- Title
-
Zebrafish akt2 is essential for survival, growth, bone development, and glucose homeostasis
- Authors
- Zhang, D., Wang, J., Zhou, C., Xiao, W.
- Source
- Full text @ Mech. Dev.
|
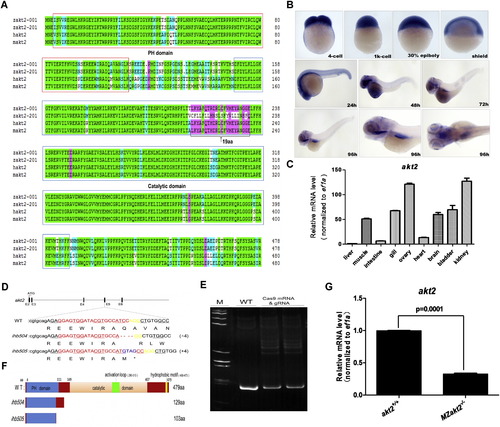
Expression of akt2 and disruption of akt2 via CRISPR/Cas9 technology. (A) Alignment of amino acid sequences of human akt2 (ENSG00000105221), mouse akt2 (ENSMUSG00000004056), and zebrafish akt2 (ENSDARG00000011219). Zakt2-001 and zakt2-201 represent the amino acid sequences of two transcripts of akt2 gene in zebrafish. The homologous degrees of amino acid sequence from high to low are marked by green, pink, light blue, and white. (B) Whole-mount in situ hybridization staining for zebrafish akt2 expression at different developmental stages during embryogenesis. (C) The expression level of akt2 in nine tissues of adult zebrafish as revealed by semi-quantitative RT-PCR assays. (D) The location of sgRNA targeting sites on zebrafish akt2, and the sequences of targeting sites from the mutant akt2ihb504 and the mutant akt2ihb505. Dashes, deletion; purple letters, insertion. (E) Verification of the efficiency of CRISPR/Cas9-mediated zebrafish akt2 disruption by HMA. Genomic DNA was prepared from the pools of injected embryos. The injected (Cas9 mRNA & gRNA) pools contain heteroduplex band, indicating that the site has been disrupted. (F) Schematic representation of the putative peptides of two wild-type transcripts and the two mutated akt2 peptides from the targeted allele. (G) The relative levels of the wild-type and homozygous mutant (akt2ihb504/ihb504) transcripts from larvae at 5 dpf were detected by RT-PCR. MZakt2−/− indicated maternal-zygotic mutants obtained from the offspring of homozygous intercrosses. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. PHENOTYPE:
|
|
Akt2 mutants exhibit growth retardation. (A) The morphology of wild-type and maternal-zygotic homozygous (MZakt2−/−) larvae from 24hpf to 72hpf. (B) At 5 dpf (days post-fertilization), heterozygous (Makt2+/−) larvae were indistinguishable from their homozygous (MZakt2−/−) siblings in morphology obtained from male akt2ihb504/+ × female akt2ihb504/ihb504 (left panel). Genotyping was conducted using HMA (Heteroduplex mobility assay) (right panel). (C) At 16 dpf, homozygous MZakt2−/− larvae were smaller than their heterozygous (Makt2+/−) siblings in body size obtained from male akt2ihb504/+ × female akt2ihb504/ihb504 (upper panel). Genotyping was conducted by HMA (lower panel). (D) At 51 dpf, homozygous MZakt2−/− zebrafish (akt2ihb504/ihb504) were much smaller than their wildtype (akt2+/+) siblings. (E) At 46 dpf, the body length of homozygous (MZakt2−/−) mutants (akt2ihb504/ihb504) was significantly shorter than that of wildtype siblings (n = 20; p < 0.0001). (F) At 46 dpf, the body weight of homozygous (MZakt2−/−) mutants (akt2ihb504/ihb504) was significantly lighter than that of wildtype siblings (n = 20; p < 0.0001). (G) Pituitary development in homozygous (MZakt2−/−) larvae (5 dpf) was indistinguishable from that of their wildtype siblings, as revealed by pomca staining. (H) Pituitary development in homozygous (MZakt2−/−) larvae (5 dpf) was indistinguishable from that of their wildtype siblings, as revealed by gh1 staining. (I–J) At 10 dpf, the mRNA level of gh1, pomca, sst1.2, sst2 and sst3 in MZakt2−/− mutants compared to wild-type siblings. |
|
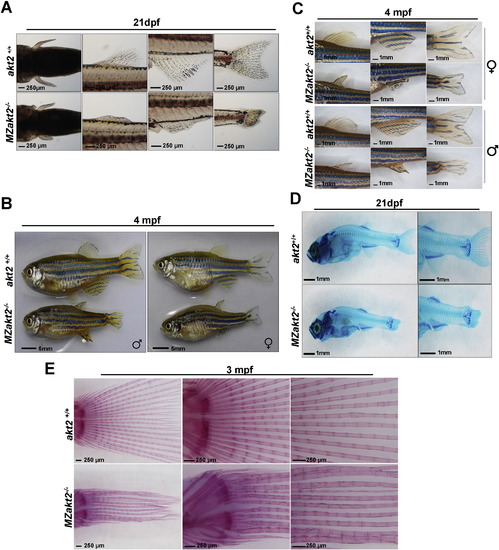
Akt2 mutants exhibit delayed fin generation. (A) At 21 dpf, MZakt2−/− mutants displayed shorter protrusions, aberrant extension, and reduced number of segments of fin rays in pectoral fin, dorsal fin, canal fin, and caudal fin compared with their wildtype siblings. (B–C) At adult stage (4 months), MZakt2−/− mutants displayed delayed fin generation. (D) At 21 dpf, the joint between the muscle and dorsal fin or anal fin, and the connection between the caudal fin and the spine were normal in MZakt2−/− mutants compared with their wildtype siblings, as revealed by Alcian blue staining. (E) Alizarin red-staining of caudal fin from wild-type and homozygous adult zebrafish. Scale bars are shown in the pictures. PHENOTYPE:
|
|
Akt2 mutants display impaired glucose homeostasis. (A) Pancreas development in homozygous (MZakt2−/−) larvae was indistinguishable from their wildtype siblings, as revealed by insulin (endocrine pancreas) and trypsin (exocrine pancreas) staining; try, trypsin. (B) Blood glucose concentration of MZakt2−/− mutants (3-months old; n = 5) was higher than that of their wildtype siblings (3-months old; n = 5) after being fully fed. Zebrafish were fully fed for 15 min, and blood glucose concentration was measured after 8 h. (C) Blood glucose concentration of MZakt2−/− mutants (3-months old; n = 3) was higher than that of their wildtype siblings (3-months old; n = 3) after intraperitoneal injection with glucose solution. Zebrafish were intraperitoneally injected with glucose solution, and blood glucose concentration was measured after 4 h. (D) The mRNA level of insulin in MZakt2−/− mutants (3-months old; n = 6) was much lower than that of their wildtype siblings after being fully fed. Zebrafish were fully fed for 15 min, and total RNA was extracted from heart, muscle, and liver (ventral lobe) after 4 h. (E) The mRNA level of insulin in MZakt2−/− mutants (3-months old; n = 6) was much lower than that of their wildtype siblings after fasting for 2 d. (F) The protein level of insulin in the liver-pancreas of MZakt2−/− mutants (3-months old; n = 6) was lower than that of their wildtype siblings after being fully fed. Zebrafish were fully fed for 15 min, and protein was extracted from the liver-pancreas after 4 h and detected via the ELISA method. (G) The mRNA level of glut1 in MZakt2−/− and Makt2+/− embryos (5 dpf, ♀akt2−/− × ♂akt2+/−) was lower than that of wildtype embryos (5 dpf, akt2+/+ × akt2+/+). (H) Immunofluorescent staining for insulin expression in pancreas of akt2+/+ and akt2−/−; the magnification of images is the same. |

ZFIN is incorporating published figure images and captions as part of an ongoing project. Figures from some publications have not yet been curated, or are not available for display because of copyright restrictions. |
Reprinted from Mechanisms of Development, 143, Zhang, D., Wang, J., Zhou, C., Xiao, W., Zebrafish akt2 is essential for survival, growth, bone development, and glucose homeostasis, 42-52, Copyright (2017) with permission from Elsevier. Full text @ Mech. Dev.