- Title
-
A Zebrafish Chemical Suppressor Screening Identifies Small Molecule Inhibitors of the Wnt/beta-catenin Pathway
- Authors
- Nishiya, N., Oku, Y., Kumagai, Y., Sato, Y., Yamaguchi, E., Sasaki, A., Shoji, M., Ohnishi, Y., Okamoto, H., Uehara, Y.
- Source
- Full text @ Chem. Biol.
|
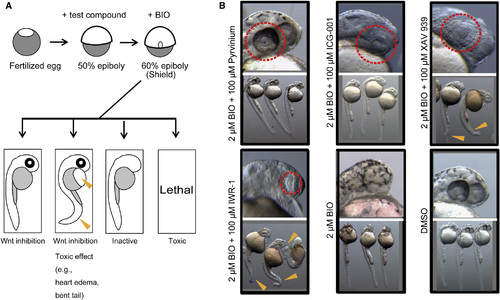
A Zebrafish Assay for Chemical Suppressors Confirms Inhibitory Activities of Known Inhibitors of the Wnt Pathway (A) A schematic diagram of the zebrafish chemical suppressor screening is shown. Chemical suppressors of the eyeless phenotype were identified. Embryos were pretreated with test compounds at 50% epiboly and treated with BIO (a GSK3 inhibitor) at the shield stage, then incubated for 18 to 24 hr. Effects of the compounds were evaluated under a dissecting microscope. When eye development was restored to normal, the test compound was classified as a positive candidate for Wnt pathway inhibitors. When a developmental defect was observed, the adverse effect might reflect potential toxicity of the compound. Arrowheads indicate sites of toxic effects (e.g., heart edema, bent tail). (B) Known Wnt pathway inhibitors restored eye development in the presence of BIO. Pyrvinium (a casein kinase activator), ICG-001 (an inhibitor of interaction between β-catenin/TCF and CREB-binding protein), XAV939 (a tankylase inhibitor), and IWR-1 (an Axin stabilizer) at least partially reversed the BIO-induced eyeless phenotype at 100 µM (dotted lines). Treatments of XAV939 and IWR-1 resulted in malformation in tails (arrowheads). Images were taken at 30 hpf. |
|
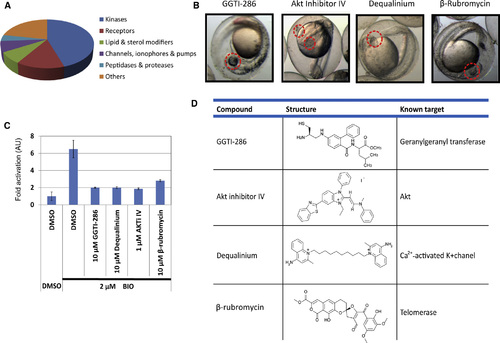
A Zebrafish Chemical Screening Identifies Inhibitors against GGTase, Akt, Telomerase, and K+ Channel as Chemical Suppressors of the Wnt/ß-catenin Pathway (A) Compounds from SCADS chemical libraries were classified into subcategories of the target molecules. (B) The 20 µm GGTI-286, Akt inhibitor IV (AKTiIV), dequalinium, and ß-rubromycin rescued the BIO-induced eyeless phenotype. Images were taken at 30 hpf. (C) GGTI-286, AKTiIV, dequalinium, and ß-rubromycin reduced ß-catenin/TCF-dependent transcriptional activity in CHO cells. Cells were transiently cotransfected with Super 8 x TOPFlash for monitoring ß-catenin/TCF-dependent transcriptional activity and with pRL-CMV for normalizing transfection efficiency. The cells were pretreated with 10µm compounds for 30 min to 6 hr and treated with 2µm BIO for 18 hr. Cells were lysed, assayed for firefly luciferase activity, and then assayed for Renilla luciferase activity. Normalized relative luciferase activities are shown as fold activation to DMSO-treated cells. Values are ± SEM (n = 3). AU, arbitrary units. (D) Structures and known target molecules of the positive compounds are summarized. |
|
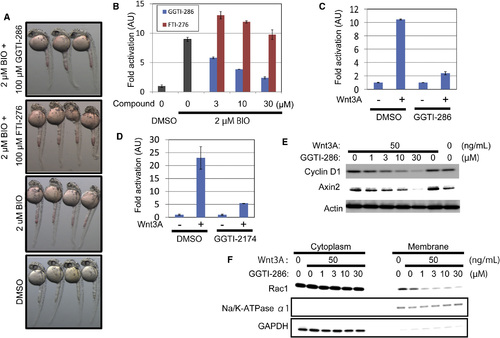
GGTIs Specifically Inhibit the Wnt Canonical Pathway (A) GGTI-286 rescued the eyeless phenotype in zebrafish embryos, but FTI-276, a farnesyltransferase inhibitor, did not. Embryos were pretreated with 100µm GGTI-286 or 100 µM FTI-276 at 50% epiboly, treated with 2µm BIO at the shield stage, and then incubated for another 24 hr. Images were taken at 30 hpf. (B) GGTI-286 inhibited ß-catenin/TCF-dependent transcriptional activity, but FTI-276 did not. CHO cells were transiently transfected with the TOPFlash reporter plasmids. The cells were pretreated with 3, 10, or 30µm GGTI-286 or FTI-276 for 2 hr and treated with 2µm BIO for 18 hr. Firefly and Renilla luciferase activities were measured. Normalized relative luciferase activities are shown as fold activation to DMSO-treated cells. (C and D) GGTI-286 (C) or GGTI-2174 (D) inhibited Wnt3A-induced transcriptional activation of the β-catenin/TCF complex. MDA-MB-231 cells were transiently transfected with the reporter plasmids. The cells were pretreated with 10µm GGTI-286 or 30µm GGTI-2174 for 6 hr and treated with 50 ng/ml Wnt3A for 18 hr. Firefly and Renilla luciferase activities were measured. Normalized relative luciferase activities are shown as fold activation to the absence of Wnt3A. Values are ± SEM (n = at least 3). (E) GGTI-286 diminished expression levels of cyclin D1 and Axin2 in MDA-MB-231 cells. The cells were pretreated with 1 to 30µm GGTI-286 for 6 hr and treated with 50 ng/ml Wnt3A for 18 hr. Cell lysates were analyzed by Western blotting with antibodies against cyclin D1, Axin2, and actin. (F) GGTI-286 reduced membrane localization of Rac1 in MDA-MB-231 cells. The cells were pretreated with 1 to 30µm GGTI-286 for 6 hr and treated with 50 ng/ml Wnt3A for 18 hr. Cell lysates were fractionated into the cytoplasmic and membrane fractions by ultracentrifuge and analyzed by Western blotting with antibodies against Rac1, Na, K-ATPase α1 subunit, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH). |
|
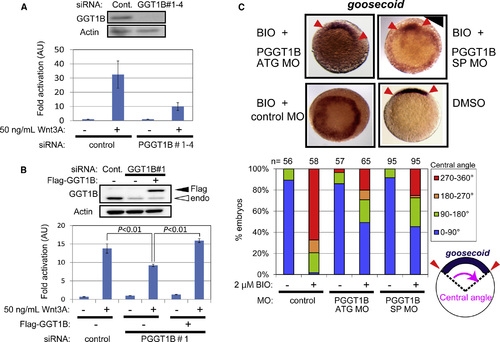
GGTase Is Required for Wnt/ß-catenin Signaling (A) Knockdown of the ß subunit of PGGT1B reduced Wnt3A-induced transcriptional activation of the ß-catenin/TCF complex. MDA-MB-231 cells were transfected with a mixture of four siRNAs for PGGT1B and the reporter plasmids. The cells were treated with 50 ng/ml Wnt3A for 18 hr. Cont., control. (B) Reconstitution of PGGT1B with Flag-tagged PGGT1B reversed PGGT1B knockdown-mediated reduction in transcriptional activation of β-catenin/TCF. MDA-MB-231 cells were transfected with a siRNA targeting the 32 untranslated region (32 UTR) of pggt1b gene, an expression vector for Flag-tagged PGGT1B deleted the 32 UTR, and the reporter plasmids. The cells were treated with 50 ng/ml Wnt3A for 18 hr. Fold activation to the absence of Wnt3A is shown. Values are ± SEM (n = at least 3). PGGT1B expression levels were analyzed by Western blotting with antibodies against PGGT1B and actin. (C) Knockdown of PGGT1B inhibited BIO-induced goosecoid expression in zebrafish embryos. Embryos were injected with antisense MOs that target translation (ATG MO) or splicing (SP MO) of the zebrafish pggt1b gene at the one-cell stage and treated with 30 µM BIO or DMSO for 8 min at 32- to 64-cell stage. At 50% epiboly, embryos were fixed and subjected to in situ hybridization with an antisense goosecoid probe. Embryos are animal pole view, dorsal up if it can be distinguished. Arrowheads indicate tips of the goosecoid expression area. The central angles of goosecoid expression area were measured and scored into four classes: 0° to 90°, 90° to 180°, 180° to 270°, and 270° to 360° (n = at least 56). |
|
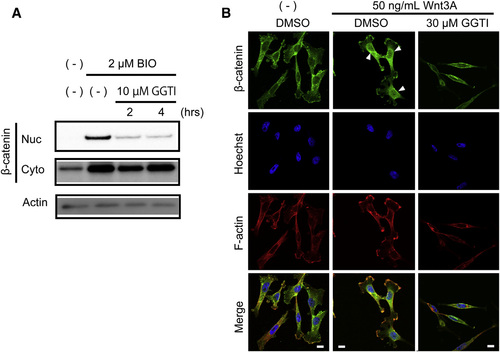
GGTI-286 Inhibits Nuclear Accumulation of ß-catenin (A) GGTI-286 diminished ß-catenin nuclear accumulation. CHO cells were pretreated with 10µm GGTI-286 for 2 or 4 hr, treated with 2µm BIO for 2 hr, and then mechanically lysed and fractionated. The nuclear (Nuc) or cytoplasmic (Cyto) fractions of ß-catenin were analyzed by Western blotting with an antibody against ß-catenin. An anti-actin antibody was used for the loading control. (B) GGTI-286 reduced Wnt3A-induced nuclear accumulation of ß-catenin. MDA-MB-231 cells were pretreated with 30µm GGTI-286 for 4 hr, treated with 50 ng/ml Wnt3A for 18 hr, and then stained with an anti-ß-catenin antibody, Hoechst33342, and phalloidin. Fluorescent images were observed under a confocal microscope. Scale bars represent 10µm. |
|
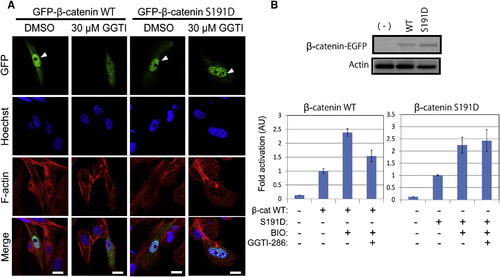
ß-catenin S191D, a Phospho-Mimicking Mutant of ß-catenin, Overcomes GGTI-286-mediated Inhibition in Wnt/ß-catenin Signaling (A) ß-catenin S191D, a pseudophosphorylated mutant of ß-catenin Ser191, reversed GGTI-286-mediated inhibition of β-catenin nuclear localization. CHO cells were transiently transfected with expression vectors for EGFP-fusion ß-catenin wild-type (WT) or S191D. The cells were treated with 30 µM GGTI-286 for 18 hr and stained with Hoechst33342 and phalloidin. Fluorescent images were observed under a confocal microscope. Scale bars represent 10µm. (B) ß-catenin S191D reversed GGTI-286-mediated inhibition of ß-catenin/TCF-dependent transcriptional activation. CHO cells were transiently transfected with expression vectors for β-catenin WT or S191D and the reporter plasmids. The cells were treated with 2µm BIO for 18 hr. Luciferase activities were measured. Normalized relative luciferase activities are shown as fold activation to the absence of BIO. Values are ± SEM (n = at least 3). |
|
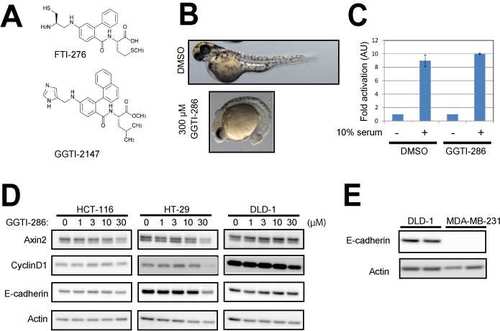
Related to Figure 3. (A) Structures of FTI-276 and GGTI-286 are shown. (B) GGTI-286 reduced the posterior structure of zebrafish embryos. Embryos were treated with 300 µM GGTI-286 at 50% epiboly. Images were taken at 57 hpf (C) GGTI-286 did not inhibit serum-induced AP1 promoter activity. CHO cells were plated in 0.2% FBS/DMEM and transfected with SEAP (secreted alkaline phosphatase) reporter plasmids for AP1. The cells were pre-treated with 10 µM GGTI-286 for 6 hours, and treated with10% FBS for 18 hours. Culture media were assayed for SEAP activities. The normalized relative luciferase activities are shown as fold activation to cells cultured in 0.2% FBS/DMEM. Values are means +/- SEM (n= at least 3). (D) GGTI-286 was less effective on expression levels of Cyclin D1 and Axin2 in colon cancer cell lines with E-cadherin expression. The cells were treated with 1-30 µM GGTI-286 for 18 hours. Cell lysates were analyzed by western blotting with antibodies against Cyclin D1, Axin2, E-cadherin, and Actin. Because these colon cancer cell lines possess activated Wnt/-catenin signaling, Wnt3A stimulation was omitted. (E) MDA-MB-231 cells lack endogenous E-cadherin expression. |