- Title
-
A zebrafish model of inflammatory lymphangiogenesis
- Authors
- Okuda, K.S., Misa, J.P., Oehlers, S.H., Hall, C.J., Ellett, F., Alasmari, S., Lieschke, G.J., Crosier, K.E., Crosier, P.S., Astin, J.W.
- Source
- Full text @ Biol. Open
|
Colitogenic challenge is associated with increased intestinal lymphangiogenesis. (A-C) Lateral images of lyve1:DsRed2;kdrl:EGFP larvae at 7dpf (A), treated with TNBS (B) or DSS (C). Asterisks indicate intestinal lymphatic sprouts (ILSs). A′-C′ shows the left intestinal vasculature and A′′′-C′′′ shows the trunk vasculature in the DsRed channel only. A′′-C′′ are schematic diagrams of arteries (red), veins (blue), lymphatic vessels (green) and ILS (magenta) of A-C. (D-G) Quantification of ILS number (D,E) and total ILS length (F,G) in TNBS (D,F) and DSS (E,G) treated larvae compared with untreated, 5-aminosalicylic acid (5-ASA), and TNBS/DSS+5-ASA (ne20). Error bars, ±s.d. *P<0.05, **P<0.01, ***P<0.001 by one-way ANOVA with Dunnett′s multiple comparison test. DA, dorsal aorta; L-IL, left intestinal lymphatics; PCV, posterior cardinal vein; TD, thoracic duct. Scale bar: 100µm. |
|
Treatment with 5-aminosalicylic acid (5-ASA) reduces TNBS-mediated macrophage recruitment to the intestine. (A-C) Lateral images of the intestine of untreated (A), TNBS-treated (B) and TNBS+5-ASA (C) mpeg1:EGFP larvae at 7dpf. The intestine is outlined with a yellow dotted line. (D-G) Three-dimensional reconstruction of the intestine in untreated (D,F) and TNBS-treated (E,G) mpeg1:EGFP;i-fabp:RFP larvae at 7dpf showing increased recruitment of mpeg1-expressing macrophage lineage cells to i-fabp-expressing intestinal epithelial cells following TNBS treatment. D,E and F,G represent two different angles taken from the same animal. (H) Quantification of macrophages in the intestine of TNBS-treated larvae compared with untreated and TNBS+5-ASA at 7dpf (ne22). (I) FACS analysis of EGFP-positive cell counts from untreated and TNBS-treated 7dpf mpeg1:EGFP larvae (ne3, each replicate contains 30-40 larvae). Error bars, ±s.d. *P<0.05, ***P<0.001, by one-way ANOVA with Dunnett′s multiple comparison test (H) or unpaired t-test (I). Scale bars: 100µm. |
|
Zebrafish macrophages and neutrophils express lymphatic growth factors. (A) RT-PCR of vegfaa, vegfc, vegfd, csf1ra, mpx, cdx1b, and ef1-a from RNA isolated from untreated and TNBS-treated 7dpf zebrafish macrophage-lineage cells. (B) qPCR analysis of vegfc mRNA in TNBS-treated 7dpf zebrafish macrophages relative to untreated 7dpf zebrafish macrophages (n=3). (C) RT-PCR of vegfaa, vegfc, vegfd, csf1ra, mpx, cdx1b, and ef1-a from RNA isolated from untreated and TNBS-treated 7dpf zebrafish neutrophils. The colours in the gel images are inverted and the numbers in brackets indicate reproducibility over three experiments. Error bars, ±s.d. n.s. P>0.05 by unpaired t-test. |
|
The Vegfr pathway is required for zebrafish intestinal inflammatory lymphangiogenesis. (A-C) Lateral images of intestine in 7dpf lyve1:DsRed2 larvae exposed to TNBS+1% DMSO (A), TNBS+2.5nM tivozanib (B) and TNBS+150nM sunitinib (C). Asterisks indicate intestinal lymphatic sprouts (ILSs). (D,E) Quantification of ILS number (D) and total ILS length (E) in TNBS+1% DMSO treated larvae compared with 2.5nM tivozanib, TNBS+2.5nM tivozanib, 150nM sunitinib and TNBS+150nM sunitinib treated larvae (n≥20). Error bars, ±s.d. **P<0.01, ***P<0.001 by one-way ANOVA with Dunnett′s multiple comparison test. L-IL, left intestinal lymphatics; TD, thoracic duct. Scale bar: 100µm. |
|
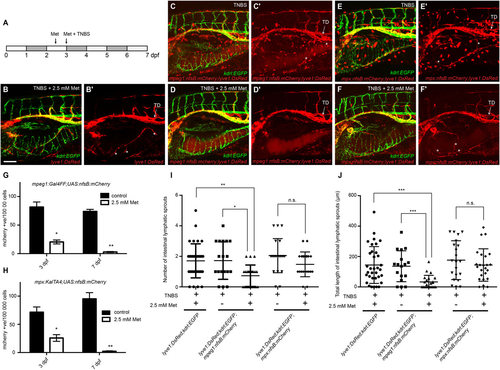
Macrophages are required for intestinal inflammatory lymphangiogenesis. (A) Schema outlining metronidazole and TNBS treatments. (B) Lateral image of the intestine in 7dpf lyve1:DsRed2; kdrl:EGFP larvae exposed to TNBS+2.5mM metronidazole (Met). (C,D), Lateral image of the intestine in 7dpf lyve1:DsRed2;kdrl:EGFP;mpeg1:Gal4FF;UAS:nfsB:mCherry larvae exposed to TNBS (C) and TNBS+2.5mM metronidazole (D). (E,F), Lateral image of the intestine in 7dpf lyve1:DsRed2;kdrl:EGFP;mpx:KalTA4;UAS:nfsB:mCherry larvae exposed to TNBS (E) and TNBS+2.5mM metronidazole (F). (B2-F2) shows expression in the DsRed/mCherry channel only. Asterisks indicate intestinal lymphatic sprouts (ILSs). (G,H) FACS analysis of mCherry-positive cell counts from TNBS-treated (control) and TNBS+2.5mM metronidazole-treated 3 and 7dpf mpeg1:Gal4FF;UAS:nfsB:mCherry larvae (G) or mpx:KalTA4;UAS:nfsB:mCherry larvae (H) (ne3, each replicate contains 30–80 larvae). (I,J) Quantification of ILS number (I) and total ILS length (J) (ne17). Error bars, ±s.d. n.s. P>0.05, *P<0.05, **P<0.01, ***P<0.001, by unpaired t-test (G,H) or by one-way ANOVA with Tukey′s multiple comparison test (I,J). TD, thoracic duct. Scale bar: 100µm. |
|
Treatment with 5-aminosalicylic acid (5-ASA) reduces TNBS-mediated neutrophil recruitment to the intestine. (A-C) Lateral images of untreated (A), TNBS-treated (B) and TNBS + 5-ASA treated (C), lyz:EGFP larvae at 7 dpf. The intestine is outlined with a yellow dotted line. (D) Quantification of neutrophil number in the intestine of larvae treated with TNBS, or TNBS + 5-ASA at 7 dpf (ne25). Error bars, ±s.d. ***P<0.001 by one-way ANOVA with Dunnett’s multiple comparison test. Scale bars: 200 µm. |
|
Addition of 50 nM tivozanib or 3 uM sunitinib prevents the development of trunk and intestinal lymphatics. (A-J) Lateral images showing of 7 dpf lyve1:DsRed2;kdrl:EGFP embryos showing the trunk (A,C,E,G,I) and right intestinal (B,D,F,H,J) vasculature after treatment with 1% DMSO (A,B), 2.5 nM tivozanib (C,D), 50 nM tivozanib (E,F) 150 nM sunitinib (G,H) or 3 uM sunitinib (I,J) at 3 dpf (anterior to the right). (A′-J′) shows expression in the DsRed channel only. (K) Quantification of thoracic duct development in 7 dpf larvae treated with Vegfr inhibitors (ne25). Error bars, ±s.d. n.s P>0.05, ***P<0.001 by one-way ANOVA with Dunnett’s multiple comparison test. R-IL, right intestinal lymphatics; TD, thoracic duct. Scale bars: 100 µm. |
|
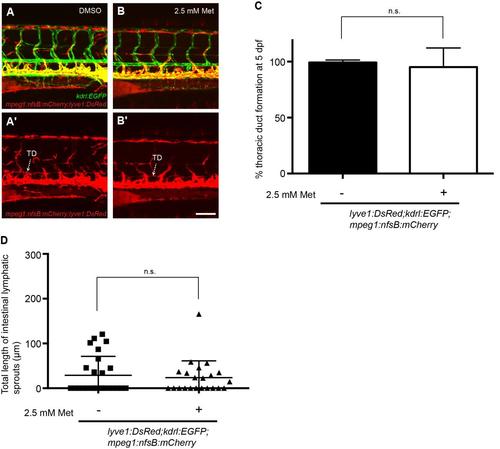
Ablation of macrophage-lineage cells does not inhibit developmental lymphangiogenesis. (A-B) Lateral image of the trunk vasculature in 5 dpf lyve1:DsRed2;kdrl:EGFP;mpeg1:Gal4FF;UAS:nfsB:mCherry treated with DMSO (A) or 2.5 mM metronidazole (B). (A′-B′) shows expression in the DsRed/mCherry channel only. (C) Quantification of thoracic duct development at 5 dpf (ne19). (D) Quantification of ILS development at 7 dpf (n≥22). Error bars, ±s.d. n.s. P>0.05, by Mann-Whitney test (C) or unpaired T-test (D). TD, thoracic duct. Scale bar: 100 µm. |