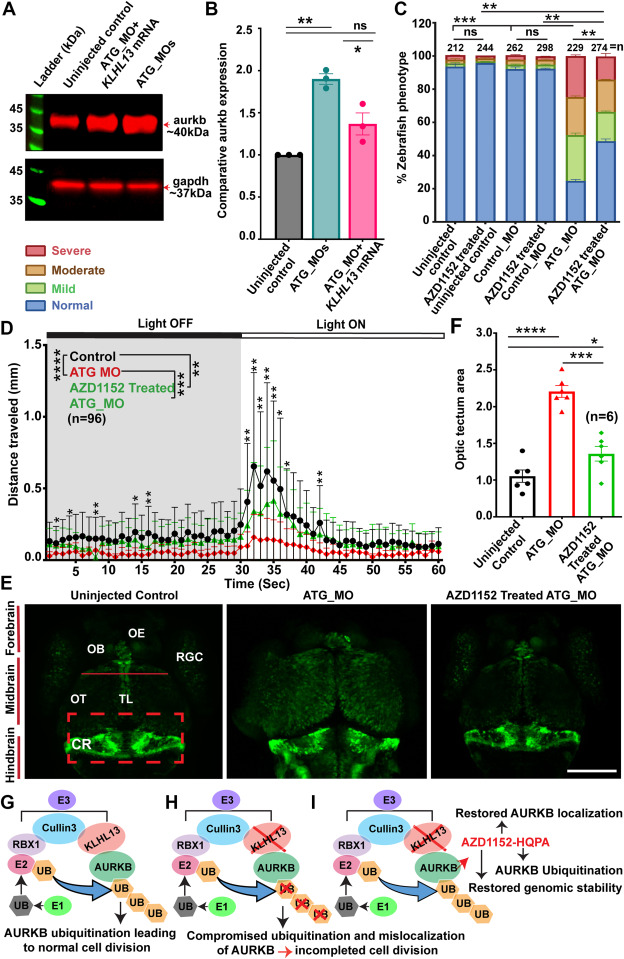
Fig. 5 AZD1152-HQPA treatment rescued developmental and behavioral deficits in klhl13 zebrafish morphants. A. Endogenous zebrafish aurkb expression level at 3 dpf in the uninjected control, KLHL13WT mRNA + ATG_MO coinjected, and in the ATG_MOs alone injected larvae. B. Quantification of the aurkb expression. The klhl13 morphants (ATG_MOs) revealed significantly high aurkb levels (∗∗P <.0048) compared with uninjected controls. Intriguingly, coinjections with the human KLHL13WT mRNA significantly (∗P >.037) reduced the aurkb levels in the klhl13 morphants. C. Treatment of klhl13 ATG_MO-injected zebrafish larvae within 24 hours after fertilization with AZD1152-HQPA inhibitor (30 nM) significantly (∗∗P <.001, ∗∗∗P < 002) rescued the number of larvae in the normal developmental class. D. Visual motor startle behavior analysis demonstrated a significantly impaired locomotor activity in ATG_MO injected larvae during the dark-to-light transition period in terms of distance (mm) traveled. Intriguingly, AZD1152-HQPA (30 nM) treatment significantly improved locomotor activity when compared with the untreated ATG_MO alone injected group (∗P <.01, ∗∗P <.001, ∗∗∗P <.0001, and ∗∗∗∗P <.00001). E. Brain morphology of neurod1-EGFP zebrafish at 5 dpf. ATG_MO-injected larvae after treatment with AZD1152-HQPA (30 nM) had a significant improvement in the overall normal brain size (comparable to controls), particularly the optic tectum, when compared with the untreated ATG_MO, quantified in panel (F). Moreover, the MHB was also intact in the AZD1152-HQPA treated ATG_MO-injected larvae (∗P <.0419, ∗∗∗P <.0001, ∗∗∗∗P <.00001). Scale bar: 200 μm. G. Schematic of known Kelch-dependent E3 ubiquitin ligase-mediated ubiquitination of the AURKB pathway. H. Based on the data presented in this study, we hypothesize that the impairment of KLHL13 function in the recognition and presentation of AURKB to the ubiquitin (UB) molecules for the ubiquitin-dependent proteasomal degradation caused abnormal cell-cycle regulation. I. AZD1152-HQPA mediated inhibition of AURKB can restore genomic stability and AURKB levels at the central spindle in cells expressing KLHL13 pathogenic variants. Thus, the AURKB inhibitor (AZD1152-HQPA) could be further developed and evaluated as a potential treatment option for brain disorders stemming from KLHL13 dysfunction.
Image
Figure Caption
Figure Data
Acknowledgments
This image is the copyrighted work of the attributed author or publisher, and
ZFIN has permission only to display this image to its users.
Additional permissions should be obtained from the applicable author or publisher of the image.
Full text @ Genet. Med.