- Title
-
Involvement of the Sonic hedgehog, patched 1, and bmp2 genes in patterning of the zebrafish dermal fin rays
- Authors
- Laforest, L., Brown, C.W., Poleo, G., Géraudie, J., Tada, M., Ekker, M., and Akimenko, M.-A.
- Source
- Full text @ Development
|
Induction of shh during regeneration of the caudal fin. shh expression was determined (A,E) 2 days; (B,D,F-I) 4 days, (C) 12 days after amputation. Two fin rays are shown in A and D, three fin rays in B and branches of three fin rays are shown in C. Shh is confined to one or two subsets of cells in individual fin rays on each side of the fin (BD). Shh is expressed in only one subset of cells as soon as a bifurcation is formed (C) or in the lateral-most fin rays of a caudal fin which never form branches (marked with an asterisk in B). (D) The distal part of the fin is slightly tilted; the open triangles and the filled triangles indicate the two subsets of shh-expressing cells on each side of the ray. (E-G) Proximodistal and (H,I) transverse sections of regenerating caudal fins, 2 days (E) and 4 days (F-I) after amputation. The planes of sections are shown in B. (G) Higher magnification of a section comparable to that shown in F with shh-expressing cells delimited by brackets. The scleroblasts can be distinguished from the rest of the blastemal cells by their location and by their more condensed appearance. The region containing shh-expressing cells seems to overlap the limit where lepidotrichial material can be observed in the epidermal-blastemal interface. (H) shh is expressed in a unique broad domain when no bifurcation is forming. (I) shh expression in two subsets of cells on one side of the fin ray precedes the formation of a bifurcation of the lepidotrichia. The large arrows in A, B and E indicate the level of amputation. P (in B) indicates the proximal direction; b, blastema; l, lepidotrichia; e, epidermis; s, scleroblasts. Scale bars, A-D, 80 μm; E,F,H,I, 30 μm; G, 4 μm. EXPRESSION / LABELING:
|
|
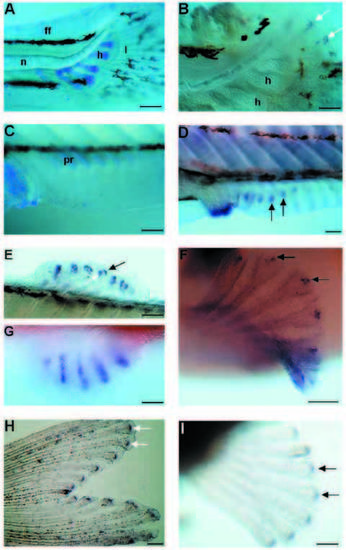
Shh and ptc1 expression in the developing fins of zebrafish larvae. (A,B) Caudal fins of 5.5 mm larvae. (C,D) Anal fins of 6.5 mm larvae. (E) dorsal fin, (F) pectoral fin and (G) pelvic fin of 7.2 mm larva. (A) Alcian blue staining of the cartilage. The lepidotrichia stained in light blue are already well developed in the caudal fin fold. (B) Caudal part of a similar larva to the one in A showing shh expression in each developing lepidotrichia. Note that whole-mount in situ hybridization procedure causes a shrinkage of the tissues. (C) Alcian blue staining of the cartilage of the anal fin. Some elements of the endoskeleton as well as the exoskeletal lepidotrichia are not yet apparent. (D) In a comparable fin to that in C, shh transcripts are already found in the fin fold in subset of cells where the lepidotrichia are forming. Shh is also strongly expressed in the analia-genitalia region. (E-G) Shh expression subsequently appears in dorsal (E), pectoral (F), and pelvic (G) fin rays. The waving of the dorsal fin (E) shows the two groups of cells, one per side, expressing shh. Ptc1 expression in a caudal fin (H) and a pectoral fin (I) of a 7 mm larva. The caudal fin is already heavily pigmented. Arrows in B,D-F and in H,I indicate shh and ptc1 expression, respectively. Anterior is to the left; h, cartilage condensation of hypural bones; l, lepidotrichia; n, notochord; ff, fin fold; pr, proximal radial of the endoskeleton of an anal fin. Scale bars, A, 200 μm; B, 100 μm; C,D,E,G,H,I, 160 μm; F, 80 μm EXPRESSION / LABELING:
|
|
Expression of ptc1 and bmp2 in the regenerating caudal fin. Expression of ptc1 (A-E) was determined at 2 days (A,C) and 4 days (B,D,E) after amputation. (C-E) Proximodistal sections. (C) Two days after amputation, ptc1 is expressed in the basal epithelial layer of the wound epidermis just distal to the stump. (D,E) Four days after amputation, ptc1 is expressed at the same proximodistal level as shh but in 71% of the fins analyzed, it is not only expressed in the basal epithelial layer but also in the adjacent scleroblasts (D); in the remaining 29%, ptc1 is only expressed in a subset of cells of the basal epithelial layer (E). Expression of bmp2 (F-K) was determined at 4 days (F,I-K), 30 hours (G) and 2 days (H) after amputation. G-K are proximodistal sections. (H) Two days after amputation, bmp2 is expressed in cells of the basal epidermal layer just distal to the stump bone on each side of the fin and in adjacent scleroblasts. (I,J) At 4 days after amputation, bmp2 like ptc1 is expressed in 71% of the fins in the basal epithelial cells as well as in the scleroblasts (I); in the remaining 29%, bmp2 transcripts are only found in the epidermal compartment (J). (K) Another site of bmp2 at 4 days is a group of cells invading the space between the newly forming lepidotrichia and the epidermis as indicated by the single arrowhead. The double arrowhead in K indicates the distal domain of bmp2 expression. Arrows indicate the level of amputation. b, blastema; l, lepidotrichia; e, epidermis; s, scleroblasts. Scale bars, A,B, 80 μm; C,D,E,H,K, 30 μm; F, 65 μm; G,I,J, 25 μm. EXPRESSION / LABELING:
|
|
Effect of amputation level on shh expression. (A) Schematized representation of the levels of amputation with respect to the first bifurcation of the caudal fin rays. A “short cut” was done within 1-2 segments after the bifurcation. A “long cut” was made further than 2 segments distal to the bifurcation. The small insert shows the ray fusions induced by the short, but not by the long cut. (B) Representative fusions of caudal fin. Due to the uneven level of the first bifurcations across the fin, some rays underwent a “short cut” (s), whereas some underwent a “long cut” (l). Only rays with short cuts have fused branches. (C-E) Shh expression in “short cut” fin rays at 3 days (C), 4 days (D), and 5 days (E). The cells expressing shh form more diffuse patterns at 3 and 4 days compared to fins that had a long cut (see Fig. 2). The two groups of cells expressing shh, announcing a bifurcation, are clearly resolved at 5 days. The bracket in E indicates ray fusion. Arrows in C,D,E, indicate the level of amputation. Scale bars, 80 μm. EXPRESSION / LABELING:
|
|
Retinoic acid impairs shh and ptc1 expression in regenerating caudal fins. (A-C) Zebrafish were treated with 10-6 M all-trans-RA for a period of 1 day, starting 1 day after amputation. Shh expression was determined (A), 1 day; (B), 3 days; (C), 6 days after the end of the RA treatment. The curve in the caudal fin in A and B is due to the fusion of rays. Shh expression is abolished in treated fins at 1 or 3 days but gradually re-established at 6 days. (D-F) Alcian blue and Alizarin red stainings showing the lepidotrichial matrix and the mineralized bones of the stump, respectively, in half-fins treated with RA (D,E) or in untreated fins (F). Three days after the end of the treatment, the regenerate is developing but there is little lepidotrichial matrix deposition at the tip of each native fin rays (D); in situ hybridization on the second half of this fin shows a weak shh expression (not shown). Five days after the end of the treatment (E), the lepidotrichial matrix does not extend in the regenerate as far as in the control fin (F). Expression of shh (G-I) or of ptc1 (J-L) is abolished by short RA treatments. Zebrafish were treated with 10-6 M RA for 1 hour (H,K), or 4 hours (I,L), starting 3 days after amputation. Expression of shh and ptc1 was determined immediately before RA treatment (G,J); or immediately after the end of the treatment (H,I,K,L). Three days after amputation, shh (G) and ptc1 (J) transcripts are clearly detectable in fin rays, but markedly decreased (H,K) or undetectable (I,L) after 1 and 4 hours of RA treatment, respectively. Arrows indicate the level of amputation; dotted lines in D-F indicate the distal limit of the regenerate. Scale bars, C-L, 80 μm. EXPRESSION / LABELING:
|