- Title
-
Phospholipase D1 is required for angiogenesis of intersegmental blood vessels in zebrafish
- Authors
- Zeng, X.X., Zheng, X., Xiang, Y., Cho, H.P., Jessen, J.R., Zhong, T.P., Solnica-Krezel, L., and Brown, H.A.
- Source
- Full text @ Dev. Biol.
|
Activation of zebrafish Pld1 in HEK293 cells. Recombinant human PLD1 (hPLD1) or zebrafish Pld1 (zPld1), was transiently expressed in HEK293 cells, and activity was measured, in vivo (A) and in vitro (B). Cells were transfected with an empty pEGFP-C1 vector, or a vector encoding wild-type hPLD1 (hPLD1 wt), or zPld1 wt. In addition, catalytically inactive mutants of hPLD1 K989R and zPld1 K846R were transfected. (A) In vivo activity was measured as the transphosphatidylation of radiolabeled endogenous substrate to 1-butanol, in response to 100 nM PMA compared to basal. (B) In vitro PLD activity was measured using 20 mg total cell lysates for either basal or with the following recombinant protein additions: Arf1 (275 nM), PKCα (1 μM), or Arf1 (275 nM) + PKCα (1 μM) for each transfection condition. Data are expressed as mean ± SEM (*, p < 0.03 when compared to the basal activity of each transfection) of triplicate samples from three independent experiments. (C) Representative Western blot analysis of the transiently expressed N′-GFP tagged PLD1 proteins (GFP-PLD) or GFP vector control (GFP) used in the PLD activity assay. Equal loading of the protein was standardized by GAPDH. |
|
Spatio-temporal expression of pld1 during zebrafish embryogenesis. (A) pld1 transcripts are ubiquitously distributed at 16-cell stage (1.5 hpf). At 11.3 hpf (B, C) and 15 hpf (D, E), pld1 expression is restricted to the dorsal notochord (nc) and the adjacent forming somites (sm). (F–I) pld1 expression in somites (sm) becomes more pronounced during late segmentation stages. (H, I) Cross section of an embryo at 21.5 hpf shows pld1 expression in the somites (sm) in the anterior trunk region (H) but in notochord (nc) in the posterior trunk (I). (J, K) At 24 hpf, pld1 is primarily expressed in the somites (sm). (L) At 2 dpf, the expression of pld1 is strongly reduced in the somites and appears in the pharyngeal pouch (pp). (M, N) At 3 dpf and 4 dpf, pld1 is detected mainly in the liver. (A) Animal view: (B, D) dorsal views, animal pole towards the top; (C,E,F,K,L,M) lateral views, anterior to the left; (G, N) dorsal view, anterior to the left. Animal pole (☆, ★). Vegetal pole (#). Dorsal (D). Scale bar represents 100 μm (H, J) and 250 μm (A, C, E, F, K, L). |
|
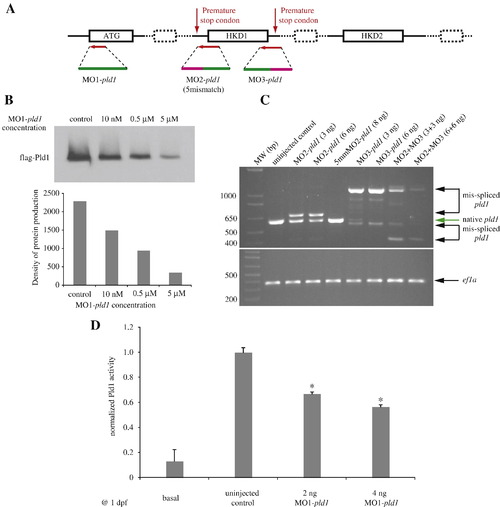
Design and evaluation of effectiveness of antisense oligonucleotides targeting pld1 translation and splicing. (A) The scheme of morpholinos designed to target pld1. (B) in vitro transcription and translation experiment testing the efficiency of MO1-pld1 to inhibit pld1 translation in a dose-dependent manner. (C) Gel electrophoresis of RT-PCR amplified pld1 fragments to monitor interference by MO2-pld1 and MO3-pld1 compared to the normal splicing of pld1. ef1a is used as a loading control. 5mmMO2-pld1: 5mismatch MO2-pld1. (D) The basal and PMA stimulated PLD activity were measured using a stable isotope method for detecting the formation of PtdBuOH by mass spectrometry. Enzyme activity is significantly reduced in MO1-pld1 injected embryos (*, p < 0.01). Data is from three independent experiments. Each condition represents the average measurement from 50 embryos at 1 dpf. Error bars depict SEM. EXPRESSION / LABELING:
|
|
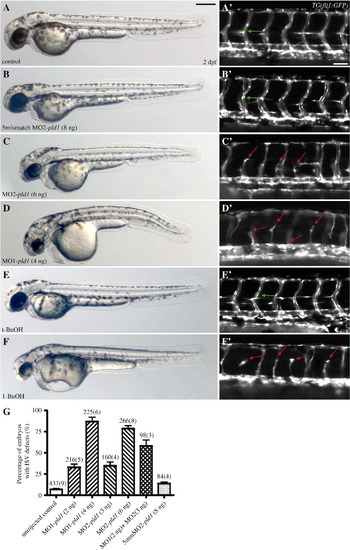
Disruption of Pld1 signaling impairs development of intersegmental vessels. (A,B,C,D,E,F) Nomarski and (A′,B′,C′,D′,E′,F′) fluorescent images of trunk region of TG(fli1:GFP) embryos at 2 dpf. (A,A′) Uninjected control embryos, (B,B′) embryos injected with 8 ng 5mismatch MO2-pld1 and (C,C′) embryos injected with 6 ng MO2-pld1, or (D,D′) 4 ng MO1-pld1, (E,E′) 0.3% t-butanol treated embryo and (F,F′) to 0.3% 1-butanol treated embryo. (A′,B′,E′) Green arrows point to ISVs with normal morphology, whereas (C′,D′,F′) red arrows point to ISV defects, where ISVs stop in the middle along the ventral–dorsal axis or show completely blocked sprouting from the ventral blood vessels (D′,F′). Scale bar represents 20 μm (A′) and 250 μm (A). (G) A bar graph shows the percentage of ISV defects in embryos that were injected with different morpholinos. The total number of embryos is indicated above the bar as well as the number of experimental repeats (in parentheses). PHENOTYPE:
|
|
Effect of Pld1 deficiency on ISV and surrounding tissue development. (A, B) Loss of Pld1 function impairs ISV development. Whole mount in situ hybridization showing expression of tie1 (A) and flk1 (B) at 30 hpf in the trunk region of uninjected control embryos, and embryos either injected with MO1-pld1 or treated with 1-butanol (1-BuOH). Red arrows indicate stunted ISVs. (C–E) Somite muscle and notochord development is relatively normal in embryos with disrupted Pld1 signaling. Representative confocal images of whole-mount immunohistochemistry show the morphology of somites, slow muscle and notochord in MO2-pld1 injected embryos at 2 dpf. Lateral view, anterior to the left. (C) F59 antibody was used to stain slow muscle Myosin. (D) Titin antibody was used to label the organization of muscle cell “Z-discs”. (E) Laminin immunoreactivity at the myosepta and the notochord region in control sibling and pld1 morphants embryos. Scale bar represents 10 μm (D), 20 μm (C, E) and 40 μm (A). |
|
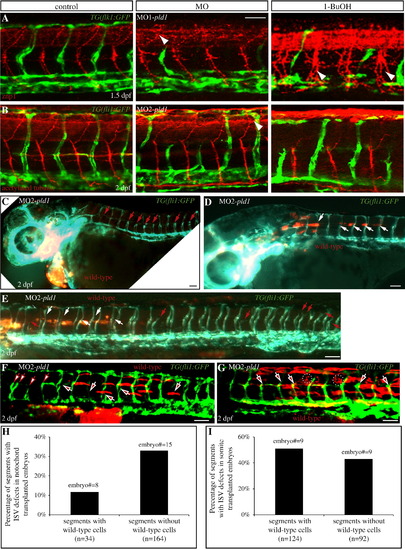
(A,B) Disruption of Pld1 signaling differentially affects ISV and motor axon dorsal–ventral morphogenesis. (A) Uninjected control TG(flk1:GFP) embryos were stained with znp1 antibody to visualize primary motor neurons (red), which extend from the dorsal to the ventral side along with ISV (green). (B) Anti-acetylated tubulin antibody detects both the primary and secondary motor neuron cell body and axons (red) in the trunk region of uninjected control TG(fli1:GFP) embryos, and ISV (green) is positioned in between. Embryos with MO1-pld1 injection or 1-butanol treatment exhibit impaired ISV morphology compared with control embryos; however, all the motor neurons are able to connect from the ventral side to the dorsal side. Interestingly, the motoneurons are more branched when Pld1 signaling is disrupted (white arrowheads), compared to control embryos. (C–E,H) Transplanted wild-type cells in the notochord partially rescue the ISV defects in pld1 morphants. (C–E) Fluorescent images of TG(fli:GFP) pld1 morphants containing Rhodamine-dextran labeled cells (red) transplanted from wild-type donor embryos, and localized in the prechordal plate (C) or anterior notochord (D,E). Lateral view, anterior to the left. ISVs are visualized in green by TG(fli:GFP). White arrows point to normal ISVs in the regions of the chimeric embryos possessing transplanted wild-type cells, whereas red arrows indicate defective ISVs in segments lacking wild-type cells. (H) In chimeric embryos harboring transplanted cells in the notochord, the percentage of ISV defects in segments with wild-type cells (12%, segments number = 34, 8 embryos) compared to segments without wild-type cells (33%, segments number = 164, 15 embryos). (F,G,I) Transplanted wild-type cells in the somites cannot rescue the ISV defects in pld1 morphants. (F,G) Fluorescent images of TG(fli:GFP) pld1 morphants containing Rhodamine-dextran labeled cells (red) transplanted from wild-type donor embryos, and localized in the somites. Lateral view, anterior to the left. ISVs are visualized in green by TG(fli:GFP). White arrows with red filling point to defective ISVs in the regions of the chimeric embryos possessing transplanted wild-type cells, whereas red arrows with white filling indicate defective ISVs in segments lacking wild-type cells. (I) In the chimeras exhibiting transplanted cells in the somites, the percentage of ISV defects in segments with wild-type cells (51%, segments number = 124, 9 embryos) was comparable to segments without wild-type cells (43%, segments number = 92, 9 embryos). Scale bar represents 20 μm (A) and 30 μm (C–G). |
|
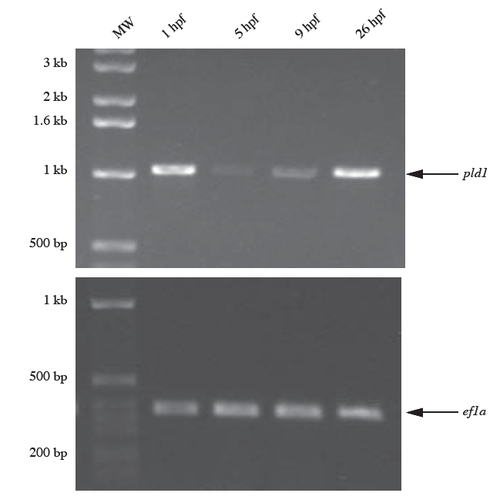
The expression levels of pld1 mRNA detected by RT-PCR at the indicated development stages. ef1a primers were used as a control in RT-PCR reaction. |
|
Loss of Pld1 function disrupts the cranial vessels and blood cells formation. (A) At 48 hpf, Control TG(flk1:GFP)/(gata1:DsRed) embryos show a normal formation of ISV and axis vessels (green) in the trunk region (lateral view) with flowing blood cells in red, and 0.3% 1-BuOH treated embryos (B) show defective ISV formation and slightly reduced axis formation at the similar area, however blood cells are much more strongly affected. At 48 hpf, compared to control embryos (C), major cranial vessels in the head region are mainly formed in MO2-pld1 injected embryos (D), with minor disorganization of CCtA (Cerebellar central artery) (white arrows) and reduction of IOC vessels (Inner optic circle, white dotted circle) in the retinal area. At 12 hpf (6-somite stage), uninjected control embryos (E, group; F, dorsal view) express gata1 bilaterally in the lateral plate mesoderm, however the expression are strongly reduced or absent in MO2-pld1 injected embryos (G, group; H, dorsal view). EXPRESSION / LABELING:
PHENOTYPE:
|
Reprinted from Developmental Biology, 328(2), Zeng, X.X., Zheng, X., Xiang, Y., Cho, H.P., Jessen, J.R., Zhong, T.P., Solnica-Krezel, L., and Brown, H.A., Phospholipase D1 is required for angiogenesis of intersegmental blood vessels in zebrafish, 363-376, Copyright (2009) with permission from Elsevier. Full text @ Dev. Biol.