- Title
-
Metalloproteinase Adamts16 Is Required for Proper Closure of the Optic Fissure
- Authors
- Cao, M., Ouyang, J., Guo, J., Lin, S., Chen, S.
- Source
- Full text @ Invest. Ophthalmol. Vis. Sci.
|
Adamts16 is expressed at the closing OF. (A–D) Adamts16 mRNA in situ hybridization of mouse embryonic sagittal eye sections. (A) E10.5 OC. (B) E11.5 central OC. (C) E11.5 OC more proximal, close to the OS. (D) E13.5 OC. Arrows point to the specific Adamts16 signals in the OF region. (E–H) Lateral views of adamts16 mRNA whole-mount in situ hybridization of (E) 24-hpf, (F) 36-hpf, (G) 48-hpf, and (H) 72-hpf zebrafish embryos. (E', F') Section in situ hybridization images of the corresponding (E–H) stages. The red arrow in (F') points to the adamts16 signal at the edge of the closing OF. Scale bar in (C): 50 μm, applies to (A–C); in (D): 50 μm; in (H): 100 μm, applies to (E–H); and in (H'): 50 μm, applies to (E'–H').
EXPRESSION / LABELING:
|
|
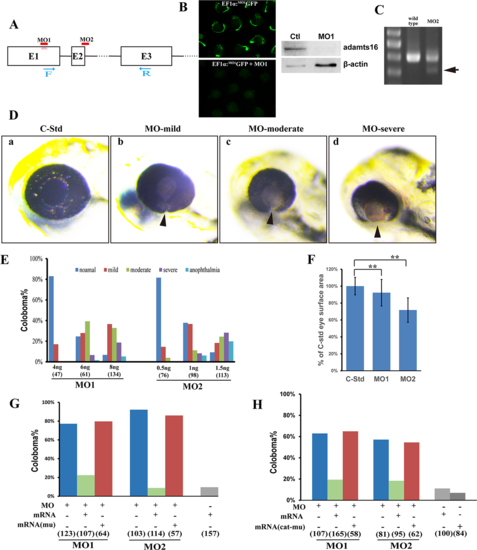
adamts16 is required for zebrafish OF closure. Diagram illustrating a portion of the zebrafish adamts16 gene, MO1 and MO2 targeting sites, and primers used to examine splicing products before and after MO2 injection. (B) Left: MO1 knockdown efficacy reporter assay. Right: Western blot analysis showing adamts16 protein depletion from MO1-morphant fish bodies. (C) Testing MO2 knockdown efficacy by RT-PCR using primers indicated in (A); amplicons from control fish RNA were 401 bp, while MO2 disruption of splicing resulted in the appearance of smaller 319-bp amplicons (arrow). (D) Knockdown of adamts16 by either MO1 or MO2 resulted in coloboma and microphthalmia. (F) Eye surface area of MO1- and MO2-morphant zebrafish. Data were normalized as the percentage of average eye surface area to the standard control MO-injected zebrafish. (G) Quantification of coloboma prevalence shows that coloboma phenotype of MO1- or MO2-morphants can be rescued by adamts16 mRNA injection. (H) Quantification of coloboma prevalence shows that the coloboma phenotype in MO1- or MO2-morphants could not be rescued by the catalytic site–mutant form of adamts16 mRNA injection. Numbers under each column in (E), (G), and (H) denote the number of zebrafish injected in each group.
PHENOTYPE:
|
|
adamts16-morphant OCs fail to disassemble the basement membrane at the closing OF. The 60-hpf zebrafish embryonic eye sagittal sections were stained with anti-laminin (A–C) or anti-fibronectin (D–F) antibodies. (A, D) Control eyes. Arrows point to the location of the previous OF. (B, E) adamts16-morphant eyes with mild coloboma. Arrows point to the unfused OF with intact basement membrane. (C, F) adamts16-morphant eyes with moderate coloboma. Arrows point to the unclosed OF. Scale bar: 50 μm.
PHENOTYPE:
|
|
adamts16-morphant OCs proliferate slowly, and exhibit increased apoptosis activity. (A, B) Images of 36-hpf zebrafish eye sagittal sections stained with anti-BrdU antibody (red). (C) Percentage quantification of BrdU+ cells/eye in control and MO1- and MO2-morphant groups. (D, E) TUNEL staining (green) of 36-hpf zebrafish eye sagittal sections. (F) Quantification of TUNEL+ cells/section in control, MO1-, and MO2-morphant groups. (G) Quantification of coloboma prevalence shows that the coloboma phenotype in MO1- or MO2-morphants could not be rescued by p53 MO knockdown. Numbers under each column denote the number of zebrafish injected in each group. Scale bars in (B) and (E): 50 μm.
PHENOTYPE:
|
|
Gene expression changes in adamts16-morphant eyes. (A–L) Expression of various genes essential for key cellular events of OF closure examined by in situ hybridization. tbx5: dorsal retina marker; vax2: ventral retina marker; aldh1a2: RA-synthesizing enzyme and dorsal retina marker; aldh1a3: RA-synthesizing enzyme and ventral retina marker; foxC1: POM marker and regulator; pax2: OF marker and regulator. (M) Heatmap of differentially expressed genes in 36-hpf control, MO1-, and MO2-morphant eyes analyzed by high-throughput sequencing. Left: Downregulated genes. Right: Upregulated genes. Green circles highlight cell cycle regulators and purple circles highlight cell death regulators. (N–O) In situ hybridization of fgf8 in control and MO1-morphant heads. Red arrowheads point to the midbrain-hindbrain boundary. Red arrows point to the OS. (P) Quantification of coloboma prevalence shows that coloboma phenotype of MO1- or MO2-morphants can be rescued by fgf8-MO coinjection. Scale bar: 100 μm.
EXPRESSION / LABELING:
PHENOTYPE:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. EXPRESSION / LABELING:
|

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. EXPRESSION / LABELING:
|