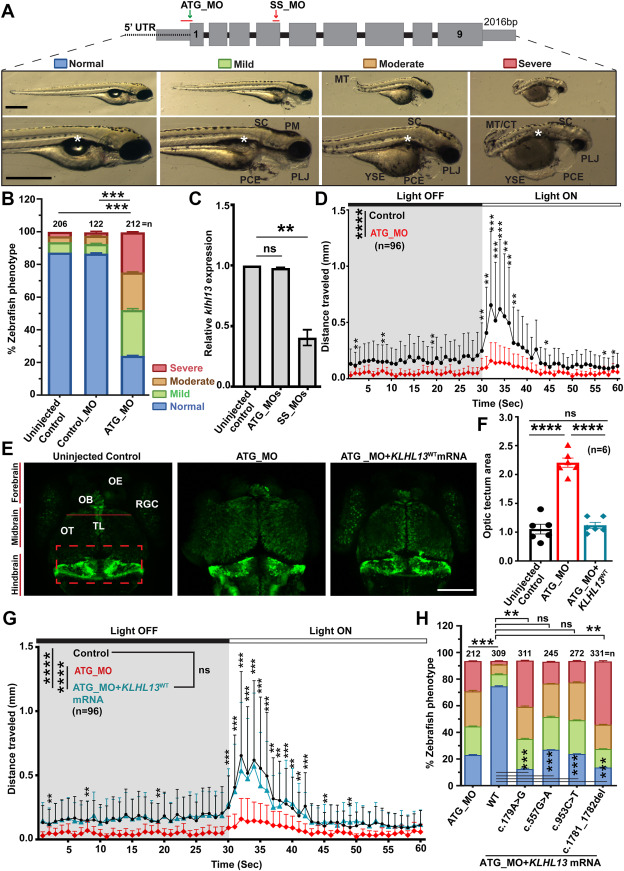
Fig. 4 Morpholino-mediated klhl13 knockdown caused developmental delay and macrocephaly in zebrafish. A. Schematic representation of the zebrafish klhl13 gene (top), along with the location of ATG-blocking morpholino (MO). Shown also are representative images (bottom) of resulting phenotypes in ATG_MO injected zebrafish larvae at 3 dpf. Based on the phenotypic severity, larvae were categorized into four classes MT/CT, malformed curled tail; PCE, pericardial edema; PLJ, protruding lower jaw; PM, populated head melanocytes; SC, scoliosis; YSE, yolk sac edema. White star marked absence of the swim bladder. Scale bar: 10 μm. B. The graph represents the percentages of controls (uninjected and control MO_injected larvae) along with klhl13 ATG_MO. A significantly reduced (∗∗∗P < .0001) number of ATG_MO-injected morphants was observed in the normal morphological class of larvae when compared with control groups. C. Graph representing the relative klhl13 expression in the ATG_MO and SS_MOs compared with the uninjected control, normalized to gapdh. SS_MOs showed a significant reduction in the relative klhl13 expression (∗∗P <.0044), whereas ATG_MO revealed a comparable klhl13 expression (ns: P <.0955) to uninjected control. D. Visual motor startle behavior analysis demonstrated delayed motor reflexes during the dark-to-light cycle transition stage in ATG_MO injected morphants compared with control-injected larvae (∗P <.01, ∗∗P <.001, ∗∗∗P <.0001, ∗∗∗∗P <.00001). E. Representative confocal images of 5 dpf neurod1-EGFP transgenic zebrafish larvae. Compared with the control groups (uninjected and control_MO-injected), larvae injected with ATG_MO exhibited a significantly enlarged brain (∗∗∗P <.00001; quantified in panel F), particularly in the optic tectum (highlighted by red lines). Additionally, the MHB appeared disrupted in ATG_MO-injected larvae (indicated by red boxes). Anatomical regions are labeled: CR, cerebellum; OB, olfactory bulb; OE, olfactory epithelium; OT, optic tectum; RGC, retinal ganglion cells; TL, torus longitudinalis. Scale bar: 200 μm. F. Coinjection of human KLHL13WT mRNA with ATG_MO significantly rescued the brain phenotype, restoring brain size and the integrity of the MHB (∗∗∗∗P <.0001 compared with ATG_MO alone). G. Visual motor startle behavior analysis showed a significant improvement in the overall locomotor activity in the human KLHL13WT mRNA and ATG_MO coinjected larvae in the light/dark transition period compared with the ATG_MO injected larvae (∗∗P <.001, ∗∗∗P <.0001, ∗∗∗∗P <.00001). H. The graph represents the number of ATG_MO injected morphants, as well as human KLHL13WT and NDD-associated variants mRNA coinjected morphants in various morphological categories. Microinjections of human KLHL13WT encoding mRNA, along with ATG_MOs, significantly rescued the number of larvae in the normal developmental class compared with ATG_MO alone (∗∗∗P <.0002). However, NDD-associated KLHL13 hemizygous variants (c.179A>G, c.557G>A, c.953C>T, and c.1781_1782del) encoding mRNAs did not rescue the phenotype when compared with the normal class of KLHL13WT encoding mRNA (∗∗∗P <.0001), supporting their pathogenic nature. Moreover, significantly more larvae in the severe class were found when ATG_MO was coinjected with human KLHL13 mRNAs harboring c.179A>G (∗∗P <.0012) and c.1781_1782del variants (∗∗P <.0052).
Image
Figure Caption
Figure Data
Acknowledgments
This image is the copyrighted work of the attributed author or publisher, and
ZFIN has permission only to display this image to its users.
Additional permissions should be obtained from the applicable author or publisher of the image.
Full text @ Genet. Med.