- Title
-
Antagonistic role of vega1 and bozozok/dharma homeobox genes in organizer formation
- Authors
- Kawahara, A., Wilm, T., Solnica-Krezel, L., and Dawid, I.B.
- Source
- Full text @ Proc. Natl. Acad. Sci. USA
|
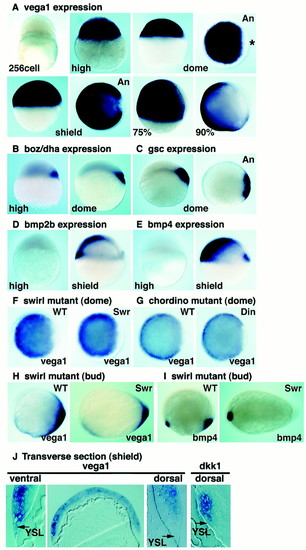
Vega1 is activated at the MBT and expressed in a complementary pattern to the dorsal genes, boz/dha and gsc. Whole-mount in situ hybridization of vega1 (A, F-H), boz/dha (B), gsc (C), bmp2b (D), and bmp4 (E and I). (A-G) Dorsal is to the right where known; An, animal view; 75% and 90%, 75% and 90% epiboly stage. Asterisk in dome stage animal view points to vega1-depleted region. (H-I) Anterior is to the left, dorsal up. (F and G) Expression of vega1 is not changed in swirltc300/tc300 and chordinott250/tt250 mutant embryos at dome stage. Genotyping of mutant embryos was performed by PCR. (H and I) At bud stage, vega1 is partly down-regulated in swirltc300/tc300 mutant embryos, whereas bmp4 expression in the posterior region is abolished. (J) Transverse section of shield stage embryo stained by in situ hybridization for vega1 or dkk1. It is noteworthy that the dkk1 expression domain is vega1 negative. Arrows indicate yolk syncytial layer. |
|
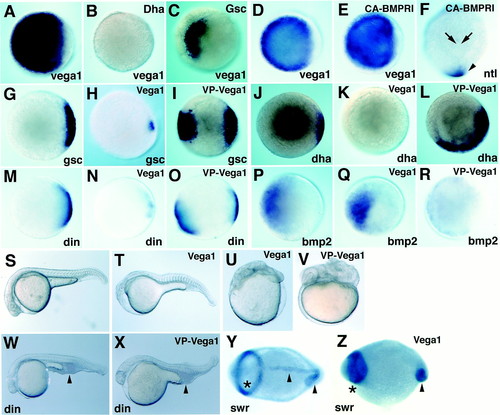
Regulation of vega1 expression and analysis of vega1 function. Whole-mount in situ hybridization for vega1 (A-E), ntl (F), gsc (G-I), boz/dha (J-L), chordin (M-O), and bmp2b (P-R); animal views (A-E, G-R), dorsal view (F), dome stage (A-E, G-I, M-O), 30% epiboly stage (P-R), shield stage (J--L), bud stage (F). (A-C) boz/dha mRNA (B; 25pg), gsc mRNA (C; 50pg) were injected into one to two cell stage embryos; (A) uninjected control. (D-F) RNA injections; CA-BMPRI mRNA (E and F; 200 pg) was injected into one to two cell stage embryos; (D) uninjected control. (E) Vega1 expression was unchanged. (F) Expression of ntl in the chordamesoderm region was suppressed (arrows), but not affected in the tailbud domain (arrowhead). (G-R) vega1 mRNA (H; 250 pg; and K, N, and Q, 100 pg) and VP16-vega1 mRNA (I, L, O, and R, 50 pg) were injected into one to two cell and two to eight cell stage embryos, respectively. (G, J, M, and P) Uninjected controls. (S-V) Phenotypic effects; lateral views, prim-6 (25 hpf) stage. Embryos were injected with vega1 mRNA (T and U; 100pg) or VP16-vega1 mRNA (V; 50pg) at the one to two cell stage. (S) Uninjected control. (W and X) Embryos from a dintt250/+X dintt250/+ cross were injected with VP16-vega1 mRNA (X; 100pg) at one to two cell stage. VP16-vega1 could not restore the ventral tail phenotype (arrowhead), whereas wild-type embryos injected with VP16-vega1 were dorsalized (V). (W) Uninjected dintt250/tt250 mutant. (Y and Z) Whole-mount in situ hybridization for pax2.1 plus ntl; 5-somite stage, dorsal view. Asterisk and arrowhead show pax2.1 and ntl staining domain, respectively. (Z) Embryos from a swirltc300/+X swirltc300/+ cross were injected with vega1 mRNA (150pg) at one to two cell stage. (Y) Uninjected swirltc300/tc300 mutant. |
|
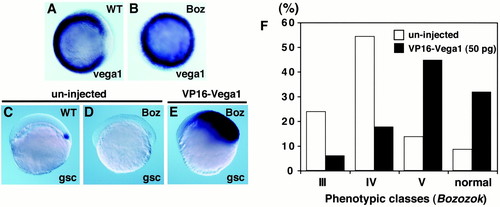
VP16-vega1 can rescue both gsc expression and boz phenotype. Whole-mount in situ hybridization for vega1 (A and B, shield stage, animal view) and gsc (C-E, 40% epiboly stage, lateral view). Genotyping of boz mutants was carried out by restriction fragment polymorphism (11). (A) bozm168/+ phenotypically wild type. (B) bozm168/m168 mutant. (C) Uninjected wild-type embryo. (D) Uninjected bozm168/m168 embryo. Expression of gsc was suppressed in all uninjected boz embryos (n = 15). (E) VP16-vega1 mRNA (100pg) was injected into one-cell stage of embryos obtained from a bozm168/m168 x bozm168/m168 cross. Expansion of gsc was observed in all VP16-vega1-injected boz embryos (100%, n = 39). (F) Suppression of boz phenotype by VP16-vega1 injection. VP16-vega1 mRNA (50 pg) was injected into one-cell stage of embryos obtained from bozm168/m168 x bozm168/m168 crosses. The boz phenotype was classified by morphological criteria at 30 hpf as described previously (11). The number of uninjected and VP16-vega1-injected embryos was 59 and 85, respectively. Class V has a small break in trunk notochord with normal head structure; there is a two-to-several somite wide gap in the trunk notochord of class IV; class III shows partial cyclopia with anterior head deficiency and usually a large gap in the trunk notochord. Essentially identical results were obtained in several independent experiments. |