- Title
-
Rag1 immunodeficiency-induced early aging and senescence in zebrafish are dependent on chronic inflammation and oxidative stress
- Authors
- Novoa, B., Pereiro, P., López-Muñoz, A., Varela, M., Forn-Cuní, G., Anchelin, M., Dios, S., Romero, A., Martinez-López, A., Medina-Gali, R.M., Collado, M., Coll, J., Estepa, A., Cayuela, M.L., Mulero, V., Figueras, A.
- Source
- Full text @ Aging Cell
|
|
|
|
|
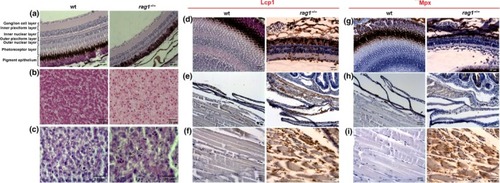
Senescence detection in |
|
Gene expression of typical SASP components in |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |