- Title
-
Slow muscle regulates the pattern of trunk neural crest migration in zebrafish
- Authors
- Honjo, Y., and Eisen, J.S.
- Source
- Full text @ Development
|
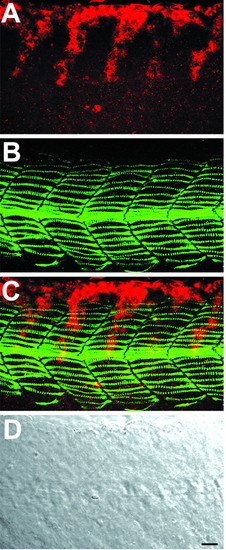
Neural crest migration in wild-type embryos. Whole-mount staining of 21 hpf wild-type embryo with crestin riboprobe (red) and F59 antibody (green) to reveal trunk neural crest and slow muscle cells. (A) Neural crest alone, (B) slow muscle alone, (C) the merged image, (D) a Nomarski differential interference contrast (DIC) image. Neural crest migrates in the middle of the medial aspect of the myotome in one- to two-cell wide streams. Slow muscle fibers extend throughout the anteroposterior axis of each somite. At this stage of development, midtrunk level neural crest cells are in a more medial focal plane than slow muscle fibers, but they are seen together in this z-projection. Similar z-projections are shown in subsequent figures. Lateral view, anterior toward the left. Scale bar: 20 µm. EXPRESSION / LABELING:
|
|
Neural crest cells migrate in an unrestricted pattern in spt mutants and ntl mutants injected with spt MOs. (A-D) Whole-mount staining of 21 hpf spt mutant with crestin riboprobe (red) and F59 antibody (green). (A) Neural crest alone, (B) slow muscle alone, (C) the merged image, (D) a DIC image. F59 antibody staining reveals that spt mutants have few slow muscle cells. Neural crest cells migrate in spt mutants, but migration is not restricted to a specific pathway, thus there are no migration streams. (E-H) Whole-mount staining of 21 hpf ntl mutant injected with spt MOs. (E) Neural crest alone, (F) slow muscle alone, (G) the merged image, (H) a DIC image. F59 antibody staining reveals that ntl mutants injected with spt MOs have no muscle. As in spt mutants, neural crest migrates in these embryos, but migration is not restricted to a specific pathway and there are no migration streams in ntl mutants injected with spt MOs that have no muscle cells, as shown by absence of myod expression (H). Scale bar: 20 µm. EXPRESSION / LABELING:
|
|
Disruption of neural crest migration is correlated with segmentation defects. Whole-mount staining with crestin riboprobe (red) and F59 antibody (green) of bea (A-D; posterior of somite 9) and fss mutants (E-H) at 21 hpf. Streams of neural crest cells (red, A,C,E,G) are present, but less regular than in wild types and the streams show some branching, consistent with abnormal somite shape and size. However, neural crest still migrates generally in the middle of the medial aspect of the myotome in both mutants. (D,H) DIC images of bea and fss mutants, respectively. Scale bar: 20 µm. EXPRESSION / LABELING:
|
|
In segmentation mutants, neural crest migration is disrupted in the same somites in which segmentation is disrupted. Whole-mount staining with a crestin riboprobe of wild-type (A) fss (B), bea (C) and smu mutants (D) at 21 hpf to reveal neural crest cells. (A) In wild-type embryos, neural crest cells migrate in segmental streams along the entire AP axis. (B) Neural crest migration is abnormal from the first somite in fss mutants. (C) In bea mutants, neural crest migration is disrupted posterior to somite 6. (D) In smu mutants there is no clear segmental pattern of neural crest migration; in addition, neural crest cells stall at the level of the dorsal aspect of the notochord. (E,F) EB165 antibody staining reveals that in smu mutants (F) somites are smaller along the DV axis, but a significant amount of fast muscle is still present, although there may be less than in wild types (E). Scale bar: 60 µm in A-D; 25 µm in E,F. EXPRESSION / LABELING:
|
|
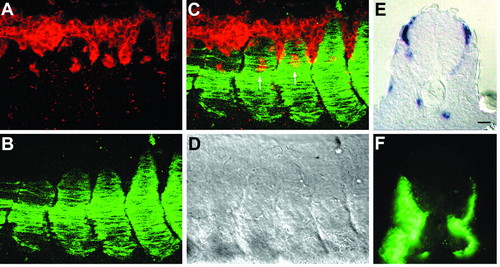
Fast muscle is not essential for neural crest migration on the medial pathway. (A-D) Whole-mount staining of shh mRNA-injected embryo at 21 hpf. (A) crestin RNA expression (red) in neural crest cells. (B) F59 protein localization (green) in slow muscle. (C) Merged image. (D) DIC image. In the absence of fast muscle, the somites do not adhere to one another, creating a somite-free space in the intersomitic cleft. Neural crest migrates in this region, presumably because neural crest cells are motile in the absence of somites. However, neural crest also migrates in normal streams medial to the middle of somites (arrows in C). (E,F) Cross-section of shh mRNA-injected embryo. The somite on the left side of this embryo has no fast muscle and excess slow muscle (all the muscle is slow, as indicated by F59 staining in F); however, neural crest cells (crestin riboprobe, purple in E) have migrated to the same extent as on the right side, where fast muscle is present and slow muscle has migrated to the myotome periphery, as in wild type. Scale bar: 20 µm. |
|
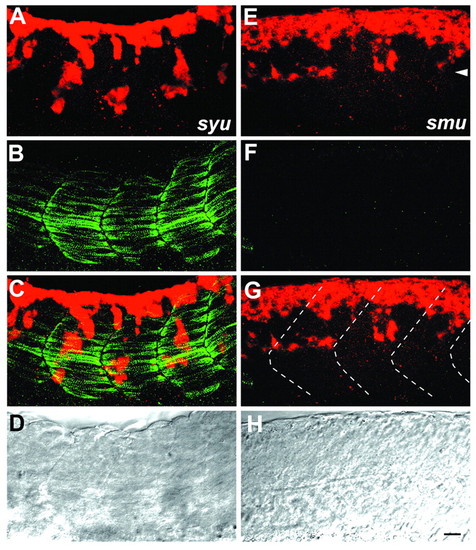
Neural crest migration is disrupted in Hedgehog signaling mutants. Whole-mount staining (A-C,E-G), and DIC image (D,H) of syu (A-D) and smu mutants (E-H) at 21 hpf with crestin riboprobe and F59 antibody. syu mutants have a significant number of slow muscle cells. Consistent with this, neural crest migration is fairly normal, although the streams are less regular than in wild types. By contrast, smu mutants entirely lack slow muscle and neural crest cells are abnormally patterned and stall at the level of the dorsal aspect of the notochord (arrowhead in E). Somite boundaries are shown as broken lines in G. Scale bar: 20 µm. EXPRESSION / LABELING:
|
|
Slow muscle regulates neural crest migration. (A-C) Removal of adaxial cells (black box in A; absence of orange stain) results in disruption of neural crest cell (purple) migration. F59 staining reveals slow muscle cells (orange). (B,C) Higher magnification views of where adaxial cells were removed (B, black box in A) and normal slow muscle cells are present (C, blue box in A). (D) Control experiment in which adaxial cells were removed, but slow muscle cells are present at 21 hpf and neural crest migration is essentially normal. (E) Nomarski DIC image of wild-type embryo shows neural crest migrates in the middle of somite. (F-H) Transplantation of adaxial cells in smu mutants. (F,G) Nomarski DIC images show neural crest migration (crestin riboprobe; purple) and transplanted slow muscle cells (red). Somite boundaries are shown as broken lines. (H) F59 staining (green) of the same embryo shows wild-type adaxial cells. (G) Higher magnification of F. Wild-type adaxial cells transplanted dorsal of the notochord partially restored neural crest migration in the middle of the somite, whereas neural crest migrates on the boundaries of somites without slow muscle. Scale bar: 85 µm in A; 75 µm in B,C; 70 µm in D-F; 40 µm in G,H. |
|
Slow muscle removal results in aberrant DRGs. (A-D) Views of a single 72 hpf embryo: (A,B) control side; (C,D) experimental side. All figures are printed with anterior toward the left and dorsal toward the top for ease of comparison. (A,C) Anti-HuC antibody staining (red) reveals clusters of DRG neurons adjacent to the neural tube. DRGs are indicated only in the first segment in A (arrow). Enteric neurons are indicated with asterisks. (B,D) F59 staining reveals slow muscle cells (green). (A,B) There is a single DRG per somite in most segments, although one segment (open arrowhead) has two DRG neuron clusters. (C,D) Adaxial cells were removed from the experimental side and F59 staining shows less slow muscle. In many segments there are two DRG neuron clusters per somite; the second cluster is often much further ventral than the normal DRG position (arrowheads in C). (E,F) Schematic showing DRG formation in somites 3-10 of five embryos from which adaxial cells were removed from somites on one side; each color represents a different embryo. (E) Control side. (F) Experimental side; the colored lines represent the somites with reduced or missing slow muscle. Scale bar: 20 µm. EXPRESSION / LABELING:
|