- Title
-
Extra-embryonic syndecan 2 regulates organ primordia migration and fibrillogenesis throughout the zebrafish embryo
- Authors
- Arrington, C.B., and Yost, H.J.
- Source
- Full text @ Development
|
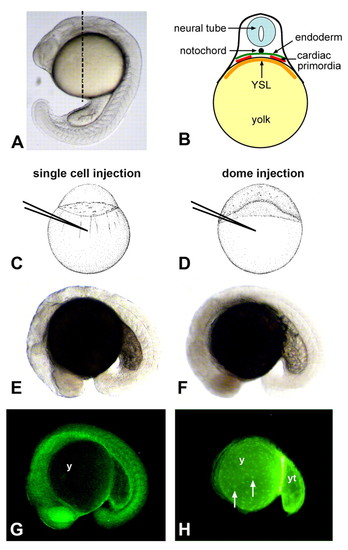
Testing the role of embryonic and nonembryonic cell lineages in cardiac and gut morphogenesis. (A) Lateral view of a zebrafish embryo at the 21-somite stage. Dashed line represents the approximate anterior-posterior location of cardiac primordia at this stage. (B) Diagram represents a transverse section at the level of the dashed line shown in A. Cardiac precursors migrate between the endoderm and the extra-embryonic yolk syncytial layer (YSL) towards the midline. Early gut primordia are positioned analogously but are located posterior to the cardiac primordia. (C,E,G) Morpholino injection into an embryo at the 1-cell stage (C), and light (E) and fluorescent (G) microscopy of a traditional morphant at the 18-somite stage (lateral view). In single-cell injections, fluorescent morpholino is enriched in embryonic tissues and is significantly reduced in the yolk (y). (D,F,H) Morpholino injection into an embryo at the dome stage (D), and light (F) and fluorescent (H) microscopy of a yolkMO embryo at the 18-somite stage (lateral view). In dome injections, fluorescent morpholino is restricted to the extra-embryonic yolk (y), posterior yolk tube (yt) and yolk syncytial nuclei (white arrows). Little, if any, fluorescence is detected within embryonic tissues. |
|
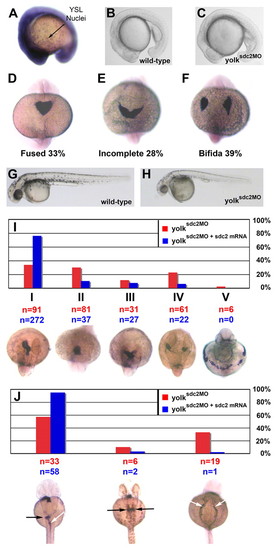
Impaired cardiac primordia and primitive foregut migration in yolksdc2MO embryos. (A) Ubiquitous expression of sdc2 at the 16-somite stage. Strong expression is seen throughout embryonic tissues and within individual yolk syncytial nuclei. (B,C) Lateral views of wild-type (B) and yolksdc2MO (C) embryos at the 18-somite stage. Patterning of yolksdc2MO embryos is relatively normal at this stage. (D-F) Expression of cmlc2 in embryos at the 22-somite stage (dorsal view). Bilateral cardiac primordia were completely fused in 100% of wild-type embryos but only in 33% of yolksdc2MO embryos (D) at this stage. Cardiac primordia migration was defective in yolksdc2MO embryos, with 28% manifesting incomplete heart-field fusion (E) and 39% with cardia bifida (F). (G,H). Lateral views of wild-type (G) and yolksdc2MO (H) embryos at 40 hpf. The overall patterning in yolksdc2MO embryos remains normal, but the embryos are smaller, with a defective posterior yolk tube, larger yolk cell, pericardial edema and decreased pigmentation (H). (I) Bar graph depicting the spectrum of cardiac defects seen in yolksdc2MO embryos (red bars) and yolksdc2MO embryos co-injected with sdc2 mRNA (blue bars) at 40 hpf. Below the graph, the expression of cmlc2 is shown in embryos displaying the spectrum of cardiac phenotypes seen in yolksdc2MO embryos (dorsal view, anterior up for class I-IV embryos; ventral view, anterior up for class V embryo). Cardiac phenotypes vary in severity from left to right and include normal heart tube looping (class I), impaired elongation of the cardiac cone to form the primitive heart tube (class II), incomplete fusion of bilateral heart fields (class III), cardia bifida (class IV) and a band-like distribution of myocardial cell precursors (class V). Numbers of embryos displaying each phenotype are noted in red for yolksdc2MO embryos and in blue for yolksdc2MO + sdc2 mRNA embryos. In rescued embryos, the percentage of embryos with cardiac anomalies decreased from 66% to 24% (P<0.01). (J) Bar graph depicting the spectrum of gut defects seen in yolksdc2MO embryos (red bars) and yolksdc2MO embryos co-injected with sdc2 mRNA (blue bars) at 40 hpf. Below the graph, the expression of foxa3 is shown in embryos displaying the spectrum of gut phenotypes seen in yolksdc2MO embryos (dorsal view, anterior up). From left to right, embryos display normal gut looping (white arrow) and asymmetric hepatic/pancreatic bud formation (black arrow), duplicated hepatic/pancreatic buds (black arrows), and bilateral condensation of the posterior endoderm resulting in the formation of a two-sided foregut (white arrows). EXPRESSION / LABELING:
PHENOTYPE:
|
|
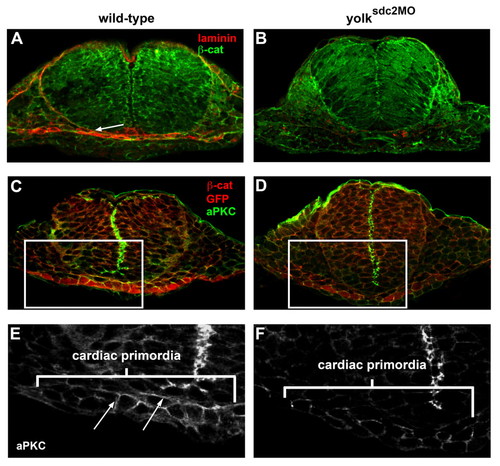
Basement membrane and epithelial cell polarity are disrupted in yolksdc2MO embryos. (A,B) Transverse sections of wild-type (A) and yolksdc2MO (B) embryos at 22 somites reveal laminin deposition (white arrow) between the cardiac and ventral foregut primordia in wild-type embryos (3/3) and reduced laminin in yolksdc2MO embryos (5/6). Laminin, red; β-catenin, green. (C-F) Transverse sections of Tg(cmlc2:GFP) embryos at 22 somites immunostained for the cell-junction protein aPKC (green) and cardiac primordia (red). Magnified views of cardiac precursors (marked by rectangles in C and D) reveal apicolateral localization of aPKC protein (arrows) in noninjected embryos (E) and diminished aPKC in yolksdc2MO embryos (F). EXPRESSION / LABELING:
PHENOTYPE:
|
|
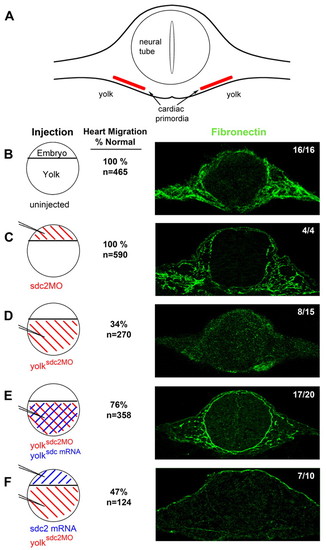
Extra-embryonic Sdc2, but not embryonic Sdc2, is required for fibronectin fibrillogenesis throughout the embryo. (A) Diagram of an embryo cut in cross-section at the level of the cardiac primordia. Fibronectin fibrils form in the extracellular space adjacent to the cardiac primordia and surrounding the neural tube. (B-F) Transverse sections (right) of 22-somite embryos immunostained for fibronectin (green). Circular diagrams (left) represent different injection patterns of sdc2 morpholino (red) and sdc2 mRNA (blue). (B) Uninjected wild-type embryos (16/16 embryos) and (C) traditional sdc2 morphants (4/4 embryos) had normal fibrillogenesis, indicating that embryonic Sdc2 is not required for fibrillogenesis. (D) yolksdc2MO morphants had no fibril formation in 8/15 embryos. With co-injection of sdc2 mRNA into the yolk at the dome stage, fibrillogenesis was rescued in 17/20 embryos (E). By contrast, injection of sdc2 mRNA at the 1-to-2-cell stage, to overexpress Sdc2 in embryonic cells, followed by injection of sdc2 morpholino into yolk at the dome stage (F), did not rescue fibrillogenesis (7/10 embryos). As noted by the numbers adjacent to the confocal microscopy images, a higher percentage of normal cardiac migration correlates with the presence of fibronectin fibrils. PHENOTYPE:
|
|
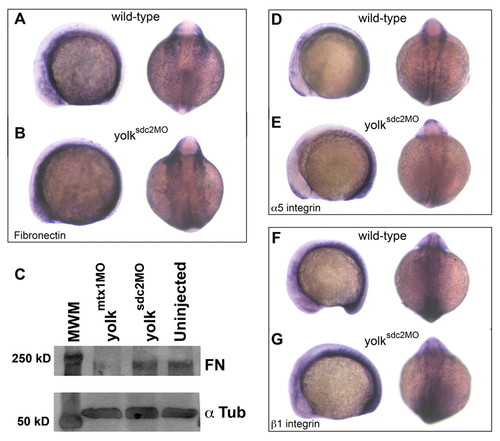
Extra-embryonic Sdc2 controls fibronectin polymerization. (A,B) Lateral and dorsal views of wild-type (A) and yolksdc2MO (B) embryos at the 12-somite stage. fibronectin 1 mRNA expression patterns were normal in yolksdc2MO embryos. (C) Western blot experiment showing normal accumulation of fibronectin (FN) protein monomers in wild-type and yolksdc2MO embryos. yolkmtx1MO embryos have decreased fibronectin monomer accumulation due to decreased transcription of fibronectin 1 (Sakaguchi et al., 2006). α-tubulin (α Tub) was used as a loading control. (D-G) Lateral and dorsal views of wild-type (D,F) and yolksdc2MO (E,G) embryos at the 12-somite stage reveal similar expression patterns for mRNA encoding α5 integrin (D,E) and β1 integrin (F,G). EXPRESSION / LABELING:
|
|
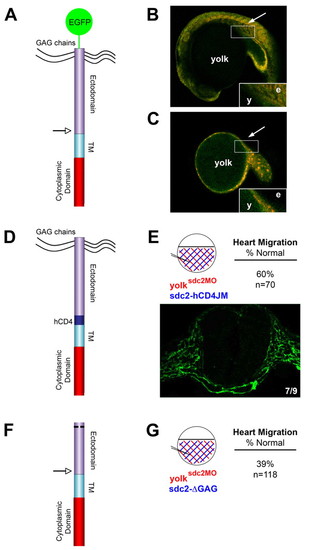
Non-shed extra-embryonic Sdc2 is sufficient for embryonic organ primordia migration and fibrillogenesis. (A) Schematic of the GFP-sdc2 construct with an arrow marking the location of the putative juxtamembrane cleavage site. TM, transmembrane domain. (B,C) Confocal microscopy images reveal GFP (green) expression in embryonic tissue (arrow) with injection of the GFP-sdc2 construct at the single-cell stage (B) but restriction of GFP expression to the YSL/yolk in embryos (n=10) injected at the dome stage (C; arrow shows where embryonic tissue is located). The distribution of co-injected rhodamine-tagged control morpholino (red) correlates with the GFP expression. Insets in B and C show GFP distribution at higher magnification. e, embryo; y, yolk. (D) Schematic of the sdc2-hCD4JM construct. In this construct, 15 juxtamembrane amino acids, including the putative cleavage sequence for proteoglycan shedding, were replaced with a noncleavable sequence from the juxtamembrane domain of human CD4 (hCD4). (E) Co-injection of sdc2 morpholino and sdc2-hCD4JM mRNA into yolk at the dome stage (injection pattern shown in the circular diagram) rescued fibronectin fibrillogenesis in 7/9 embryos (fibronectin fibrils in green) and cardiac migration in 60% of embryos (n=70). (F) Schematic of the sdc2-ΔGAG construct that contains the N-terminal membrane localization sequence but lacks all GAG attachment sites. (G) In contrast to sdc2-hCD4JM, co-injection of sdc2 morpholino and sdc2-ΔGAG mRNA into the yolk at the dome stage did not rescue cardiac primordia migration (normal in only 39% of embryos, n=118). |
|
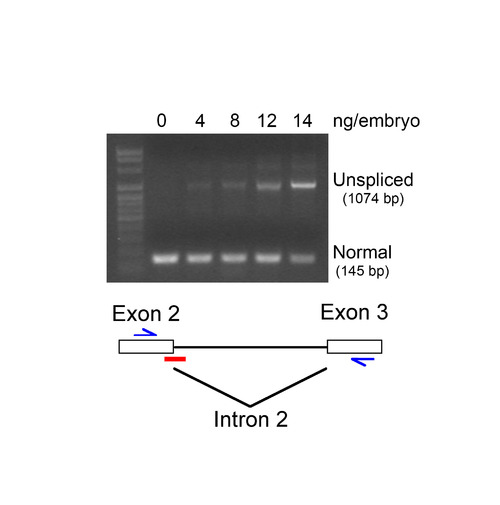
Specificity of sdc2 MO. Embryos were injected at the single-cell stage with sdc2 splice-blocking morpholino (SB MO) at the indicated concentrations. RNAs were extracted and analyzed by RT-PCR. An increased concentration of SB MO correlated with an increased accumulation of intron 2 (929 bp) in sdc2 RNA and a decreased accumulation of sdc2 spliced mRNA (145 bp). Diagram indicates the position of SB MO (red bar) and RT-PCR primers (blue arrows). Retention of intron 2 is predicted to lead to premature translation termination. |