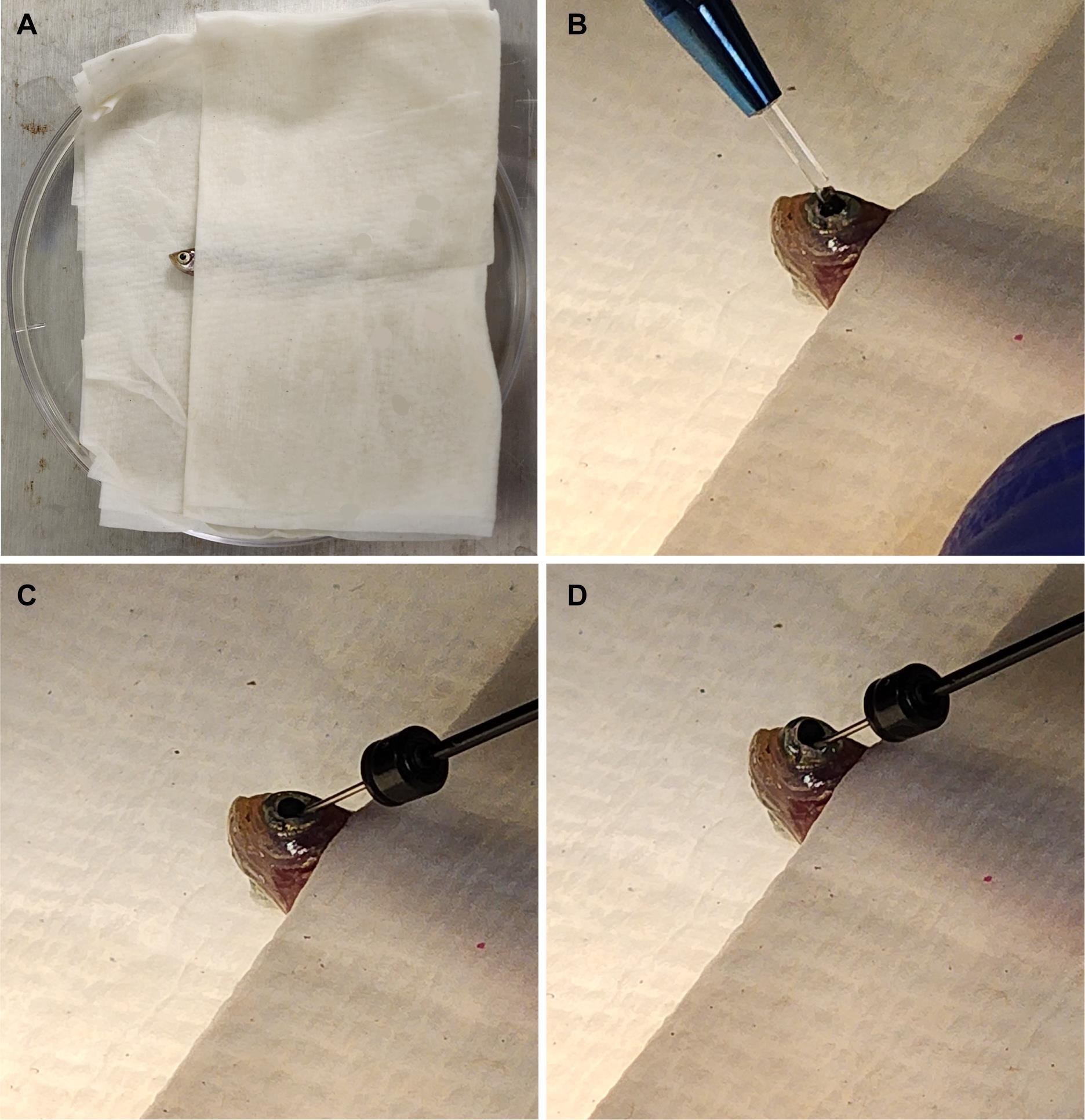
Fig. 1 Fish setup for intravitreal injections. (A) Fish are placed in a moist towel so that only their heads are exposed for intravitreal injections after being anesthetized. (B) A sapphire blade is used to make an incision between the pupil and the outer edge of the iris. (C) A blunt syringe is used to remove 1.0 μL of vitreous humor from the eye. (D) 1.0 μL of the solution of interest is injected at the incision site. This image was adapted from Shihabeddin et al. [17]. 3. Place the Petri dish lid with zebrafish under a microscope, viewing at approximately 10×. 4. Use the sapphire blade to make an incision between the pupil and the outer edge of the iris (Figure 1B). 5. Insert the syringe at the site of incision and remove 1.0 μL of vitreous humor. It is best to angle the needle toward the lens to avoid damaging the retina (Figure 1C). Important: Removal of the vitreous humor is critical for the oligonucleotides to reach and penetrate the retina. 6. Remove the syringe from the incision site and expel the vitreous humor collected. 7. Use a syringe to collect 1.0 μL of either Vivo-morpholino or siRNA solution. 8. Re-insert the syringe at the site of incision and inject solution into the eye (Figure 1D), being careful to avoid touching the retina. 9. Remove the syringe and place the zebrafish in the recovery tank. 10. Use a disposable pipette to aerate the tank until the zebrafish awakens.
Image
Figure Caption
Acknowledgments
This image is the copyrighted work of the attributed author or publisher, and
ZFIN has permission only to display this image to its users.
Additional permissions should be obtained from the applicable author or publisher of the image.
Full text @ Bio Protoc