- Title
-
Crossveinless 2 is an essential positive feedback regulator of Bmp signaling during zebrafish gastrulation
- Authors
- Rentzsch, F., Zhang, J., Kramer, C., Sebald, W., and Hammerschmidt, M.
- Source
- Full text @ Development
|
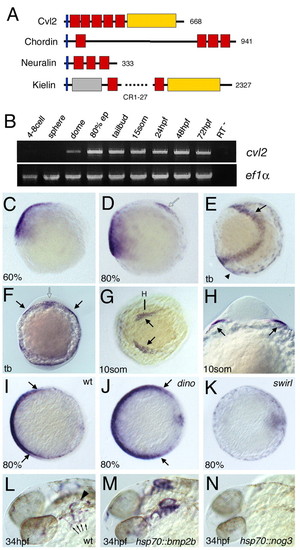
cvl2 is co-expressed with bmps and positively regulated by Bmp signaling. (A) Domain composition of Cvl2 compared with the CR domain proteins Chordin (Sasai et al., 1994), Neuralin (Coffinier et al., 2001) and Kielin (Matsui et al., 2000). Blue boxes indicate signal peptide; red boxes indicate CR domains; yellow boxes indicate vWFd domain; gray box indicates the Thrombospondin domain; numbers one the right indicate total number of amino acid residues. (B) Temporal expression profile of cvl2 determined by RT-PCR. cvl2 is only zygotically expressed. (C-K) Spatial expression pattern determined by in situ hybridization; developmental stage is indicated in lower left-hand corner; genotype of embryos is indicated on the right-hand side. (C-E) Lateral views, ventral leftwards. (F,H) Frontal views, focal plane of H is indicated in G. (G) Animal pole view on head, ventral leftwards. Filled arrows in E-H indicate anterior lateral mesoderm; open arrows in D,F indicate prechordal plate; arrowhead in E indicates ventral tailbud mesoderm. (I-K) Vegetal views, ventral leftwards, with dorsal borders of the expression domain indicated by arrows. (I,J) cvl2 expression is expanded dorsally in chordin mutants. (I,K) Ventral expression is absent in bmp2b mutants. (L-N) Late cvl2 expression at 34 hpf in otic vesicle (arrowhead in L) and pharyngeal pouches (arrows in L) is elevated in hsp70::bmp2b transgenic (M), but downregulated in hsp70::noggin3 transgenic (N) embryos after a 30 minutes heatshock (28°C to 39°C) at 26 hpf. EXPRESSION / LABELING:
|
|
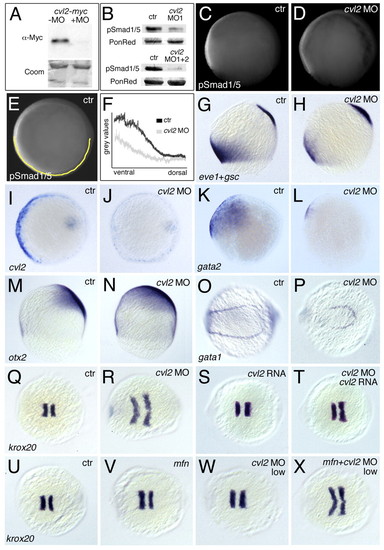
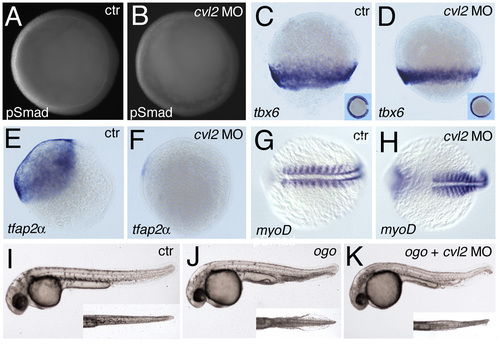
cvl2 is required for ventral specification and Bmp signaling. (A) Western blot; translation of myc-tagged cvl2 mRNA is suppressed by cvl2 ATG-MO. (B) Western blots, revealing strongly reduced pSmad1/5 levels in embryos of 80% epiboly stage after injection of cvl2 MO1 (upper panel) or co-injection of cvl2 MO1+2 (lower panel). (C,D) Whole-mount immunostaining, lateral view at 80% epiboly; cvl2 MO1 leads to a reduction of ventral α-pSmad1/5 immunoreactivity. (E,F) p-Smad1/5 intensity along dorsoventral axis of cvl2 morphant (gray in F) and control sibling (black in F) at 80% epiboly stage. Signal intensities were measured from vegetal views, along a yellow line indicated in E for one of the investigated controls (n=7). (G-X) Whole-mount in situ hybridization with probes indicated in lower left corners. (G-N) Eighty percent epiboly stage. (G,H,K-N) Lateral views; (I,J) vegetal views, dorsal rightwards. Ratios of affected embryos were: eve1, 33/39; cvl2, 66/71; gata2, 31/32; otx2, 45/53. (J) Only the ventral expression of cvl2 is reduced in the morphant, while expression in the prechordal plate is normal. (O,P) Ten-somite stage, view of tailbud, anterior leftwards; expression domain of the blood precursor marker gata1 is reduced in cvl2 morphant (29/37 embryos). (Q-X) Five-somite stage, anterior leftwards; the krox20 expression domains are ventrally expanded in cvl2 morphant (R, 53/68 embryos), but rescued to near wild-type condition in embryo co-injected with cvl2 MO and Drosophila cvl2 mRNA (T, 46/47 embryos). (U-X) Low doses of cvl2 MO1 enhance the mild dorsalization of tolloid/minifin mutants (40/43 in cross of two mfn homozygotes, 22/97 in cross of two mfn heterozygotes), while having no or a much weaker effect in wild-type embryos injected in parallel (0/33). Control embryos are injected with a mismatch cvl2 morpholino. Abbreviations: Coom, Coomassie Blue staining of gel loaded with same amounts of protein; PonRed, Ponceau Red staining of blots after immunoreaction. EXPRESSION / LABELING:
|
|
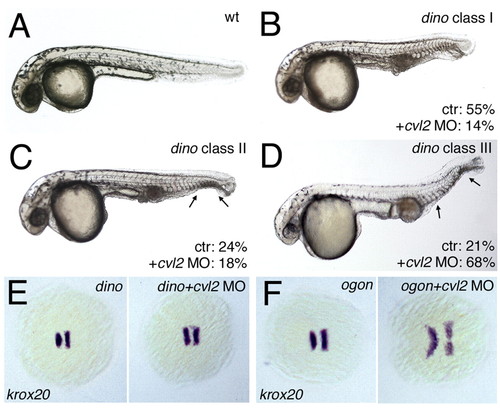
Loss of cvl2 only weakly affects chordin/dino mutants. (A-D) Lateral views of 32 hpf embryos; different classes of dino mutants displaying an increasing loss of ventral tail fin tissue (arrows in C,D). Percentages indicate the fraction of embryos displaying the shown tail fin phenotype among uninjected mutants (n=132) and mutants from the same crosses injected with cvl2 MO (n=215). Injection of cvl2 MO2 increases the percentage of dino mutants with class III phenotype, reflecting a stronger dorsalization within the ventral tissue fin tissue. (E,F) krox20 staining, five-somite stage, animal view, anterior leftwards. cvl2 MO1 clearly expands the neuroectoderm in ogontm305 mutants (F, 41/42 embryos), but only has a weak effect in chordin morphants (E, 55/55 embryos), indicating that ventralized traits of chordin can neither be rescued nor enhanced upon loss of Cvl2 function. EXPRESSION / LABELING:
|
|
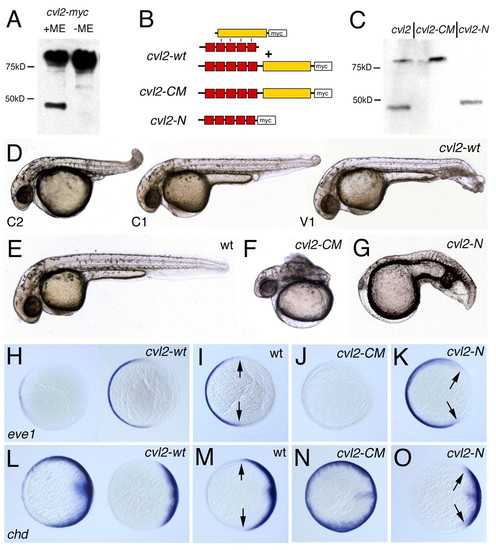
Proteolytic cleavage converts Cvl2 from an anti- to a pro-Bmp factor. (A) Western blot with anti-Myc antibody after SDS-PAGE with embryonic protein extracts of cvl2-myc mRNA-injected embryos. Under reducing conditions [+mercaptoethanol (ME)], full-length Cvl2 and a C-terminal cleavage fragment are detectable. Under non-reducing conditions (without ME), the cleavage products remain associated, and only the high molecular weight band is detectable. (B) Illustration of Cvl2 proteins encoded by the different mRNAs. The protein encoded by cvl2-WT is either cleaved, with the two fragments remaining associated via disulfide bounds of thus far unidentified residues (top), or uncleaved, resembling Cvl2-CM (lower panel). (C) Western Blot showing that Cvl2-CM is not cleaved in embryos, and that Cvl2-N is readily synthesized. (D-G) Lateral views of 32 hpf embryos; (H-O) animal views, dorsal rightwards, after in situ hybridization at 80% epiboly for the ventral marker eve1 (H-K) or the dorsal marker chordin (chd; L-O) (Schulte-Merker et al., 1997). Injection of cvl2-wt mRNA leads to both weakly ventralized (V1 in D and right embryos in H,L) and weakly (C1 in D) or moderately dorsalized embryos (C2 in D and left embryos in H,L). Injection of cvl2-CM mRNA causes strong dorsalization (F,J,N), while cvl2-N mRNA causes ventralization (G,K,O). Arrows in I,K,M,O indicate borders of the eve1 or chd expression domains. Ratios of obtained phenotypes were: (D-G) see Table 1; (H) dorsalization, 17/44; wild type, 22/44; mild ventralization, 5/44; (J) 52/55; (K) 21/43; (L) dorsalization, 21/56; wild type, 29/56; mild ventralization 6/56; (N) 45/48; (O) 21/36. EXPRESSION / LABELING:
|
|
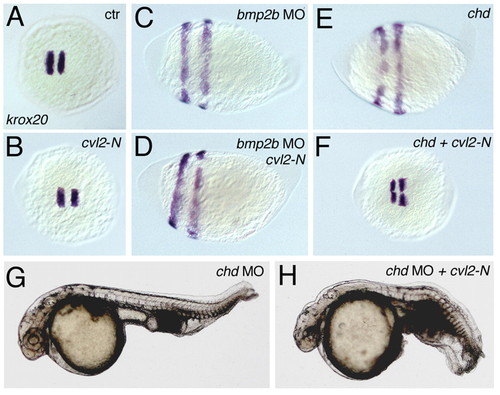
The ventralizing effect of Cvl2-N depends on Bmp2b, but is partly independent of Chordin. (A-F) krox20 staining. (A) Wild type, (B) wild type injected with cvl2-N mRNA, (C) bmp2b morphant, (D) bmp2b morphant injected with cvl2-N mRNA, (E) chd mRNA injected embryo and (F) chd plus cvl-N mRNA injected embryo. cvl2-N mRNA can rescue dorsalization caused by chd mRNA but not by bmp2b MO. (G,H) Lateral views of 32 hpf embryos. The ventralization of (G) chd MO injected embryos is enhanced by co-injection of (H) cvl2-N mRNA. EXPRESSION / LABELING:
|
|
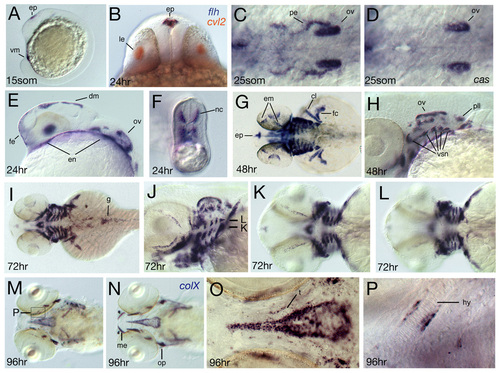
Spatial expression of crossveinless2 at later developmental stages. Developmental stage is indicated in the lower left corner. (A, C-M, O-S) In situ hybridization with a cvl2 antisense probe, (B) double in situ staining with cvl2 in red and floating head (flh) in blue, and (N) in situ hybridization with a collagen X probe. (A,E) Lateral views, (B) frontal optical cross-section showing co-expression of cvl2 and flh in the epiphysis. (C,D) Dorsal view of (C) wild type and (D) casanova mutant. Expression in the pharyngeal endoderm is absent in casanova mutants, (F) optical cross-section of the tail at the level of the yolk extension showing expression in medially migrating neural crest cells, (G,I,K,L) dorsal views, focal planes of K,L are indicated in J. (K) Expression in putative medioventral cartilage condensations (see also J). (L) Expression in more dorsolateral areas that separate the arches (compare also with J). (M-P) Ventral views, expression domains of cvl2 and the bmp target gene collagen X overlap. (O) Higher magnification of the area of the hypophyseal fenestre showing that cvl2 is not expressed in the stacked mature chondrocytes of the trabeculae. (P) Higher magnification of the hyoid arch as indicated in M. cvl2 expression is restricted to tissue surrounding the cartilage. cl, cleithrum; dm, dorsal diencephalic midline; em, eye muscles; en, endoderm; enp, endodermal pouches; ep, epiphysis; fc, finbud cartilage; fe, facial ectoderm; g, gut; hy, hyoid arch; le, lens; nc, neural crest; pe, pharyngeal endoderm; pll, posterior lateral line ganglion; op, operculum; ov, otic vesicle; t, trabeculae; vm, ventral mesoderm; vsn, visceral sensory neurons. EXPRESSION / LABELING:
|
|
Loss of Cvl2 causes dorsalization and rescues the ventral tail fin duplication of ogon/sizzled (ogo) mutants. (A,C,E,G,I,J) Control embryos, (B,D,F,H,K) cvl2 MO injected. (A-H) Immunostainings or whole-mount in situ hybridization with antibodies or probes indicated in lower left corners. (A-F) Eighty percent epiboly stage, dorsal towards the right. (A,B) Vegetal views of embryos also shown in Fig. 2C,D; (C-F) Lateral views; insets in E,F show lateral views of same embryos. (G, H) Ten-somite stage, dorsal views. In the injected embryo, the anterior somites are laterally expanded. (I-K) Live embryos at 36 hpf, lateral views; insets show magnified ventral views on tail. There is duplication of the ventral tail fin in the ogon mutant (J), which is rescued close to wild-type condition (I) after injection of cvl2 MO (K). The small eyes phenotype of the ogon mutant results from brain necrosis, which is most probably not due to the loss of sizzled, but to a thus far unidentified other gene deleted in the used m60 deficiency allele. Accordingly, it is not rescued upon injection of cvl2 MO. EXPRESSION / LABELING:
|