- Title
-
Functional interactions of genes mediating convergent extension, knypek and trilobite, during the partitioning of the eye primordium in zebrafish
- Authors
- Marlow, F., Zwartkruis, F., Malicki, J., Neuhauss, S.C.F., Abbas, L., Weaver, M., Driever, W., and Solnica-Krezel, L.
- Source
- Full text @ Dev. Biol.
|
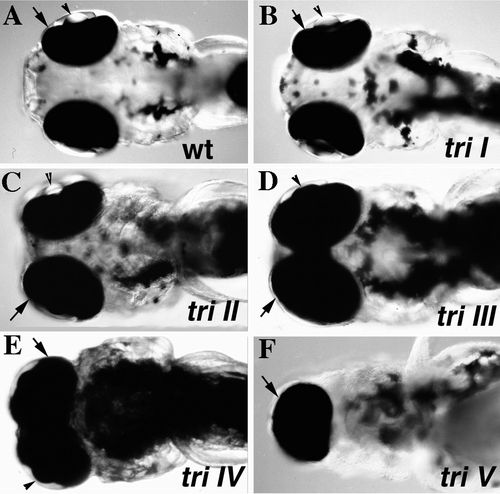
Degrees of cyclopia in trim209 mutants. Micrographs of live embryos at 3 dpf, ventral view of head region. Five classes of the cyclopia phenotype can be distinguished in trim209 mutants. In class I mutants (B) the eye spacing is comparable to that observed in a wild-type sibling (A). In mutants of class II (C) the eye spacing is decreased. (D) In mutants of class III eyes are marginally fused. (E) The eyes are completely fused in mutants of class IV. (F) One cyclopic eye is observed in class V mutants. Retina covered by pigment epithelium (arrow). |
|
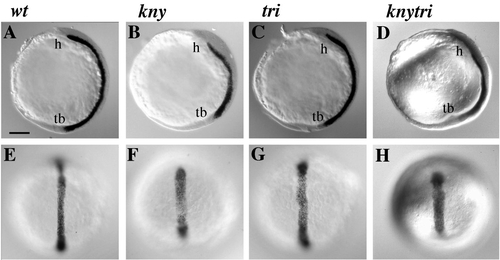
knym119 trim209 double-mutant embryos exhibit additive defects in convergent extension during somitogenesis and 1 dpf, and a synergistic cyclopia defect, compared to single mutants. (A, C, E, G) Dissecting microscope images, lateral view of live embryos at 30 hpf. The length of the embryo is decreased in knym119 (C) and trim209 (E) mutants and dramatically decreased in the double mutant (G) compared to wild type (A). Abnormal shape of the yolk cell and cyclopia are seen in the double mutant (G, inset). (B, D, F, H) Dorsal views of live embryos at 13 hpf using Nomarski optics. The mediolateral width and AP extension of somites (s), notochord (n), and neural keel (nk) are affected in an additive fashion in double mutants (H) compared to knym119 (D) and trim209 (F) single-mutant embryos. fb, forebrain; hb, hindbrain; mb, midbrain; mb/hb, midbrain–hindrain boundary; ye, yolk extension. PHENOTYPE:
|
|
Comparative expression of shh in wild-type (A, E), trim209 (C, G), knym119 (B, F), and kny tri (D, H) embryos at bud stage (10.5 hpf). The shh expression domain is shortened in the AP direction and broadened in the mediolateral direction; this phenotype is more pronounced in knym119 (B, F) and knym119 trim209 (D, H) than in trim209 (C, G) embryos. (A–D) Lateral views, dorsal to the right and anterior at the top; (E–H) dorsal views, anterior toward the top. Tailbud (tb), prospective head region (h). Bar, 120 um. |
|
Abnormal expression of shh gene at 30 hpf is correlated with the cyclopia phenotype in trim209 and knym119 trim209 mutant embryos. (A–D) Lateral views. shh expression in wild-type (A) and knym119 mutants (B) extends to the anterior tip of the embryo. In contrast, the shh expression domain is compressed along the AP axis and fails to extend to the anterior tip of the embryo in cyclopic trim209 (C) and knym119 trim209 (D) mutants. The telencephalon occupies a more dorsal location in trim209 (C) and knym119 trim209 (D) embryos than observed for wild-type embryos (A). (E–H) Dorsal views. shh expression in the hypothalamus is compressed and positioned more posteriorly in trim209 (G) and knym119 trim209 (H) mutant embryos compared to wild-type (E) and knym119 (F) embryos. Arrows, middiencephalic boundary; mb, midbrain; ey, eye; hb, hindbrain; fb, forebrain; fp, floor plate expression in the spinal cord. |
|
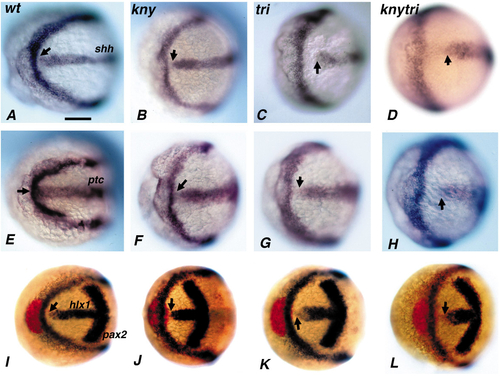
Comparative expression of shh (A–D), ptc (E–H), and prechordal plate markers hgg1 and hlx1 (I–L) relative to dlx3, baseball-stitch band around the anterior edge of the neural plate in wild-type (A, E, I), knym119 (B, F, J), trim209 (C, G K), and knym119 trim209 (D, H, L) visualized by in situ hybridization. The shh expression domain in knym119 (B), trim209 (C), and knym119 trim209 (D) is localized abnormally with respect to the anterior edge of the neural plate at the one-somite stage. This abnormal localization is also observed for ptc1 expression. A gap is observed between ptc1 and dlx3 for trim209 (G) and knym119 trim209 (H) mutant embryos, but not for wild-type (E) or knym119 (F) embryos. In addition, a gap is observed between hlx1 and dlx3 for all embryos including wild type (I–L). hgg1 (red) is positioned anterior to the neural plate in wild-type (I), trim209 (K), and knym119 trim209 (L) embryos and underlies the neural plate in knym119 embryos (J). Arrows indicate the anterior extent of shh (A–D), ptc (E–H), or hlx1 (I–L) expression. Bar, 60 um. |
|
Abnormal morphology of the optic primordia and ectopic expression of pax6 and pax2 genes in the proximal anterior neural plate in knym119 trim209 embryos during the formation of a cyclopic eye. (A, B) Nomarski images of live embryos at the 16-somite stage. In wild-type embryos (A) two separate eye placodes are seen (ey), and the prospective telencephalon (t) and diencephalon (d) are positioned between the eyes at the anterior edge of the neural keel (arrow). In knym119 trim209 mutant embryos (B) one continuous eye placode (ey) is observed with the prospective telencephalon and diencephalon positioned posterior and dorsal to the forming eye. (C, D) pax6 and (E, F) pax2 expression visualized by whole-mount in situ hybridization. pax6 expression in wild-type embryos (C) is detected in distal regions of the anterior neural plate in the future diencephalon and eyes and is absent from the median region of the anterior neural plate (arrow). knym119 trim209 double mutants have ectopic pax6 expression in the median region of the neural keel (arrow), while expression in the prospective diencephalon is compressed and located posterior to the eye field. pax2 in wild type is expressed in two optic stalks (os) separated by the midline; in contrast, one continuous domain is observed in knym119 trim209 double mutants with ectopic expression in the median region of the anterior neural keel (arrow). PHENOTYPE:
|
|
Effects of tiggy winkle (twhh) ectopic expression on eye development in wild-type (A, B) and trim209 mutant embryos (C–E) at 48 hpf. twhh mRNA (0.1 ng) was injected into zebrafish embryos at the one- to eight-cell stage. In injected wild-type and mutant embryos unilateral or bilateral reduction of retina and pigment epithelium was observed. Notably synopthalmia and cyclopia were still observed in affected mutant embryos (D, E). Bar, 63 um. |
Reprinted from Developmental Biology, 203, Marlow, F., Zwartkruis, F., Malicki, J., Neuhauss, S.C.F., Abbas, L., Weaver, M., Driever, W., and Solnica-Krezel, L., Functional interactions of genes mediating convergent extension, knypek and trilobite, during the partitioning of the eye primordium in zebrafish, 382-399, Copyright (1998) with permission from Elsevier. Full text @ Dev. Biol.