- Title
-
Early development of the zebrafish pronephros and analysis of mutations affecting pronephric function
- Authors
- Drummond, I.A., Majumdar, A., Hentschel, H., Elger, M., Solnica-Krezel, L., Schier, A.F., Neuhauss, S.C., Stemple, D.L., Zwartkruis, F., Rangini, Z., Driever, W., and Fishman, M.C.
- Source
- Full text @ Development
|
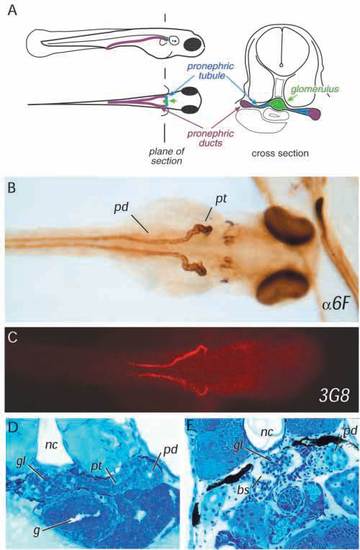
The zebrafish pronephros. (A) Diagram of the three elements of the larval pronephros at 72 hpf showing the position of glomerulus under the notochord, the pronephric tubules extending laterally to connect with the pronephric ducts which serve the function of the collecting system. (B) Horseradish peroxidase whole-mount immunostaining of a 84 h embryo with anti-Na+/K+-ATPase alpha subunit monoclonal antibody (α6F;) shows the overall anatomy of the pronephric ducts (pd) and pronephric tubules (pt), which become progressively convoluted. The glomerulus is unstained at the embryo midline. (C) The monoclonal antibody 3G8 stains the anterior half of the pronephric ducts as well as the lateral part of the pronephric tubules. (D) Cross section of a 72 h embryo showing that the glomerulus (gl), the pronephric tubule (pt) and the pronephric duct (pd) are fully formed between the gut (g) and the notochord (nc). (E) In a 6.5-day-old larva, Bowman’s space (bs) is clearly evident in the glomerulus (gl). EXPRESSION / LABELING:
|
|
Pronephric nephron development. (A) Cross section of wild-type pronephroi at 24 hpf shows the forming nephron primordium (np) as an invagination of the coelomic lining. The lumen of the nephron precursor, or nephrocoel, is still connected to the coelom (c) at this stage by a transient nephrostome (nst; arrow). (B) Sagittal section at 24 h shows the anterior pronephric duct (pd) abutting on the lateral aspect of the nephron primordium (np). C) At 32 h, the nephron primordium is closed off from the coelom and appears as a double-layered sac of cells with a central lumen. (D) A sagittal view at 32 h reveals early stages of nephron morphogenesis with lateralmost cells in the position of future tubules (tubule primordium; tp) becoming distinct from medial cells in the position of the future glomerulus (glomerular primordium; gp). (E) By 40 h, the nephron primordium has become partitioned into a glomerular domain medially directly under the dorsal aorta (da) and tubule domains laterally with a distinct basement membrane surrounding the developing glomerulus (arrow). Pigment cells envelope the pronephros at this stage. (F) A sagittal view of the 40 h pronephric nephron shows the medial paired glomeruli (gl) with the pronephric tubules (pt) fused to their posterior side. da, dorsal aorta. (G) Longitudinal sections through the forming glomerulus (gl) at 40 h show its position below the notochord (nc) and its intimate association with the dorsal aorta (da) and cells (arrow), which surround the forming glomerular basement membrane. (H) Sagittal view of the pronephros at 50 h with the glomeruli (gl) fused at the midline and the pronephric tubule (pt) arcing over to connect to the pronephric duct (pd). (I) The pronephros at 2.5 days postfertilization shows paired glomeruli (gl) ventral to the aorta and pronephric tubules (pt) connecting laterally to the pronephric ducts. (J) By 3 days, nephron formation is essentially complete and the direct connection between Bowman’s space and the lumen of the pronephric tubules is evident (arrows). On the right are schematized versions of the micrographs in B, D, F and H, derived from serial sections, illustrating the morphological transitions that occur during pronephric nephron morphogenesis. In all cross sections, the top of the figure is dorsal and in all sagittal sections the top of the figure is anterior. |
|
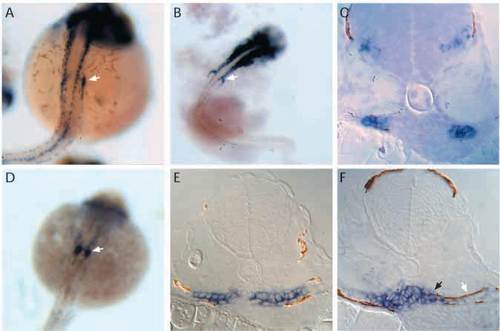
Expression of pax2.1 and wt1 in the forming pronephros. (A) Whole-mount in situ hybridization shows pax2.1 is expressed in the anterior pronephric ducts (arrow) at 24 h as well as in the spinal cord. (B) At 30-32 h, a new domain of pax2.1 expression is observed anterior and medial to the pronephric ducts (arrow), in the position of the future pronephric tubules. (C) Histological sections of pax2.1 expression in the presumptive tubule primordium cells show that these cells constitute the lateral half of the nephron primordium and that the medial halves of the nephron primordium are negative. (D) Whole-mount in situ hybridization shows wt1 expression in the paired nephron primordia at 24h (arrow). (E) At 30-32 h, wt1 is uniformly expressed throughout the nephron primordia. (F) By 36 h, wt1 expression becomes restricted to a mass of cells at the midline in the position of the future glomerulus (black arrow). Lateral cells within the forming nephron in the position of the future tubule do not express wt1 (white arrow). |
|
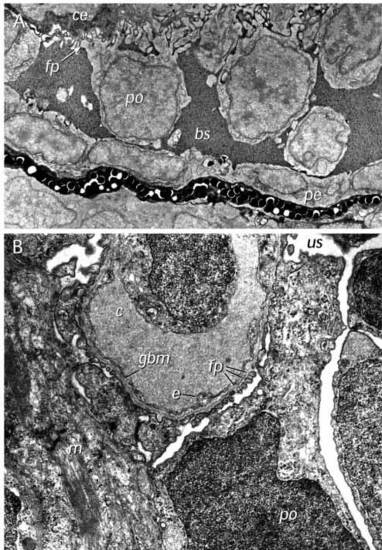
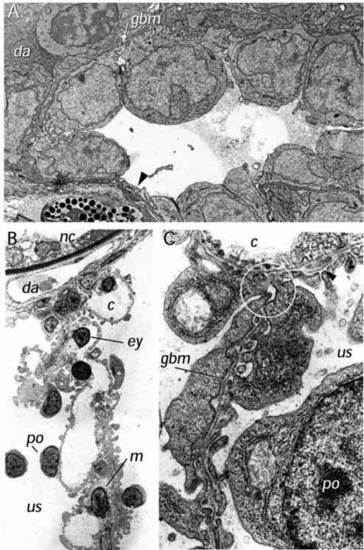
Electron microscopy of the wild-type glomerulus. (A) Ultrastructure of the forming glomerulus at 40 hpf. A longitudinal section (similar to that presented in Fig. 2D) shows podocytes (po) extending foot processes (fp) in a dorsal direction and in close contact with capillary endothelial cells (ce). Bowman’s space (bs) appears filled with an electron-dense precipitate and the ventralmost cells appear to be taking on the flattened appearance of the parietal epithelium (pe) that will line Bowman’s capsule. (B) Glomerulus and filtration barrier of the wild-type pronephros at 3.5 days postfertilization. The filtration barrier is well developed displaying an endothelium with open pores (without diaphragm), a thin trilaminar glomerular basement membrane (gbm) and podocytes with primary processes and numerous interdigitating foot processes (fp). The filtration slits between the foot processes are bridged by one or sometimes two slit membranes. Mesangial cells (m) are also evident. us, urinary space; c, capillary space; e, endothelial cell. ×36,500. |
|
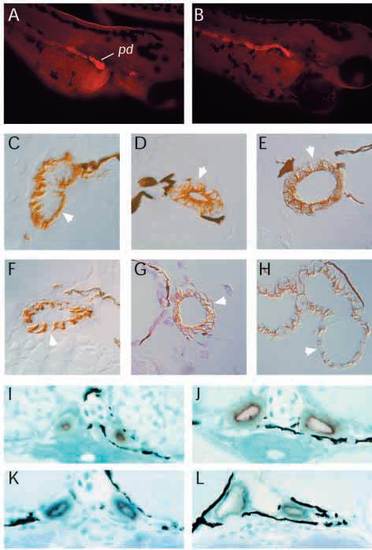
Onset of glomerular filtration. (A) Wild-type embryos injected with rhodamine dextran (10,000 Mr) into the heart at 48 hpf show extensive accumulation of dye in interstitium and in basement membranes but not in the lumen of the gut (g). Dye is present in the lumen of the pronephric duct indicating that the pronephric filtration is functioning at this time. By 56 hpf (B), capillary invaginations in the glomerulus appear more extensive and dye can found in vesicles in the apical cytoplasm of the pronephric ducts. da, dorsal aorta; gl, glomerulus; g, gut; pd, pronephric duct. |
|
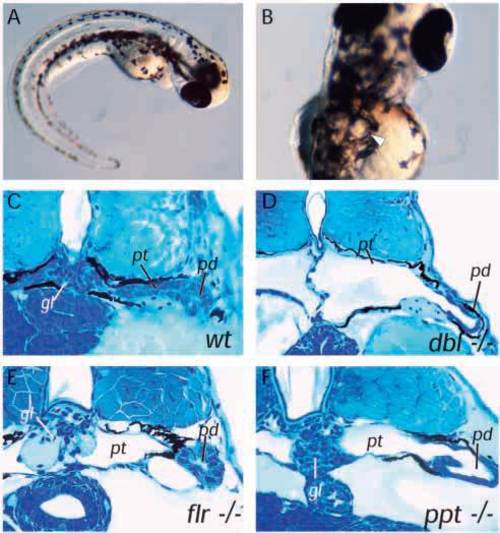
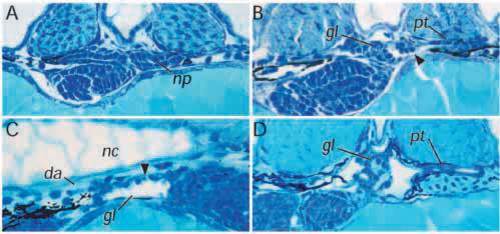
Cystic maldevelopment in zebrafish pronephric mutants. (A) The mutant double bubble (dbbm153) emerges from its chorion with a ventrally curved body axis and within 3-4 hours bilateral pronephric cysts (arrow in B) are evident. (C) Cross section of wildtype pronephros at 3.5 days pf. (D) Section of dbbm468 showing a grossly distended pronephric cyst in place of the pronephric tubule and a glomerulus reduced to a flattened septum at the midline. (E) The mutant fleer (flr) shows less severe cyst distension despite a flattened pronephric tubule epithelium and also a marked distension of the glomerular capillaries. (F) pao pao tang (pap) has an apparently intact glomerulus while the pronephric tubule epithelium is flattened and the pronephric duct is also distended. gl, glomerulus; pt, pronephric tubule; pd, pronephric duct. |
|
Early glomerular defects in the dbb-/- pronephros. dbbm468-/- embryos were examined at different stages of nephron development to determine the site of the primary defect. (A) Cross sections of dbbm468 pronephroi at 33 hpf reveal that the nephron primordium (np) is a double-layered sac of cells with a central lumen similar to wild-type pronephroi at this stage. (B) In 40 hpf mutant pronephroi, the distinct basement membrane separating glomerular and tubule domains is not seen. Instead, the forming glomerulus appears distended with fluid and cell-free areas (arrow) are evident at the junction of the glomerulus and tubule. (C) Longitudinal sections through the mutant glomerulus shows a distension of the glomerular lumen, flattening of glomerular cells and a thinning of cells associated with the glomerular basement membrane (arrow). (D) At 2.5 dpf, the dbbm468 pronephros displays a loose and disorganized glomerulus with progressive distension of the pronephric tubule. gl, glomerulus; pt, pronephric tubule; pd, pronephric duct; nc, notochord; da, dorsal aorta. |
|
Ultrastructure of the dbb-/- glomerulus. (A) Ultrastructure of the dbb-/- glomerulus at 40 hpf. Podocytes (po) are present but appear to show less extensive podocyte foot process development at this stage compared to wild type. Areas of cellular thinning (arrow) are also evident. (B) Cross section of the glomerular septum in the mutant double bubble (3.5 dpf). The glomerular capillaries (c), mesangium (m) and podocytes (po), on either side form a medial septum between the two cysts. The urinary spaces (us) on both sides of the septum represent the lumens of the cysts. ×3,500 (C) Section of part of the dbbm468-/- glomerular septum extending between two capillaries (c), (only one capillary is shown at the top). In several places, the glomerular septum consists of two podocyte layers (po) located adjacent to each other without a capillary or a mesangial layer in between. The two epithelia are focally interconnected by cell-cell junctions and form a common glomerular basement membrane (gbm), which appears not to be continuous with the GBM of the adjacent glomerular capillaries (encircled). In some places, the GBM appears to split and rejoin (arrowhead). ×35 000. da, dorsal aorta; gbm, glomerular basement membrane; nc, notochord; e, capillary endothelial cell; ey, erythrocyte; c, capillary lumen; us, urinary space. PHENOTYPE:
|
|
The pronephric duct is not obstructed in dbb-/- embryos. The lumen of the pronephric duct and cloaca is visible and patent in both wild-type (A) and dbb mutant (B) embryos indicating a lack of obstruction to the flow of fluid from the pronephros. (C) Serial sections show a patent pronephric duct lumen (arrow) in dbb homozygotes. |
|
Altered membrane protein targeting in pronephric mutants. (A) The monoclonal antibody alpha6F raised against the chicken alpha-1 subunit of the Na+/K+ ATPase reacts strongly with the pronephric duct (pd) at hatching (2.5 days) in wild-type (A) and mutant (dbbm468) (B) whole-mount embryos. In sections of wild-type duct (C), alpha6F staining is exclusively basolateral (arrow) while, in dbb (D), alpha6F staining is strongly apical and diminished on basolateral membranes (arrow). Diminished alpha6F basolateral staining is also observed in (E) fleer (flr), (F) dizzy (dzz), (G) inflated (ifl) and (H) big league chew (chw) mutant duct cells. (A,B) Cy3- labeled secondary antibody; (C-H) HRP-labeled secondary antibody. (I-L) Endogenous alkaline phosphatase staining is apical in wild-type pronephric duct (I) and is not affected in dbb (J), inflated (ifl) (K), or dizzy (dzz) (L), mutant duct cells. EXPRESSION / LABELING:
|
|
Eye defects in fleer-/- embryos. (A) Section of a wild-type eye at 72 hpf showing the characteristic stratification of retinal cell layers. (B) fleer mutant embryos at 72 hpf show excessive cell death in the photoreceptor cell layer (pcl; arrows). PHENOTYPE:
|

Unillustrated author statements PHENOTYPE:
|