- Title
-
The heartstrings mutation in zebrafish causes heart/fin Tbx5 deficiency syndrome
- Authors
- Garrity, D.M., Childs, S., and Fishman, M.C.
- Source
- Full text @ Development
|
Cardiac and pectoral fin abnormalities in hst mutant embryos. (A,C,F,H) Wild-type embryos; (B,D,E,G,I,J) hst mutant siblings. (A,B) Immunostaining with the cardiac-specific antibody MF-20 at 48 hfp in hearts of embryos near completion of looping (ventral views, anterior towards top). (C-I) Live embryos. (C-E) 3 dpf. hst mutant embryos (D,E) have mild to severe pericardial edema and an unlooped, stretched heart. (F,G) 48 hpf. (F) Wild-type embryos display pectoral fin buds with apical fold (arrowheads). (G) Pectoral fin buds are missing in hst mutant embryos (bracket). (H,I) 3 dpf. (H) Wild-type pectoral fins elongate, but (I) hst mutant embryos have still not developed fin buds (brackets). (C-G) Lateral views; (H,I) dorsal views. a, atrium, v, ventricle. EXPRESSION / LABELING:
PHENOTYPE:
|
|
Arrested pectoral fin bud induction in hst mutant embryos. (A,B) Methylene Blue-stained cross sections through 48 hpf embryos. (A) Wild-type embryos have pectoral fin buds (arrows). (B) hst mutant embryos retain small populations of presumptive fin mesenchymal cells (arrowheads) that fail to proliferate. (C) Methylene Blue-stained transverse sections of 3 dpf mutant embryos. Presumptive fin mesenchyme is usually present as a flat patch (arrowhead), but occasionally cells turn perpendicular to the basement membrane (double arrowhead), but do not proliferate further. PHENOTYPE:
|
|
Translational inhibition by tbx5 morpholino. (A-K) Live embryos; (A,C,F,I) wild-type embryos; (B,D,G) embryos injected with 12.4 ng tbx5 morpholino (tbx5-MO); (E,H,J,K) embryos injected with 1.7 ng tbx5-MO. (A,B) Injection of 12.4 ng of tbx5-MO phenocopies heart stretching and pericardial edema of hst mutant embryos, and (C,D) eliminates bud formation at 48 hfp. (E) Injection of 1.7 ng of tbx5-MO delays the onset of bud formation to shortly before 48 hpf. (F,G) 12.4 ng tbx5-MO continues to inhibit bud formation at 3 dpf. (H) Buds of 1.7 ng-injected embryos grow slowly. (I-K) 6 dpf embryos. (I) Wild-type buds elongate to a growing fin. (J,K) 1.7 ng-injected embryos in which the initial buds develop into shorter up-turned fins. (J) Some embryos develop only one bud and hence one fin. (A-E) Lateral views; (F-K) dorsal views. a, atrium; v, ventricle; arrowheads, developing pectoral fin buds; brackets, regions where fin bud failed to develop. |
|
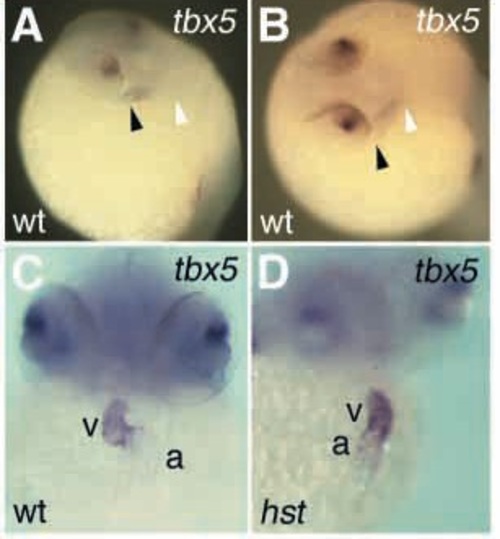
tbx5 expression in the heart tube. (A,B) In situ hybridization of 26 hfp embryos. (A) At 26 hpf, tbx5 expression is detectable first in the presumptive atrium. (B) Upon longer exposure to substrate, tbx5 expression extends through presumptive atrium and ventricle. Heart tubes of wild-type (shown) and hst mutant embryos (not shown) look identical at this stage. (C,D) In situ hybridization of 48 hfp embryos. (C, wild type; D, hst) tbx5 expression is strongest in the ventricle, and weaker in the atrium. a, atrium; v, ventricle; black arrowhead, venous (atrial) end of heart tube; white arrowhead, arterial (ventricular) end of heart tube. |
|
Early cardiac markers are normal in hst mutant embryos at the 15-somite stage. Dual in situ hybridization using: (A) nkx2.5/ntl, (B) hand2/ntl, (C) bmp4/ntl, (D) cmcl2/ntl, (E) vhmc/ntl, (F) ntl alone. The ntl probe indicates the anterior extent of the notochord (arrow in F) (Serbedzija et al., 1998). EXPRESSION / LABELING:
|
|
Myocardial differentiation in hst mutant embryos. (A-D) In situ hybridization with the ventricle-specific marker vmhc (purple) followed by immunohistochemistry with the atrial-specific S46 antibody (brown). (A, wild type; B, hst) vmhc/S46 expression was normal at 33 hpf and (C, wild type; D, hst) at 48 hpf. (E-H) versican expression in the heart. At 33 hpf (not shown), wild-type and hst mutant embryos express versican broadly in the atrium and weakly in the ventricle. (E,F) 48 hpf; (G,H), 72 hpf; (E,G) wild-type embryos restrict versican expression to the AV boundary, but (F,H) hst mutant embryos fail to undergo this transition and continue to express versican predominantly in the atrium, with weak expression in the ventricle. (I-L) bmp4 expression in the heart. (I) Wild-type and (J) hst mutant embryos express bmp4 in the ventricle and inflow tract at 48 hpf. By 72 hpf (K) wild-type embryos restrict bmp4 to the AV junction, but (L) hst mutant embryos retain ventricle-enriched expression. (M-P) versican expression in the developing ear. At 48 hpf, (M) wildtype and (N) hst mutant embryos express versican broadly in the otic placode. By 72 hpf, (O) wild-type and (P) hst mutant embryos both restrict versican to the otoliths (Mowbray et al., 2001), suggesting the heart-specific defects in hst mutant embryos are not due to general developmental delay. a, atrium; av, atrioventricular boundary; i, inflow tract; op, otic placode; ot, otoliths; pf, pectoral fin bud; v, ventricle. EXPRESSION / LABELING:
|
|
The morphology of hst hearts. (A, wild type; B, hst) Methylene Blue-stained transverse sections; hst hearts have not looped, the atrium is stretched and the ventricle slightly smaller than wild type. a, atrium; i, inflow tract; v, ventricle. PHENOTYPE:
|
|
Fin bud development in hst mutant embryos. (A-I) Wild-type embryos, (A′-I′) hst mutant embryos. (A-C′) tbx5 expression in the developing pectoral fin field. At 26 hpf, (A) wild-type embryos restrict tbx5 expression to a circular patch, the presumptive fin field, in the dorsal LPM, but (A′) hst mutant embryos display a more dispersed, less abundant expression. (B,B′) At 32 hpf and (C,C′) at 48 hpf, (B,C) wild-type embryos continue tbx5 expression in the developing bud mesenchyme, but (B′,C′) in hst mutant embryos, tbx5 expression is greatly decreased (B′) at 32 hpf and (C′) undetectable in the LPM thereafter. (D-F′) Expression of molecular markers of early limb bud induction. (D,D′) At 32 hpf, wild-type embryos express shh expression in the pectoral fin mesenchyme, but hst mutant embryos do not. (E,E′) Likewise, at 32 hpf dlx2 is expressed in the apical fold of wild-type but not hst mutant embryos. (F,F′) At 48 hpf, bmp4 is present in the apical fold of wild-type but not hst mutant embryos. (G-I′) hand2 expression in the developing fin field. (G,G′) At 24 hpf, wild-type embryos express hand2 broadly in the region of the LPM encompassing the fin field, but hst mutant embryos express hand2 faintly in only a few LPM cells. (H,H′) At 32 and (I,I′) at 48 hfp, wild-type embryos (H,I) express hand2 in the fin bud mesenchyme, but hst mutant embryos (H′,I′) have no detectable hand2 expression in the LPM. Arrows in H and G′ indicate cells weakly expressing hand2. |
|
Fin development in morpholinoinhibited buds and in hst heterozygous adults. (A-D) Methylene Blue/Azure II-stained transverse sections of pectoral fins at 6 dpf. (A) Wild-type pectoral fin, showing normal development of ventral and dorsal musculature and the endoskeletal disc. (B-D) Pectoral fins of embryos injected with 1.7 ng tbx5-MO show a range of defects, including shortened proximal-distal length and up-turned orientation. (B) Frequently, muscle and endoskeletal disc development appears normal proximally, but is poorly developed in the distal half of the fin. (C) Other fins show greatly thickened endoskeletal disc development or (D) severely stunted growth. (E,F) Pectoral fin development in hst heterozygous adults. (E,F) Alcian Blue/Alizarin Red preparations of adult pectoral fins. Lepidotrichia growth and branching are comparable in (E) wild-type and (F) hst heterozygous adults. Fin shapes and sizes are similar. dm, dorsal musculature; ed, endoskeletal disc; vm, ventral musculature. |

Unillustrated author statements |