- Title
-
Distinct amino acid motifs carrying multiple positive charges regulate membrane targeting of dysferlin and MG53
- Authors
- Zhou, L., Middel, V., Reischl, M., Strähle, U., Nienhaus, G.U.
- Source
- Full text @ PLoS One
|
(A) Domain structure of Dysf. The boxed fragment is smDysf, which was used to test accumulation at the lesion patch. (B) Representative images showing accumulation of smDysf and different mutants. Basic AAs are highlighted in red; arrows indicate the site of lesion. Z-line (z) and sarcolemmal (sc) regions are noted. (C-D) Corresponding kinetics of accumulation of zebrafish (C) or human (D) smDysf at the damage site, again with the basic AAs highlighted in red. The fluorescence intensity at the lesion was normalized to the one of the undamaged state. Intensity courses are averages over 9–15 damaged cells. Scale bars, 4 μm. |
|
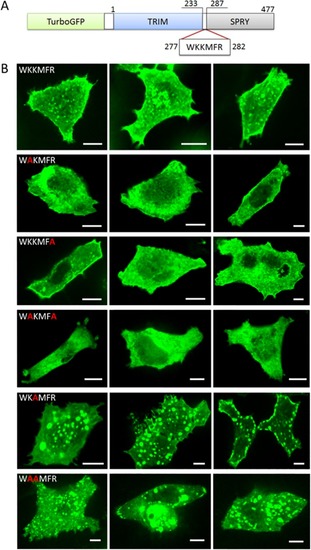
(A) Domain structure of the fusion protein turboGFP:MG53. (B) Cellular localization of (top to bottom) turboGFP:MG53 and mutants K278A, R282A, K278A/R282A, K279A, K278A/K279A in HeLa cells. Three representative images are shown for each variant, all imaged with excitation at 488 nm. Scale bars, 10 μm. |
|
Fluorescence was excited with a 488-nm laser. As in HeLa cells, K279A shows substantial vesicular localization in myoblasts. Scale bars, 10 μm. |
|
Two-channel imaging of turboGFP:MG53 (green fluorescence excited with 488-nm light, first column) with (second column) early endosomes, excited with 561-nm light (A), late endosomes, excited with 561-nm light (B), lysosomes, excited with 405-nm light (C), and inclusion bodies, excited with 561-nm light (D). Overlay images are shown in the third column. Scale bars, 10 μm. |
|
Representative images of (A) HeLa cells and(B) C2C12 myoblasts show that the charge-maintaining modification K279R rescues membrane targeting of MG53. Fluorescence was excited with a 488-nm laser. Scale bars, 10 μm. |