (V) most of the unmodified or unfolded Vangl proteins are degraded through the ERAD pathway. ERAD components HRD1 and MARCH6 are required for the degradation of Vangl and may initiate Vangl ubiquitination. (VI) VCP directly binds to Vangl at a highly conserved VIM; (VII) VCP recruits cytosolic E3 ligase KBTBD7 via its UBA-UBX adaptors (SAKS1, UBXD7, and FAF1), resulting in enhanced poly-ubiquitination of Vangl; and (VIII) extraction of highly ubiquitinated Vangl molecules for proteasomal degradation. |