- Title
-
Motoneuron fate specification revealed by patterned LIM homeobox gene expression in embryonic zebrafish
- Authors
- Appel, B., Korzh, V., Glasgow, E., Thor, S., Edlund, T., Dawid, I.B., and Eisen, J.S.
- Source
- Full text @ Development
|
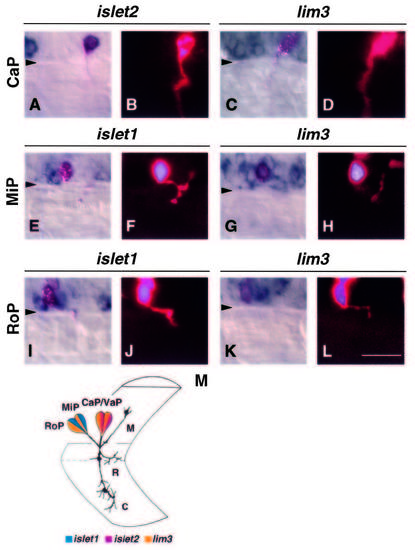
LIM homeobox gene expression in identified primary motoneurons. Primary motoneurons were injected with fluorescent dyes, fixed at 22-24 hours, and hybridized with LIM homeobox RNA probes. The fluorescent signal was enhanced using an alkaline phosphatase-catalyzed colorimetric reaction (Materials and Methods). A, C, E, G, I, K show Nomarski optics images of doubly labeled neurons. B, D, F, H, J, L are fluorescent images of the same motoneurons showing axonal trajectories. Triangles indicate the ventral boundary of the spinal cord. Scale bar equals 25 mm. (A,B) Injected CaP showing hybridization with islet2 probe. (C,D) Injected CaP hybridized with lim3 probe. At this stage, lim3 probe labels more cells than the primary motoneurons in the ventral spinal cord. (E,F) Injected MiP hybridized with islet1 probe. (G,H) Injected MiP hybridized with lim3 probe. (I,J) Injected RoP showing hybridization with islet1 probe. A second cell, dorsal to RoP, was injected but does not show islet1 labeling. Also, RoP is bordered by islet1-labeled cells. At this stage, islet1 begins to label cells in addition to the primary motoneurons (see Fig. 5). (K,L) Injected RoP hybridized with lim3 probe. (M) Schematic representation of primary motoneuron positions within the spinal cord, their axonal projections into the adjacent somite and the combination of LIM homeobox genes each expresses at axogenesis. Yellow represents lim3 expression, blue represents islet1, and red represents islet2. M indicates the MiP axon, R the RoP axon and C the CaP axon. The primary motoneurons were drawn by tracing and superimposing images of cells from different 24 hour embryos from a video display. EXPRESSION / LABELING:
|
|
Early pattern of islet1 and lim3 expression in the neural keel. Dorsal views of neural-keel-stage embryos. At this stage, nascent primary motoneurons form bilateral, discontinuous columns separated by the floorplate (fp). Note that expression patterns are not bilaterally symmetric. (A) Dorsal view of 3-somite-stage embryo probed for islet1 RNA. (B) Dorsal view of 5-somite-stage embryo probed for lim3 RNA. Dotted lines mark approximate somite boundaries. Numbers indicate somite level. Scale bar equals 20 μm. EXPRESSION / LABELING:
|
|
Developmental profile of LIM homeobox gene expression in the trunk ventral spinal cord. Expression of islet2, lim3 and islet1 RNA at similar axial positions at different times of development. All photos were taken at somite levels 6 and 7. Anterior is to the left for each panel. Dotted lines mark approximate positions of somite boundaries. Triangles indicate the ventral boundary of the spinal cord. A, F and K are dorsal views of the neural keel at the 8-somite stage. Remaining panels are lateral views with dorsal at the top. Scale bar equals 20 μm. sc, spinal cord; nc, notochord. (A) islet2 RNA is expressed in 1- 2 irregularly spaced cells per hemisegment at the 8-somite stage. (B) islet2 RNA in 1-2 midsegment cells at the 10- somite stage. (C) islet2 expression in 1-2 midsegment cells at the 12-somite stage. (D) islet2 RNA in 1-2 midsegment cells at the 16- somite stage. (E) islet2 expression maintained in 1-2 midsegment cell pattern at the 19-somite stage. Dorsal labeling, here and in D (one cell), indicates expression in cells that are probably Rohon-Beard sensory neurons, based on their locations. (F) lim3 is expressed in a variable number of cells clustered near somite boundaries in 8-somitestage embryos. (G) Clusters of cells expressing lim3 RNA near somite borders at the 10-somite stage. (H) Approximately three cells near somite borders express lim3 at the 12-somite stage. (I) lim3 RNA expressed in border and midsegment cells at the 16-somite stage. (J) Expression of lim3 in numerous ventral spinal cord cells at the 19-somite stage. (K) islet1 RNA is expressed in 2-3 variably spaced cells per hemisegment in the anterior neural keel of an 8-somite-stage embryo. (L) islet1 RNA detected in one cell near somite boundaries (black arrow) and one midsegment cell (outlined arrow) at the 10-somite stage. (M) Down regulation of islet1 RNA at the 12-somite stage. Single cells expressing high levels of islet1 RNA are seen at the anterior borders of the 6th and 8th somites (black arrows) but not of the 7th (white arrow). Low levels of RNA are seen in midsegment cells (outlined arrows). Labeling in the dorsal spinal cord represents expression in putative Rohon-Beard sensory neurons. (N) High levels of islet1 RNA in single cells at each somite border at the 16- somite stage. (O) islet1 RNA in two cells at each somite border at the 19-somite stage. EXPRESSION / LABELING:
|
|
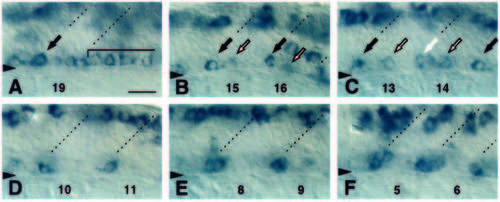
Anteroposterior progression of the islet1 RNA expression pattern in the ventral spinal cord. Panels show islet1 RNA distribution in the ventral spinal cord of a 19-somite-stage embryo at different anteroposterior positions. Anterior is to the left and dorsal is up in all panels. Approximate somite boundaries are indicated by dashed lines. Numbers at the bottom of each panel indicate somite level. Triangles mark boundary between spinal cord and notochord. Scale bar equals 20 mm. (A) Somite level 19 and unsegmented region. High level expression (arrow) at somite 19 boundary. islet1 expression in a nearly contiguous line of cells in unsegmented region (bracket). (B) Somite level 15-16. High level expression in single cells (black arrows) near somite boundaries. Low level expression in midsegment cells (outlined arrows). Dorsal labeling in putative Rohon-Beard sensory neurons. Labeling in segment 16 just dorsal of primary motoneurons represents transient expression in unidentified cells. (C) Somite level 13-14. High level of islet1 expression in single cells at 13 and 15 somite boundaries (black arrows). Low level expression (white arrow) near somite 14 boundary as well as in midsegment cells (outlined arrows). (D) Somite level 10-11. islet1 is expressed in 1 cell at each somite boundary. (E) Somite level 8-9. islet1 is expressed in 2 cells near each somite boundary. (F) Somite level 5-6. islet1 is expressed in approximately 4 cells clustered near the somite boundaries. EXPRESSION / LABELING:
|
|
Simultaneous detection of islet1 and islet2 RNA in primary motoneurons. (A and C) Double labels for islet1 RNA (magenta) and islet2 RNA (blue). (B and D) Corresponding fluorescent images showing low levels of islet1 RNA. Photos are from a 21-somite-stage embryo. Anterior is left and dorsal is up. Dashed lines represent approximate positions of somite borders. Scale bar equals 10 μm. (A,B) At somite level 14, only islet1 is expressed in a cell near the somite border (outlined arrow) and islet2, alone, is expressed in a midsegment cell (white arrow). (C,D) At somite level 17, islet1, and not islet2, RNA is detected in a cell near the somite border (outlined arrow) while both islet1 and islet2 RNAs are detected in two midsegment cells (white arrows). EXPRESSION / LABELING:
|
|
islet2 gene expression in transplanted primary motoneurons determined by in situ RNA hybridization. (A,B) Cells transplanted from the MiP position to the CaP position at different developmental stages and fixed and probed for islet2 expression after the transplanted cells developed axons. Transplanted cells are marked by red staining and islet2 localization by blue. Scale bar in A equals 25 mm for A,B. Scale bar in C equals 25 μm for C,D. (A) This cell was transplanted about 1 hour before axogenesis. It maintained the MiP fate and did not express islet2. Hybridization signal in the ventral spinal cord represents late islet2 expression, probably in secondary motoneurons. (B) This cell was transplanted 2-3 hours before axogenesis, adopted a CaP fate, and expressed islet2. (C) Image of the cell in A prior to fixation showing its MiP axonal projection. (D) Image of the cell in B prior to fixation showing its CaP axonal projection. |